Ataxias are characterized by aberrant movement patterns closely related to cerebellar dysfunction. Purkinje cell axons are the sole outputs from the cerebellar cortex, and dysfunctional activity of Purkinje cells has been associated with ataxic movements. However, the synaptic characteristics of Purkinje cells in cases of ataxia are not yet well understood. The nicotinamide antagonist 3-acethylpyridine (3-AP) selectively destroys inferior olivary nucleus neurons so it is widely used to induce cerebellar ataxia. Five days after 3-AP treatment (65mg/kg) in adult male Sprague-Dawley rats, motor incoordination was revealed through BBB and Rotarod testing. In addition, in Purkinje cells from lobules V–VII of the cerebellar vermis studied by the Golgi method, the density of dendritic spines decreased, especially the thin and mushroom types. Western blot analysis showed a decrease in AMPA and PSD-95 content with an increase of the α-catenin protein, while GAD-67 and synaptophysin were unchanged. Findings suggest a limited capacity of Purkinje cells to acquire and consolidate afferent excitatory inputs and an aberrant, rigid profile in the movement-related output patterns of Purkinje neurons that likely contributes to the motor-related impairments characteristic of cerebellar ataxias.
Las ataxias se caracterizan por patrones de movimiento aberrantes estrechamente relacionados con la disfunción cerebelosa. Los axones de las células de Purkinje son las únicas salidas de la corteza cerebelosa, y la actividad disfuncional de las células de Purkinje se ha asociado con movimientos atáxicos. Sin embargo, las características sinápticas de las células de Purkinje en casos de ataxia, aún no se comprenden del todo. El antagonista de la nicotinamida 3-acetilpiridina (3-AP) destruye selectivamente las neuronas del núcleo olivar inferior, por lo que se usa ampliamente para inducir la ataxia cerebelosa. Cinco días después del tratamiento con 3-AP (65 mg/kg) en ratas macho adultas Sprague-Dawley, se reveló una falta de coordinación motora a través de las pruebas BBB y Rotarod. Además, en las células de Purkinje de los lóbulos V-VII del vermis cerebeloso estudiadas por el método de Golgi, la densidad de las espinas dendríticas disminuyó, especialmente las delgadas y en forma de hongo. El análisis por Western blot mostró una disminución en el contenido de AMPA y PSD-95 con un aumento de la proteína α-catenina, mientras que los niveles de GAD-67 y sinaptofisina no cambiaron. Los hallazgos sugieren una capacidad limitada de las células de Purkinje para adquirir y consolidar entradas excitadoras aferentes y un perfil rígido y aberrante en los patrones de salida relacionados con el movimiento, que probablemente contribuya a las deficiencias motoras características de las ataxias cerebelosas.
Ataxias constitute a group of disabling neurological diseases of both genetic and environmental origin that are characterized by errors in the speed, direction, time, and force of movements. Consequently, they affect balance, gait, and coordination.1
Ataxias result from degenerative damage to several neural circuits and brain regions including the cerebellum.2 The cerebellum receives two afferent systems: mossy fibers and climbing fibers. The latter originate in the neurons of the inferior olivary nucleus, reach the Purkinje cells, and establish synaptic contacts with the spines of the proximal region of their dendritic arborization.3
Pharmacological lesions of the inferior olivary nucleus with 3-acetylpyridine (3-AP) have been shown to reliably replicate some of the characteristic motor impairments of cerebellar ataxias.4–6 Some experimental studies carried out using several models of ataxia have reported modifications in various parameters related to the electrophysiological activity of Purkinje cells,7,8 which may be related to the impairment of the firing pattern observed in these neurons after 3-AP treatment.9 These findings suggest abnormal modifications of the translation of excitatory inputs from lesioned climbing fibers to the Purkinje cells.
Dendritic spines regulate and translate afferent information through various local mechanisms, including those of the associative type, which are conditioned by the density of the spines on the surface of the dendrites.10 Genetic studies of various types of ataxias have reported downward variations in both the density of dendritic spines in the Purkinje cells of the cerebellum and synaptic contacts with climbing fibers.11 Additionally, dendritic spines process excitatory afferent information in a differential way depending on their geometric structure and plastic adaptations among them.12 Plastic changes occur due to the functional variability of the afferent synaptic information to the constituent neurons of the brain regions involved, including those that process information of motor meaning.13,14 However, no genetic or pharmacological studies of variations in the proportional density of the different types of dendritic spines in Purkinje cells have been conducted that could relate the hypothetically atypical differential processing of afferent synaptic information to ataxic-like motor impairments.
