Chilblains, also known as perniosis, is a condition that involves the development of inflammatory, erythematous and generally pruritic lesions in acral areas.1 These lesions generally related to marked and sustained vasoconstriction phenomena after exposure to cold have been linked to COVID-19 in the scientific literature.1 Recent studies show that these lesions would not be a specific marker of infection in otherwise asymptomatic patients.1 We report the case of a woman who developed chilblain-like lesions after receiving the first dose of Comirnaty® (mRNA, Pfizer-BioNTech).
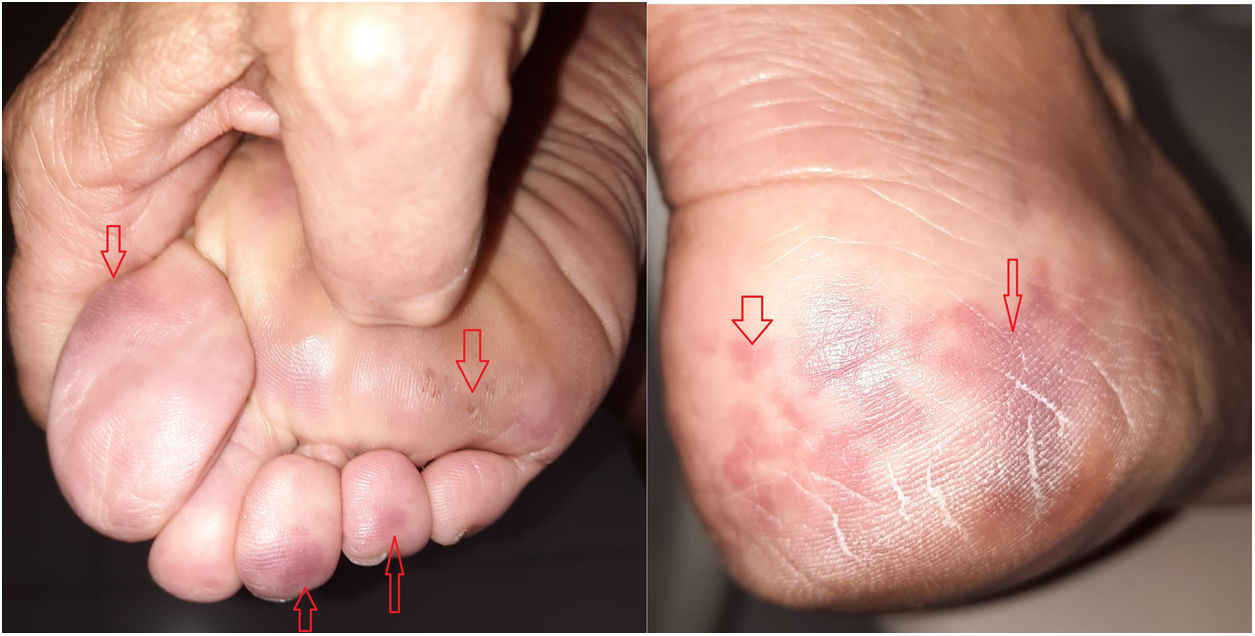
75-year-old woman with diabetes mellitus. She had no other cardiovascular risk factors. No recent changes had been made to her usual medical treatment. She visited the dermatology department for pruritic lesions of more than two weeks’ duration on both feet. The patient reported that these lesions developed five days after receiving the first dose of Comirnaty®. At the time of receiving the vaccine, the patient was asymptomatic and had no symptoms of infection. No similar previous episodes had occurred. Oedematous, erythematous-violaceous papules and plaques located on both feet, mainly on the toes and heel, were observed on examination (Fig. 1). The skin biopsy was compatible with chilblains. Blood tests for erythrocyte sedimentation rate, rheumatoid factor, coagulation and autoimmunity were normal. The diagnosis was chilblain-like lesions, and it should be noted that these lesions emerged after the first dose of the vaccine when the average temperature in Granada was 21 °C. We informed the Pharmacovigilance system and the condition resolved without treatment within three weeks.
The etiopathogenic mechanism by which chilblains occur remains unknown to date. In individuals without chilblains, exposure to cold induces vasoconstriction, which is followed by vasodilation in order to maintain adequate perfusion of the skin, especially in acral areas. Conversely, in affected individuals, cold induces a sustained constriction of the larger skin arterioles and a persistent dilatation of the smaller, superficial vessels, leading to the onset of lesions.1 Therefore, these are much more common in cold climates during winter since it is when the appropriate conditions are given for these vasoconstriction phenomena to occur.1 Numerous cases of COVID-19-associated chilblain-like lesions were reported during the last year, especially in the paediatric population and in adults with severe disease, with viral particles being described in the lesions.1–3 However, recent studies suggest that there is no such relationship and that, therefore, chilblain-like lesions would not be a specific marker of infection in healthy subjects without other symptoms of active infection.1,2 Also, during the last few months of the pandemic, vaccination has been linked to the occurrence of chilblain-like lesions. There are so far nine cases described in the literature following administration of mRNA vaccine vials, four with Moderna and five with Pfizer.2,4 The aetiological mechanism for this relationship has not yet been described, although it is well known that mRNA vaccines are capable of inducing potent type I interferon-mediated reactions and can trigger inflammatory responses involving cytokines.2
The temporal relationship between the administration of the Pfizer vial and the onset of skin lesions during the mild weeks of spring, when the average temperature was 21 °C, together with the absence of a previous history of chilblains, would support the hypothesis of a relationship between vaccination and the onset of chilblain-like lesions in our patient All currently described cases are mild and self-limiting, so they must be known and diagnosed; and the patient should be informed that the administration of the second dose is safe and should not be delayed.2
Conflict of interestsThe authors declare that they have no conflict of interest.
Please cite this article as: Pérez-López I, Gil-Villalba A, Ruiz-Villaverde R. Lesiones similares a perniosis tras vacunación con ARNm frente a COVID-19. Med Clin (Barc). 2022;158:189–190.