Lupus nephritis (LN) is a disease marked by autoantibodies against complement components. Autoantibodies against negative complement regulator factor H (anti-FH) are prevalent in aHUS, are associated with deletion of factor H-related protein 1 (FHR1) gene, and have overt functional consequences. They are also observed in C3 glomerulopathies. The frequency and relevance of anti-FH in LN are poorly studied.
AimThe aim of our investigation was to screen for the presence of anti-FH and FHR1 gene deletion in a cohort of LN patients and to evaluate their association with LN activity.
MethodELISA test and Western blot for detection of anti-FH and FHR1 deletion were used, respectively. Patients’ clinical and laboratory parameters regarding anti-FH role were processed by statistical analysis.
ResultsAnti-FH were found at low level in a small number of LN patients – 11.7% (7/60) and were not associated with deletion of FHR1. Anti-FH did not correlate with ANA titers, anti-dsDNA, C3/C4 hypocomplementemia, eGFR, proteinuria, or active urinary sediment in LN patients. A weak correlation was found between anti-FH and anti-C3 levels. Anti-FH were linked with endocapillary proliferation and histological activity index. Four anti-FH positive patients had severe to moderate LN as per the BILAG renal score.
ConclusionsAnti-FH autoantibodies are an accessory finding in LN and are more likely to manifest during the active phase of the disease. Due to their low frequency and plasma levels, they do not seem suitable for routine laboratory investigation in patients with LN.
La nefritis lúpica (NL) es una enfermedad caracterizada por autoanticuerpos contra componentes del complemento. Los autoanticuerpos contra el factor H regulador negativo del complemento (anti-FH) son prevalentes en el síndrome urémico hemolítico atípico, están asociados con la eliminación del gen de la proteína 1 relacionada con el factor H (FHR1) y tienen consecuencias funcionales evidentes. También se observan en las glomerulopatías C3. La frecuencia y la relevancia de los anticuerpos anti-FH en NL son poco conocidas.
ObjetivoEl objetivo de nuestra investigación fue detectar la presencia de anti-FH y deleción del gen FHR1 en una cohorte de pacientes con NL y evaluar su asociación con la actividad de la NL.
MétodosSe usaron pruebas ELISA y Western blot para detectar autoanticuerpos anti-FH y la deleción de FHR1. Se evaluaron los parámetros clínicos y de laboratorio de los pacientes en relación con el anti-FH.
ResultadosSe encontraron anti-FH en niveles bajos en un pequeño número de pacientes con NL: 11,7% (7/60), sin asociación con la eliminación de FHR1. Anti-FH no se correlacionó con ANA, anti-ADNbc, hipocomplementemia C3/C4, eGFR, proteinuria o sedimento urinario activo en pacientes con NL. Se encontró una correlación débil entre los niveles de anti-FH y anti-C3. Los anti-FH estaban asociados con la proliferación endocapilar y el índice de actividad histológica. Cuatro pacientes con anti-FH tenían NL severa/moderada según BILAG.
ConclusionesLos anti-FH son un hallazgo secundario en la NL y es más probable que se manifiesten durante la fase activa de la enfermedad. Por su baja frecuencia y sus niveles plasmáticos, no son adecuados para investigación de laboratorio de rutina en pacientes con NL.
Lupus nephritis (LN) is a frequent manifestation of systemic lupus erythematosus (SLE).1 Classical complement pathway plays a key role in the LN pathogenesis.2 SLE patients with LN usually are positive for anti-complement C1q autoantibodies,3 the specificity, sensitivity and significance of which are studied in details in LN. The presence of autoantibodies against proteins and regulators involved in the alternative complement activation pathway is starting to be explored in LN patients. There are already evidences for possible pathogenic role of anti-C3 and anti-properdin autoantibodies,4–6 but not many studies for the rest.
Factor H is a negative regulator of the alternative complement pathway. It serves as an inhibitor of C3 convertase and as a cofactor of C3b inactivation by factor I-mediated cleavage.7 Autoantibodies against factor H (anti-FH) are a hallmark of the atypical form of haemolytic uremic syndrome (aHUS), and to a lesser extent of C3 glomerulopathies (C3G).8–10 However, only few studies describe anti-FH autoantibodies in SLE and LN patients.11,12 The strong disease relevance of these antibodies in other renal diseases rises the interest in studying anti-FH autoantibodies in SLE and evaluating their potential functional consequences as well as the alternative complement pathway reflection of the disease activity and activation-induced self-injury.
In the current study, our aim was to screen for the presence of anti-FH autoantibodies and FHR1 gene deletion in a cohort of LN patients. We also evaluated associations between anti-FH antibodies and some clinico-morphological and histological markers of LN activity.