Thus, we evaluated the density of dendritic spines and the differential proportions of the thin, mushroom, stubby, and wide spines in the proximal arborization of Purkinje cells in the cerebellum of rats treated with 3-AP, as well as the expression of certain molecular markers of synaptic functionality in the cerebellar cortex.
Material and methodsTwentyeight adult male Sprague-Dawley rats (3 months of age) born from 6 dams were used. The rats were maintained under standard conditions: a 12h light–dark cycle (07:00–19:00), a temperature of 22±2°C, humidity at 55%, and water and food ad libitum. The animals were randomly assigned to either the control group (Saline; n=14) or the experimental group (3-AP; n=14).
Ataxia (lack of coordinated movements) was induced in the rats of the experimental group by intraperitoneal injection of a single dose of the neurotoxin 3-AP (65mg/kg of body weight, i.p.), followed 3.5h later by a single dose of nicotinamide (300mg/kg, i.p.).4 The control rats received 0.25ml of saline solution at the same times.
Based on an acute design,12,18 five days after the corresponding pharmacological treatment, both the Saline and 3-AP rats were evaluated behaviorally in a single 4-min session using the Basso-Bresnahan-Beattie (BBB) test16 in a 1.20m×1.20m open field. The BBB scale measures incremental scores, where 0 represents the complete absence of spontaneous movement and 21 a normal gait. It considers movements of the hip, knee, and ankle joints, as well as the ability to stand on both legs, the plantar position on the floor, taking steps, tail posture, and coordination. The rats were videorecorded and assessed afterwards by two independent blinded observers.
Thirty minutes after finishing the assessment with the BBB functional scale, the rats were evaluated on the Rotarod using 2 habituation procedures: first, for 15min prior to the experiment, the rats were kept in their cages in the area where the tests were to be conducted; second, each rat was placed on the stationary Rotarod for 2min. This assessment was conducted in a single session that consisted of 3 trials with a 15-min interval between them. At the beginning of the first trial, the rats were placed in an individual lane of the Rotarod and the device was turned on at a constant speed of 4rpm. We verified that they were able to walk on the Rotarod for approximately 5seconds. During testing, the Rotarod was programmed for a constant acceleration that increased from 4 to 40rpm over 5min. We recorded the latency to falls and the rotation speed (rpm) at which the animals fell off the device. After each trial, all surfaces of the Rotarod were cleansed with 70% ethyl alcohol. The data obtained from the three trials were averaged for the latency to falls and rotation speed.
Two animals per group were anesthetized with ether and given intracardiac perfusion to fix their brain tissue. The washing solution was prepared with phosphate buffer (pH 7.4, 0.1M), 1000IU/l of sodium heparin as an anticoagulant, and 1g/l of procaine chloride as a vasodilator. The fixing solution was made up of 4% formaldehyde in phosphate buffer. Once prepared, 200ml of each solution were perfused at a rate of 40ml/min. The encephalon was removed and post-fixed in 4% formaldehyde in phosphate buffer for 24h and then the cerebellar vermis was removed for further sagittal sectioning. These tissue fragments were processed and embedded in paraffin, and serial 10-mm thick sections were obtained and stained with cresyl violet.
At the conclusion of the behavioral study, six rats per group were selected at random for the analysis of their dendritic spines. They were anesthetized with 100mg/kg of ketamine and 15mg/kg of xylazine, i.p., then perfused with 200ml of a phosphate buffered saline washing solution (PBS: pH 7.4, 0.01M) containing 1000IU/l of sodium heparin as an anticoagulant and 1g/l of procaine hydrochloride as a vasodilator. Subsequently, they were perfused with 200ml of a 4% formaldehyde solution in PBS. Both solutions were delivered at a rate of 40ml/min. The brains were removed by craniotomy and fixed for 48h in 100ml of fresh fixative solution.
