To evaluate the analytical performance and clinical utility of the POC-AFIAS assay in comparison with two ELISA established assays for quantifying serum concentrations of ustekinumab.
MethodsA prospective study was conducted. Consecutive serum samples from adult patients undergoing treatment with ustekinumab were collected. Three analytical techniques were compared for the quantification of ustekinumab serum concentrations: the AFIAS-10® POC assay (POC-AFIAS), the Promonitor® ELISA assay (ELISA-PRO), and the ELISA Ridascreen® assay (ELISA-RDSC). Ustekinumab concentrations were evaluated within three therapeutic ranges: <1μg/mL, 1–4.5μg/mL, and >4.5μg/mL. Statistical analysis included Pearson correlation, intra-class correlation coefficient, and Bland–Altman analysis.
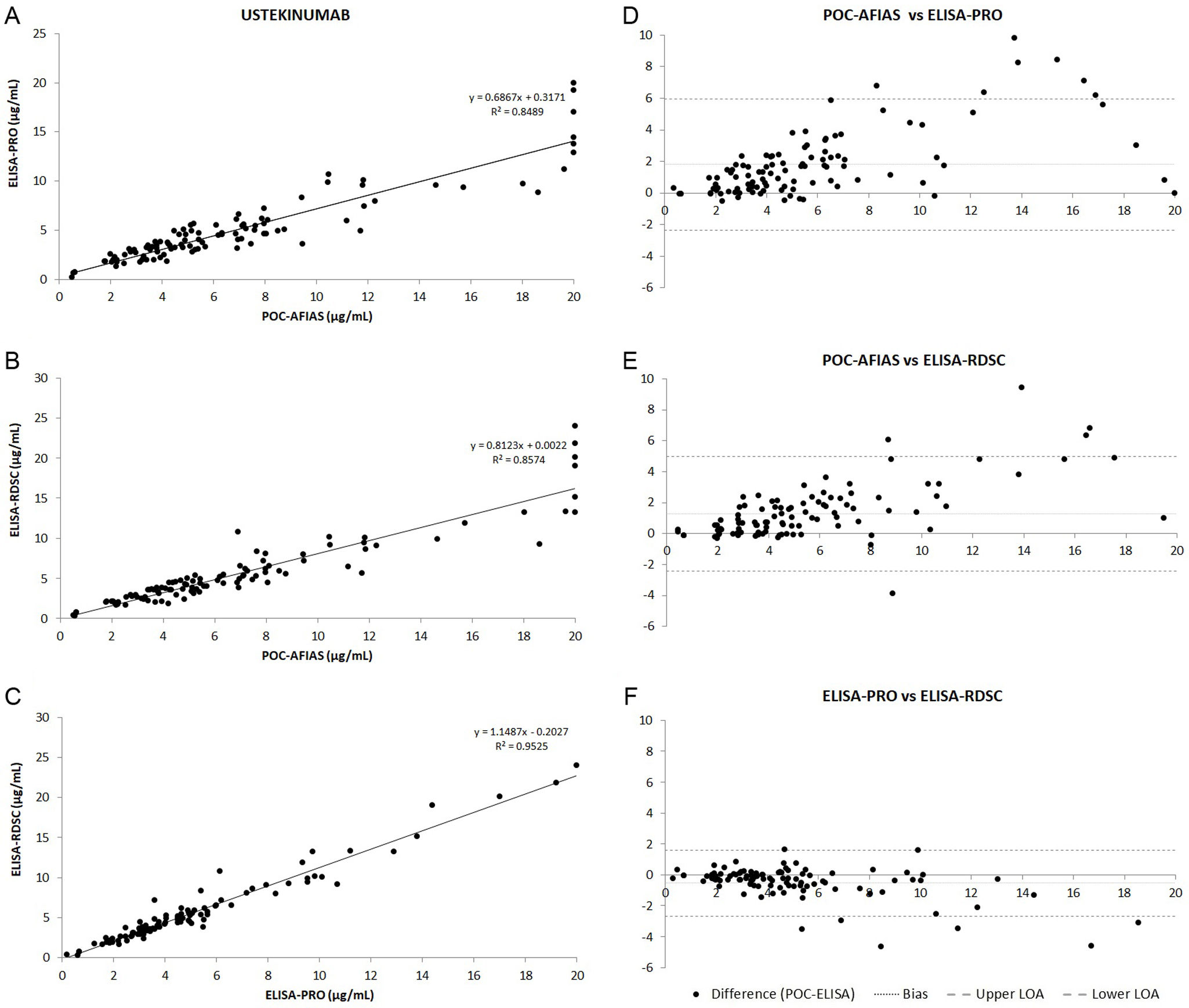
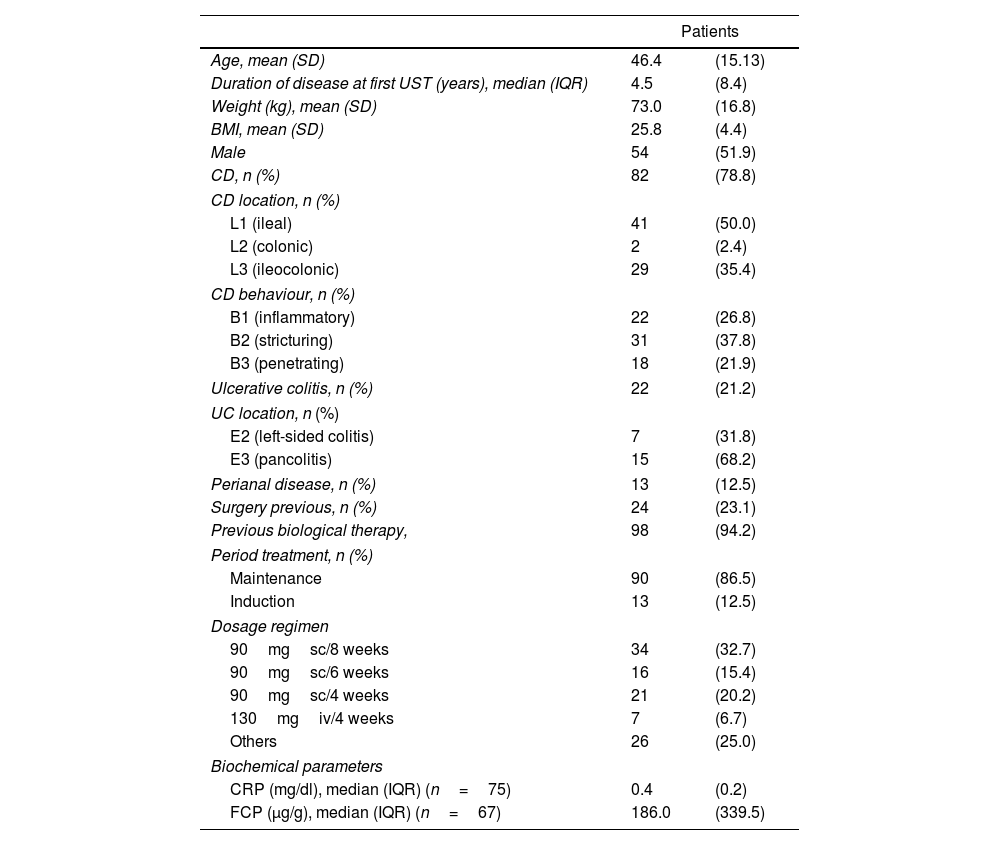
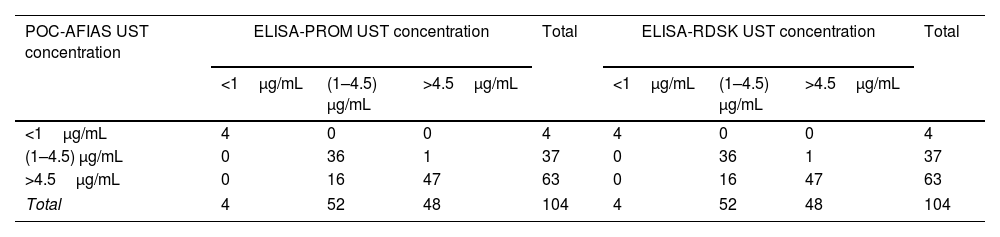
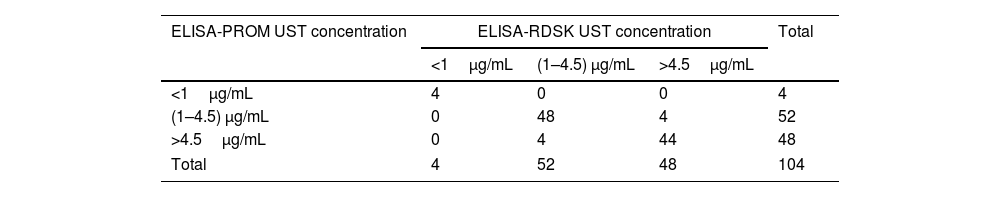
ResultsA total of 104 patients were included in the study. The median ustekinumab concentrations measured were 5.22μg/mL (POC-AFIAS), 3.99μg/mL (ELISA-PRO), and 4.50μg/mL (ELISA-RDSC). Strong correlations were observed between techniques (POC-AFIAS and ELISA-PRO: r=0.921, POC-AFIAS and ELISA-RDSC: r=0.940, ELISA-PRO and ELISA-RDSC: r=0.976). The Bland–Altman analysis revealed a bias difference of 1.81μg/mL between POC-AFIAS and ELISA-PRO, and 1.27μg/mL between POC-AFIAS and ELISA-RDSC. Agreement rates varied by therapeutic range, with the highest agreement observed within the therapeutic range (97.3%) and lower agreement for supra-therapeutic concentrations (74.6%).
ConclusionThis study demonstrated that the POC-AFIAS assay provides comparable results to established ELISA techniques for quantifying serum concentrations of ustekinumab, particularly within the therapeutic range. The findings suggest that the POC-AFIAS assay offers a rapid and effective tool for managing ustekinumab therapy in clinical practice.
Evaluar el rendimiento analítico y la utilidad clínica del ensayo POC-AFIAS en comparación con 2 ensayos ELISA establecidos para la cuantificación de concentraciones séricas de ustekinumab.
MétodosSe realizó un estudio prospectivo en el que se recolectaron muestras de suero de pacientes adultos en tratamiento con ustekinumab. Se compararon 3 técnicas analíticas: el ensayo POC-AFIAS, el ensayo Promonitor® ELISA (ELISA-PRO) y el ensayo ELISA Ridascreen® (ELISA-RDSC). Las concentraciones de ustekinumab se