Patients and methodsPatients and healthy controlsThis single-center retrospective cross-sectional study examines a SLE patients with proven lupus nephritis, followed at the Nephrology Clinic of the University Hospital “Tsaritza Yoanna – ISUL”, affiliated with the Medical University of Sofia, Bulgaria. The study included 60 patients (80% female) at a mean age of 44.9±14.8 years (ranging from 21 to 87), diagnosed with SLE based on the revised American College of Rheumatology (ACR) 1997 criteria,13 with a minimum of 4 out of 11 criteria met. At the time of enrollment in the study, 16 patients (26.7%) had reference-level proteinuria (below 0.15g/24h), 22 (36.7%) had low-grade proteinuria (between 0.15 and 1.0g/24h), 9 (15%) had moderate-grade proteinuria (between 1.0 and 3.5g/24h), and 13 (21.6%) had nephrotic-range proteinuria. These patients exhibited lupus nephritis, characterized by persistent proteinuria of 0.5g/24h or higher and/or the presence of erythrocyturia, leukocyturia, or cell cylinders upon microscopic examination of urine sediment at the time of the diagnosis. This was observed after ruling out other diseases that could potentially cause these urinary changes. The patients involved in the study had lupus nephritis confirmed either histologically (58 patients) or clinically (2 patients) during their treatment at the Nephrology Clinic. The following exclusion criteria are applied: unwillingness to participate in the study; age – under 18 years; presence of concomitant infectious inflammatory disease; presence of other concomitant autoimmune or neoplastic disease that could affect laboratory or immunological results. Complex disease activity of LN was assessed by British Islet Lupus Assessment Group (BILAG) renal.14 This cohort was stratified into four BILAG categories as follows: 22 patients (36.7%) in category A, 21 patients (35.0%) in category B, 7 patients (11.7%) in category C and 10 patients (16.7%) in category D LN. Extra-renal manifestations were reported for 85% (51/60) of the patients. According to the International Society of Nephrology and Renal Pathology Society (ISN/RPS) 2003 classification15 patients with biopsy-proven LN were distributed among the classes as follows: 4 patients (6.9%) had LN Class I, 20 patients (34.5%) had LN Class II, 3 patients (5.2%) had LN Class III, 21 patients (36.2%) had LN Class IV, 10 patients (17.2%) had LN Class V. No patient presented with LN Class VI. Histological activity and chronicity indices (histological activity index – HAI and histological chronicity index – HCI) were evaluated according to the National Institute of Health system.16
Plasma samples from 26 healthy volunteers, matched for sex and age, over 18 years of age, and without altered renal, hepatic or hematopoietic functions were collected as a control group. All plasma samples were stored at −80°C.
The study had the approval of the Ethics Review Board of Medical University of Varna, Bulgaria (protocol no. 62/04.05.2017) and conducted according to the Declaration of Helsinki. All participants in the study signed in an informed consent.
Clinical and laboratory parameters of LN patientsClinical, laboratory, immunological and histological analyses were performed in the University Hospital “Tsaritza Yoanna – ISUL” laboratories. Data for all patients were collected throughout their treatment and monitoring period at the Nephrology Clinic. Indirect immunofluorescence was used for antinuclear antibodies (ANA) titres measurement and anti-dsDNA levels were detected by ELISA (U/mL). Plasma concentration of C3 and C4 complement components was measured by immunodiffusion. The reference range for C3 between 0.75 and 1.65g/L, and that for C4 varied between 0.20 and 0.65g/L C3 hypocomplementemia was detected in 12 patients (20%, 12/60), C4 hypocomplementemia in 23 patients (38.3%, 23/60), and concomitant C3 and C4 hypocomplementemia was detected in 11 patients (18.3%). Renal biopsy specimens were examined by light microscopy and immunofluorescence, and the diagnosis of LN was made on the basis of minimum 10 glomeruli in biopsy specimen.
ELISA for detecting of autoantibodies against complement components – factor H, properdin (factor P), C1q and C3ELISA plates (Greiner bio-one®) were coated with 20μg/mL of human antigens (factor H, properdin, C1q and C3 (Complement Technology, Ins)) in sodium carbonate buffer (35mM NaHCO3, 15mM Na2CO3, pH 9.6) for overnight at 4°C. 1% BSA in PBS were used for blocking of the plates for 1h at 37°C. The plates were washed three times with 300μL/well PBS containing 0.05% Tween-20. Plasma samples were diluted 1/100 in PBS–0.05% Tween-20, except the plate coated with C1q, where PBS/750mM NaCl–0.1% Tween-20 was used to prevent the detection of C1q–IgG interaction. After washing, HRP-conjugated anti-human IgG (Southern Biotech) was applied in 1/1000 dilution in PBS-0.05% Tween-20 (100μL/well). After washing three times, the reaction was developed with 0.5mg/mL ophenylenediamine (OPD) (Thermo Scientific). The absorbance at 490nm was measured using an ELISA plate Reader – Synergy 2. A plasma sample was classified as positive for a specific autoantibody if its optical density surpassed the mean optical density of 26 healthy volunteers’ samples by more than three standard deviations.