A block of tissue from the cerebellar vermis containing lobules V-VII was removed and impregnated according to a modified Golgi method.17 The density of the spines was quantified in six Purkinje cells per animal, from lobules V–VII of the cortex of the cerebellar vermis. For this procedure, a total length of 50μm from 3–4 terminal dendritic branchlets stretching from the proximal arborization to the soma was considered. The neurons analyzed had to satisfy four morphological criteria: (a) located exclusively in lobules V–VII; (b) well-impregnated with no evidence of incomplete impregnation; (c) a fully visible cell body and branches, not obscured by non-specific precipitates, blood vessels, glia, or heavy clusters of dendrites from neighboring impregnated cells; and (d) a fully impregnated, mostly intact, arborization, with no truncated or obstructed branches. The density of the spines and the proportional density of the thin, mushroom, stubby, and wide spines were determined according to previously established criteria.12,18 Thin spines were defined as those in which the neck diameter was smaller than the total spine length, and the head diameter was larger than the neck diameter. Mushroom spines were defined as those in which both the diameter and length of the neck were shorter than the head diameter, and the length of the neck was shorter than that of the head. Stubby spines were defined as those in which the neck and head were indistinguishable, but the diameter of the spine was longer than, or equal to, its length. Wide spines closely resemble stubby spines but were defined as those in which total spine length was longer than diameter. Spine counting was performed by direct observation at 2000× using a magnification changer coupled to a light microscope with a 100× APO lens. The geometrical structure of the different spine types was verified using an image processor (LAS 4.0; Leica Microsystems, Ltd.).
For Western blot studies, protein extracts were obtained from 6 rats per group that were sacrificed with a lethal dose of sodium pentobarbital delivered i.p. After decapitation, the brains were extracted by craniotomy, and lobules V–VII from the cerebellar vermis were obtained under a stereoscopic microscope. All tissue samples were stored initially in Eppendorf tubes at −20°C. After recording the fresh tissue weight, 920μl of lysis regulator (100 mMTris [pH 7.4], 150 mMNaCl, 1mM EGTA, 1mM EDTA, 1% Triton X-100 and sodium deoxycholate 0.5%) and 80μl of protease inhibitor solution (completeTM; Sigma–Aldrich, 05 056 489 001) were added per 100mg of tissue. The tissue was homogenized using a sonicator and the samples were centrifuged for 30min at 4°C and 14,000rpm to remove cell debris. The supernatant was recovered and the amount of total protein for each tissue sample was determined using the Lowry method. From each extract, 25μg of total protein were denatured for 5min at 95°C in 5μl of sample buffer (0.125M HClTris, 4% SDS, 20% glycerol, 10% beta-mercaptoethanol, and a 0.1% bromophenol blue solution). The denatured total protein extracts were resolved on 12% polyacrylamide gels at 125V for 150min in a Tris-glycine buffer (25mM Tris–HCl, 192mM glycine and 0.1% SDS [pH 8.3]: Cat. 1,610,723; Bio-Rad Laboratories, CA, USA). The proteins were electrotransferred to a nitrocellulose membrane at 25 v for 120min in transfer buffer (Tris–HCl 25mM, 250mM glycine, and 20% methanol [pH 8.3]). The membranes were immediately incubated for 1h with blocking solution (5% fat-free milk in 0.01M PBS) to impede non-specific antibody binding, and washed three times for 5min each with PBS (PB saline)–0.05% Tween under constant stirring. The membranes were probed for 24h with the corresponding primary antibodies: anti-GAD-67 (AB5406), anti-synaptophysin (Pierce, PA1-1043), anti-AMPA (AB31232), anti-PSD-95 (AB76115), and anti-α-catenin (AB184917). After washing five times in PBS-Tween-20 (0.05%), the membranes were probed for 2h with a biotinylated goat anti-rabbit IgG secondary antibody (1: 1000, BA 1000; Vector Laboratories), followed by five more washes (in PBS-Tween-20 0.05%). Antibody binding was detected using the ABC Elite kit (PK6100; Vector Laboratories) and visualized with diaminobenzidine (D5905; Sigma). Protein expression was assessed using free ImageJ software (Wayne Rasband, National Institutes of Health, USA, version 1.50e) The data obtained were normalized and are reported as the percentages of the normalized area relative to the internal control (β-actin)19 and presented as the mean of at least six independent experiments.
Results from the Saline and 3-AP groups were compared and analyzed with a Student t test for independent samples. Data were analyzed with SPSS Statistics 24 software.
All experimental procedures were carried out to ensure the least possible pain and distress to the animals, following the NIH guidelines for the Care and Use of Laboratory Animals (NIH Publication No. 8023, revised 1978). The experimental protocol was approved by the Research Ethics Committee of the Mexican Social Security Institute (R-2017-1305-5).
