Influenza vaccine is recommended for patients with autoimmune inflammatory diseases (AIID) on biological therapy.
ObjectiveTo evaluate whether serological response to Influenza vaccine obtained in patients on biological therapy is similar to that achieved in patients on synthetic disease-modifying anti-rheumatic drugs (DMARDs) and that obtained in healthy controls.
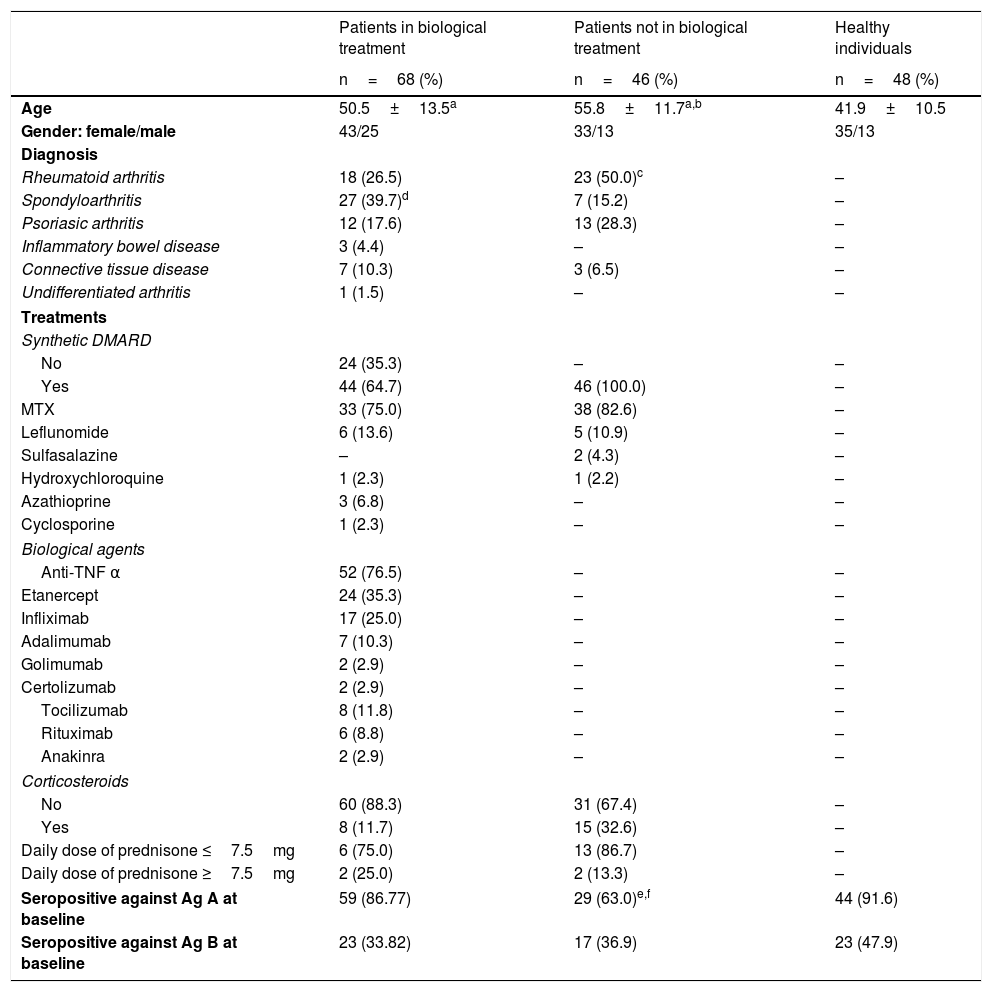
MethodsWe designed a cohort study in which patients with AIID, 68 on biological therapy and 46 on synthetic DMARDs, as well as 48 healthy controls, were included and vaccinated during the 2015–2016 influenza season. ELISA was used to measure Influenza antigen (Ag) A and B antibodies, before and after vaccination.
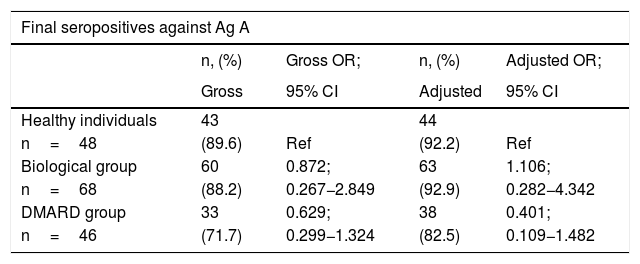
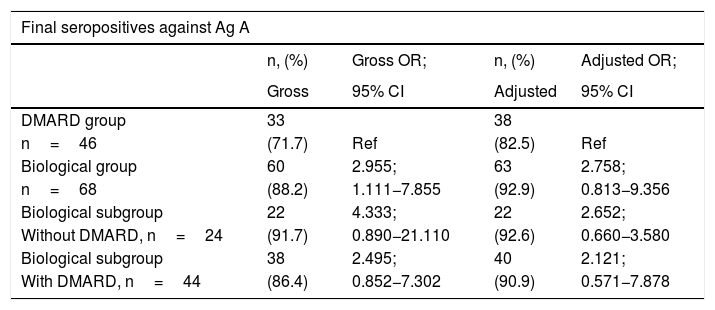
ResultsAfter vaccination, 88.24% of patients on biologics, 71.74% of those on synthetic DMARDs and 89.58% of healthy controls, presented detectable antibodies against antigen A, while 42.65% of subjects on biologics, 41.30% of those on DMARDs and 54.17% of healthy subjects were seropositive against Ag B. We did not find statistical differences.
ConclusionsIn our study, biological therapy is not associated with worse serological response.
Los pacientes con enfermedades autoinmunes inflamatorias (EAII) en tratamiento biológico deben recibir la vacunación antigripal anualmente.
ObjetivoEstudiar si la respuesta serológica a la vacuna antigripal en pacientes con EAII con tratamiento biológico es diferente de la obtenida en enfermos en tratamiento con fármacos modificadores de la enfermedad (FAME) sintéticos y de la conseguida en sujetos sanos.
MétodosSe diseñó un estudio de cohortes en el que se incluyeron pacientes con EAII, 68 tratados con biológicos (grupo biológicos) y 46 con tratamiento con FAME sintéticos (grupo FAME), así como 48 personas sanas. Todos recibieron la vacuna antigripal 2015–2016. Se midieron, mediante ELISA, los títulos de anticuerpos (Ac) frente a los antígenos (Ag) A y B, antes y después de la vacunación.
ResultadosTras la vacunación, el 88,24% de los enfermos del grupo biológicos, el 71,74% del grupo FAME y el 89,58% de las personas sanas fueron seropositivos frente al Ag A, mientras que el 42,65% del grupo biológicos, el 41,30% del grupo FAME y el 54,17% de las personas sanas fueron seropositivos frente al Ag B. No hubo diferencias significativas entre los grupos.
ConclusionesEn nuestro estudio, el tratamiento biológico no se asoció a una peor respuesta serológica.
Artículo
Comprando el artículo el PDF del mismo podrá ser descargado
Precio 19,34 €
Comprar ahora