Detection of CFHR1 deletion by Western blotPlasma samples of LN patents and healthy controls were diluted 1/100 in distilled water and mixed with non-reducing sample buffer in 4:1 ratio. After boiling for 10min, the samples were deposited on 10% NuPAGE Bis–Tris precast gel (Invitrogen) and migrated for 40min. After transfer, the nitrocellulose membrane was blotted with goat anti-human factor H antiserum (Quidel), diluted 1/1000, followed by a secondary anti-goat-HRP antibody. The signal was revealed by chemiluminescence, using the ECL detection kit and iBright imaging system (Invitrogen).
Statistical analysisStatistical analysis was carried out using software GraphPad Prism 6.01. Quantitative data were presented as either mean±3 standard deviations (SD) or median (range). The Mann–Whitney U test was employed for two-group comparisons of continuous variables, while the Kruskal–Wallis test with Dunn's multiple comparison test was used for comparisons involving more than two groups. Spearman nonparametric correlation was performed to measure the strength and direction of association between two ranked variables. With the exception of the age frequency distribution, the remaining quantitative variables have non-Gaussian distributions, hence nonparametric statistical tests have been applied. A Kolmogorov–Smirnov test has been conducted to assess the frequency distribution. Statistical significance was considered at p<0.05.
ResultsPresence of autoantibodies against factor H in LN patientsAutoantibodies against factor H were found in small proportion of patients – 11.7% (7/60). There was a statistically significant difference in the levels of anti-FH antibodies between LN patients and healthy volunteers (p=0.019, Fig. 1). The median level of anti-FH autoantibodies in LN patients was 0.089 and the median level of anti-FH in the control group was 0.119.
Since anti-FH antibodies in aHUS patients usually correlate strongly with homozygous deletion of CFHR1 and CFHR3 genes10 we searched for the presence of gene deletion of factor H-related proteins (CFHL1, CFHR1 and CFHR2) in our cohort. The anti-FH antiserum used recognizes factor H, CFHR1, CFHR2 and CFHL1 and allows detecting possible homozygous protein deficiency in the patients. The sensitivity of the method is not sufficient for determining heterozygous deficiency of factor H-related proteins. All 25 LN patients tested except one (P17), showed signal, corresponding to the two glycoforms of CFHR1 (Fig. 2). No CFHR1 deletion was found in the 20 healthy controls investigated. Therefore, the frequency of the deletion of CFHR1 in our study was very low, below 2%.
Western blot analysis for detection of CFHR1 deletion. Plasma samples of 25 LN patents (P) and 20 healthy controls were investigated. The samples were deposited on 10% NuPAGE Bis–Tris precast gel (Invitrogen), after transfer, the nitrocellulose membrane was blotted with goat anti-human factor H antiserum (Quidel), diluted 1/1000, followed by a secondary anti-goat-HRP antibody.
LN patients were divided into two groups – anti-FH positive and anti-FH negative. There were no significant differences between the clinical and laboratory parameters in the two groups (Table 1).
Clinical and laboratory parameters in positive for anti-FH patients in comparison with negative for anti-FH antibodies patients.
| Clinical and laboratory parameters | Anti-FHpositive | Anti-FHnegative | p-Value |
|---|---|---|---|
| Sex, female/male | 5/2 | 43/10 | 0.550 |
| Age, median (range) | 47 (from 35 to 75) | 41 (from 21 to 87) | 0.273 |
| Duration of disease in years, median (range) | 5.5 (0.25–24.00) | 9.00 (0.02–41.00) | 0.915 |
| Extra-renal manifestations, % | 100% (7/7) | 83.01% (44/53) | 0.439 |
| Creatinine, μmol/L, median (range) | 62 (53.00–96.00) | 68 (47.00–918.0) | 0.648 |
| eGFR, mL/min/1.73 sqm, median (range) | 91 (61.00–136.00) | 92 (6.00–135.0) | 0.656 |
| Proteinuria, g/L, median (range) | 0.45 (0.041–5.73) | 0.53 (0.02–15.72) | 0.283 |
| Active urinary sediment, % | 42.86% (3/7) | 58.49% (31/53) | 0.438 |
| Haemoglobin, g/L, median (range) | 137 (113.0–167.0) | 127.0 (55.00–181.0) | 0.244 |
| CRP, median (range) | 0.69 (0.27–1.11) | 0.37 (0.01–5.28) | 0.662 |
| (+) ANA, % | 100% (6/6) | 78% (40/51) | 0.386 |
| (+) anti-dsDNA, % | 50% (3/6) | 41% (19/46) | 0.686 |
| C3, median (range) | 1.29 (0.63–1.51) | 1.14 (0.44–1.88) | 0.931 |
| C4, median (range) | 0.15 (0.10–0.40) | 0.24 (0.03–0.53) | 0.355 |
Even though the detected levels of anti-FH autoantibodies were very low, we examined and compared their median levels in patients either positive or negative for certain clinicopathological markers of LN activity.