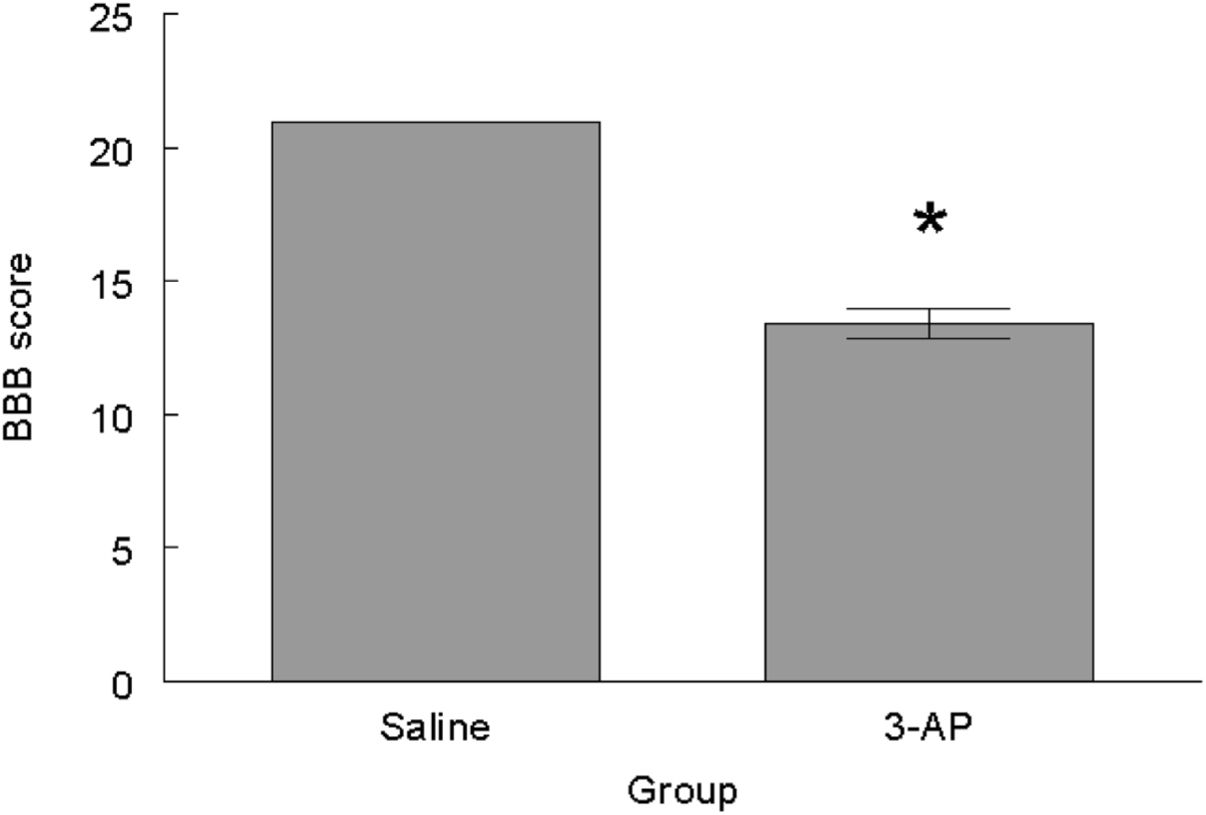
ResultsBBB testing revealed that the 3-AP group scored significantly lower than the saline control group (t=10.827, P<0.0001) (Fig. 1). With respect to the Rotarod testing, the experimental 3-AP group showed shorter latencies to falls from the rod (t=2.827, P<0.01) and lower rotation speed (t=2.885, P<.01) than the Saline control animals (Fig. 2).
Light microscopy of the lobules of the cerebellar cortex from the two groups showed a relatively uniform structure with a single row of Purkinje neurons between the molecular and granule layers. However, in the 3-AP-treated rats the alignment of the Purkinje cells was disturbed, showing irregular, scattered somas, some of which appeared to be hyperchromatic and shrunken, suggesting pyknosis. The saline controls showed normal Purkinje cell morphology and their alignment also appeared normal (Fig. 3).
Representative photomicrographs of the cerebellar cortex stained with cresyl violet. The spherical cell bodies of the Purkinje cells in the Saline control rats (A) are regularly aligned in the Purkinje cell layer (Pl) between the granular (gl) and molecular (ml) layers. In the 3-AP-treated rats (B), the Purkinje cell somata were shrunken, hyperchromatic, and scattered (arrows) within the Purkinje cell layer. Scale bar=150μm.
There was a lower density of dendritic spines on the proximal arborization of the Purkinje cells in lobules V-VII of the vermal cerebellar cortices of the rats in the 3-AP group compared to the Saline control animals (t=8.220, P<0.0001) (Fig. 4). The 3-AP rats also had fewer thin (t=5.239, P<0.0001) and mushroom (t=3.329, P<0.008) spines than Saline controls (Table 1).