evaluaron en 3 rangos terapéuticos: <1, 1-4,5 y >4,5μg/ml. El análisis estadístico incluyó la correlación de Pearson, el coeficiente de correlación intraclase y el análisis de Bland-Altman.
ResultadosSe incluyeron 104 pacientes en el estudio. La mediana de las concentraciones séricas de ustekinumab fue de 5,22μg/ml (POC-AFIAS), 3,99μg/ml (ELISA-PRO) y 4,50μg/ml (ELISA-RDSC). Se observaron fuertes correlaciones entre las técnicas (POC-AFIAS y ELISA-PRO: r=0,921, POC-AFIAS y ELISA-RDSC: r=0,940, ELISA-PRO y ELISA-RDSC: r=0,976). El análisis de Bland-Altman reveló una diferencia de sesgo de 1,81μg/ml entre POC-AFIAS y ELISA-PRO, y de 1,27μg/ml entre POC-AFIAS y ELISA-RDSC. Las tasas de concordancia variaron según el rango terapéutico, siendo la más alta dentro del rango terapéutico (97,3%) y menor para concentraciones supra-terapéuticas (74,6%).
ConclusiónEste estudio demostró que el ensayo POC-AFIAS proporciona resultados comparables a las técnicas ELISA establecidas para cuantificar las concentraciones séricas de ustekinumab, especialmente dentro del rango terapéutico. Los hallazgos sugieren que el ensayo POC-AFIAS ofrece una herramienta rápida y efectiva para la gestión de la terapia con ustekinumab en la práctica clínica.