The median anti-FH level in patients with pathological proteinuria ≥0.15g//24h was 0.100, and in patients with reference levels of proteinuria (<0.15g//24h) – 0.095. There was no difference (p=0.869, data not shown) between the two groups which was confirmed also by correlation analysis (r=−0.039, p=0.774, data not shown).
The median anti-FH level in patients with active urinary sediment above 8 erythrocytes per microliter and/or above 8 leukocytes per microliter (from non-centrifuged urine, by the Stansfield–Webb method) with or without presence of non-hyaline casts, with exclusion of other causes of hematuria, leukocyturia, and cylindruria, other than lupus nephritis activity) is 0.110, while in patients without pathologically active urinary sediment it is 0.063. The relatively higher median anti-FH levels in patients with active findings showed statistically significant difference with these of patients with non-active urine sediment (Mann–Whitney U, p=0.021, data not shown).
We did not find a link between anti-FH levels and estimated glomerular filtration rate (eGFR) (p=0.409, data not shown).
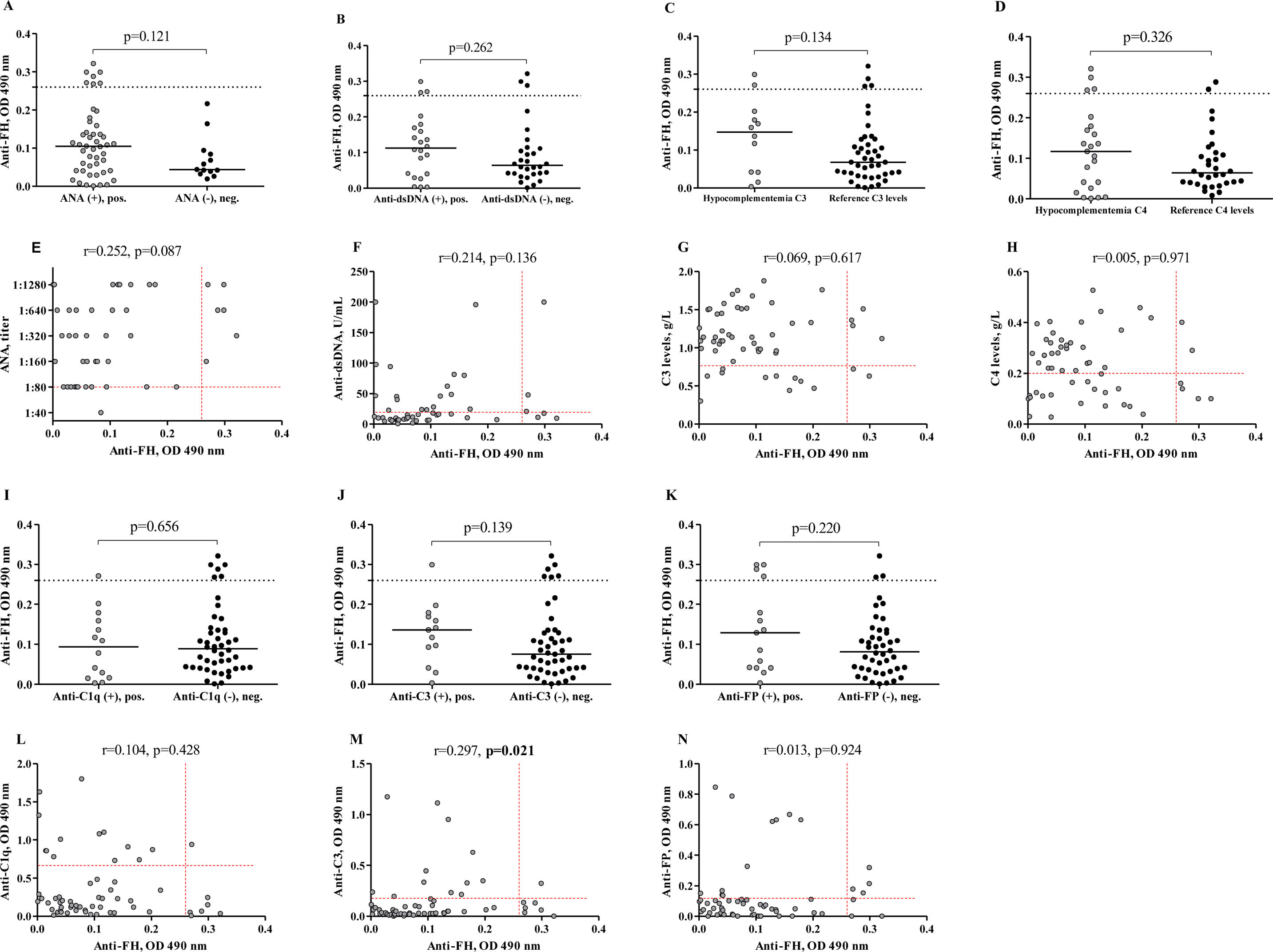
No significant difference in anti-FH levels was found between patients with pathologically elevated ANA titers and those with reference ANA titers (p=0.121, Fig. 3A). The correlation analysis did not reveal a significant correlation between the levels of anti-FH and ANA titers (r=0.252, p=0.087, Fig. 3E).
Levels of anti-FH autoantibodies in patients divided on the immunological estimation for LN activity. Levels of anti-FH in patients with LN depending on the presence or absence of pathological ANA titer (A), anti-dsDNA levels (B), C3 hypocomplementemia (C), C4 hypocomplementemia (D), anti-C1q levels (I), anti-C3 levels (J) and anti-factor P levels (K). The dashed lines in the correlation analysis graphs show cut-off for anti-FH. The medians of two groups in every graphic were compared using the Mann–Whitney test. Correlation between levels of anti-FH and levels of ANA (E), anti-dsDNA (F), C3 levels (G), C4 levels (H), anti-C1q levels (L), anti-C3 levels (M), and anti-factor P levels (N). Spearman correlation analysis was used. The dashed lines in the correlation analysis graphs show the lower reference ranges for C3 and C4 (G and H) and the cut-offs for ANA, anti-dsDNA, anti-C1q, anti-C3, anti-FP (E, F, L, M, and N) respectively.