Dendritic spines were counted in branchlets (arrows) in the proximal half (dotted line) of the dendritic arbor of the Purkinje cells in lobules V–VII of the cerebellar vermis. Scale bar=150μm. The 3-AP-treated rats had fewer spines than those in the Saline group. Mean±S.E.M. P<0.05.
Proportional density of the spine types (arrows; A: thin; B: mushroom; C: stubby; D: wide) counted in the proximal dendritic arbor of Purkinje cells from the cerebellar vermis of the control (Saline) and experimental (3-AP) groups of rats. Scale bar: 3μm.
| Group spine type | Saline | 3-AP |
|---|---|---|
| Thin | 63.1±1.0 | 54.9±1.1* |
| Mushroom | 46.4±0.5 | 42.2±1.1* |
| Stubby | 14.6±0.4 | 14.4±0.5 |
| Wide | 13.0±0.5 | 12.2±0.7 |
Mean±SEM, P<0.05.
GAD-67 (t=1.757, P<0.1) and synaptophysin (t=−0.077, P<0.9) content in the cerebellar cortex of the 3-AP animals did not differ from that of controls.
Amounts of the proteins AMPA (t=4.768, P<0.001) and PSD-95 (t=4.224, P<0.002) were lower in the 3-AP group than in the Saline rats. The α-catenin content in the cerebellar cortex of the 3-AP animals was greater than in Saline controls (t=−4.339, P<0.001) (Table 2).
Protein content in lobules V–VII of the cerebellar vermis from the control (Saline) and experimental (3-AP) rats.
| GAD-67 (51kDa) | Synaptophysin (34kDa) | AMPA (100kDa) |
| PSD-95 (95kDa) | α-Catenin (135kDa) | β-Actin (43kDa) |
| Group protein | Saline | 3-AP |
|---|---|---|
| GAD-67 | 1.068±0.2 | 0.748±0.09 |
| Synaptophysin | 0.9±0.06 | 0.908±0.08 |
| AMPA | 1.392±0.21 | 0.542±0.09* |
| PSD-95 | 1.034±0.19 | 0.56±0.09* |
| α-Catenin | 0.68±0.1 | 1.168±0.1* |
Mean±SEM, P<0.05.
The present study was designed to quantify the proportional density of spines from the proximal dendritic arborization of the Purkinje cells from lobules V–VII of the cerebellar vermis, and to determine the presence of pre- and postsynaptic molecular markers of synaptic functionality in adult ataxic rats. The lack of coordinated movements was induced by the neurotoxin 3-AP, a nicotinamide antagonist that selectively destroys inferior olivary nucleus neurons in the brain stem20 by suppressing glucose utilization21 to cause an anterograde degeneration of climbing fibers. This effect, with the consequent pathological damage induced in Purkinje cells – as observed by Janahmandi et al.22 and in our work – could impair the periodic clock that coordinates movements and motor timing,23 thus producing ataxia in the rats. In accordance with this, both the BBB test and the equilibrium and coordination testing on the Rotarod applied herein revealed the deficits in balance, gait, and coordination that characterize most cerebellar ataxias.1
Climbing fibers establish synaptic contacts with spines of the proximal dendritic arbor of the cerebellar Purkinje cells. Our study found that dendritic spines were reduced five days after administering 3-AP. Previous reports have shown that the attachment of climbing fibers to proximal spines persists, though gradual degeneration occurs from the first hours to seven days after 3-AP treatment.15 In agreement with our findings, this could induce an initial activity-dependent decrease of spines24 that, with a putative disruption of intracellular signals in the parallel fiber-dendritic spine synapses due to the lack of endoplasmic reticulum in the spines,25 would impair the organization of motor actions and lead to ataxia. Furthermore, the evidence available and our findings agree with reports showing that tetrodotoxin treatment did not affect spine density at day 7 post-treatment26 but increased it from day 7 to 3 months after 3-AP administration.15 This could occur while parallel fibers are gradually innervating some of the remaining proximal spines, inducing a later increase in their density and competing with climbing fiber sprouting until the atypical parallel fibers decrease, disappear, or come to coexist.27
Dendritic spines show several plastic changes under different conditions in addition to increasing (spinogenesis) or decreasing (pruning). These include interconversion between different types of spines and variations in their proportional density. Classical descriptions of spines refer to them as thin, mushroom, stubby, and wide.12,18,28 Thin spines are involved in the rapid transmission of synaptic impulses,29 in immediate plasticity,30 and, consequently, in the acquisition of cognitive information; that is, learning.31,32 Mushroom spines, in contrast, have been associated with the slow transmission of synaptic information33 that underlies the consolidation of long-term memory.34 The decrease of thin and mushroom spines after 3-AP treatment could be associated with a putative deficient integration of excitatory inputs to the Purkinje cells, and with an ineffective association of postsynaptic excitatory potentials.10 These findings could help to explain the difficulties involved in improving movement impairments in rehabilitation paradigms for ataxia. However, whether such a reduction of thin and mushroom spines persists over time (or to what degree) is unknown. Additional pharmacological and behavioral studies are required to elucidate this aspect and to provide evidence of possible strategies to foster restoring a balance in the proportions of the different types of spines.
Both neckless types of spines (stubby and wide) remained unchanged after 3-AP treatment. It has been proposed that they could act as regulators of neuronal excitability,12,18,35–37 which would be in line with reports showing that PCs fire highly regularly in slice preparations even in the absence of synaptic inputs,38 and since they show increased irregularity following 3-AP treatment.39 In addition, it has been reported that even after removal or inactivation of excitatory parallel fiber inputs, PCs continued to display spontaneous slow spike activity.40 In this regard, one proposal suggests that the intrinsic properties of PCs are likely to contribute to the irregularity of firing in ataxic conditions in 3-AP-treated rats.22 Thus, unchanged neckless spines could represent an additional factor that underlies the regulatory mechanisms of electrophysiological stability of Purkinje cells after climbing fiber deafferentation in ataxic rats.
Synaptophysin, a protein deeply involved in the vesiculation of neurotransmitters at the presynaptic level,41 was unchained after 3-AP treatment, suggesting an increase in neurotransmitter vesiculation by viable climbing fibers and/or parallel fibers that occupy those deafferented spines after a lesion to the olivary nucleus. GAD-67 was also unchanged after 3-AP-induced degeneration of the glutamatergic climbing fibers, suggesting an increase in the synthesis of this enzymatic protein. However, impairments in the activity of the vesicular GABA transporter have been associated with ataxia.42 Thus, the kinetics of GAD-67 in ataxia-like experimental models requires further molecular and biochemical studies, given that GAD-67 activity has been shown to be involved in human cerebellar ataxia.43
Initial activation of AMPA receptors located in the apical outer surface of dendritic spines is crucial for the synaptic stimulus to induce an excitatory postsynaptic potential. When glutamate release is insufficient to produce a detectable quantal response – as may occur in the cerebellar cortex after 3-AP-induced lesions of the climbing fibers – NMDA, but not AMPA, receptors could be activated, meaning that the synaptic impulse is not conducted. Under normal conditions, this phenomenon is known as silent synapse.44 This could be potentiated by the fact that AMPA receptors decreased in the cortex of the cerebellar vermis after 3-AP treatment, accompanied by a diminished PSD-95 content. The protein PSD-95 is an integral element of postsynaptic density linked to the synapse maturation of glutamate receptors at excitatory synapses,45 which facilitates their stability and functionality.46 Thus, a reduction of PSD-95 in the cerebellar cortex of 3-AP-treated rats could contribute to impairing the organization of their movement patterns. This would concur with our finding that α-catenin increased after 3-AP-induced degeneration of climbing fibers. The postsynaptic protein α-catenin is linked directly to spine motility and the stabilization of synaptic contacts47 by suppressing the motile activity and turnover of dendritic spines.48 Thus, overexpression of α-catenin after 3-AP treatment could reduce spine plasticity in Purkinje cells and impair the cerebellar cortex’ ability to produce flexible, versatile movement patterns.
In summary, a decrease of the dendritic spines directly related to the acquisition (thin) and consolidation (mushroom) of motor information-related synaptic excitatory inputs was observed in the cerebellar Purkinje neurons of the 3-AP-treated rats, concomitantly with a limited plastic capacity. This could contribute to the aberrant and rigid movement patterns characteristic of ataxic-like disorders.
FundingThis work was supported by Fondo de Investigación en Salud (project number: FIS/IMSS/PROT/G15/1483).
Conflict of interestsThe authors declare that they have no conflict of interest.
The authors acknowledge Professor Cohutec Vargas Genis (Instituto Nacional de Ciencias de la Rehabilitación Integral – INCRI–, A.C. México) for his kind help in logistic issues.