We did not find significant difference in anti-FH levels in between cases with and without pathologically elevated anti-dsDNA (p=0.262, Fig. 3B). Additionally, the correlation analysis revealed no significant relationship between anti-FH and anti-dsDNA (r=0.214, p=0.136, Fig. 3F).
The median anti-FH levels in LN patients with C3- and C4-hypocomplementemia were – 0.148 and 0.117 respectively. The differences between the groups – those with low C3/C4 levels and those with normal complement components – were not statistically significant (p=0.134 for C3, Fig. 3C and p=0.327 for C4, Fig. 3D). Furthermore, these differences were not supported by correlation analysis (for C3 r=−0.069, p=0.617, Fig. 3G, and for C4 r=−0.005, p=0.971, Fig. 3H). Autoantibodies against C1q are applied as reliable biomarkers for LN activity. Anti-C1q titters have prognostic value for LN flares.17 Recently, anti-C3 antibodies have been rediscovered as important players in LN pathology.5 Anti-C3 are characterised with high specificity in LN. For this reason, we examined the associations between anti-FH and anti-C1q, as well as between anti-FH and anti-C3. Only one patient had simultaneously elevated levels of both anti-FH and anti-C1q (1/60, 1.7%). Anti-FH positive patients, who were negative for anti-C1q were 6 (6/60, 10.0%). The median anti-FH level in patients with elevated anti-C1q levels (0.094) did not significantly differ from that in patients with reference levels of anti-C1q (0.089) (p=0.656, Fig. 3I). The correlation analysis confirmed the lack of dependence between the levels of anti-FH and anti-C1q (r=−0.104, p=0.428, Fig. 3L). We did not establish a difference between the medians of anti-FH levels in positive (0.136) and negative (0.075) for anti-C3 groups of patients (p=0.139, Fig. 3J) but the correlation between levels of these antibodies was significant and weak (r=0.297, p=0.021, Fig. 3M).
The relationship between anti-FH and relatively novel in LN autoantibodies against another complement regulator, factor P was also studied. However, no significant difference was established between the median levels of anti-FH in patients with different anti-factor P status (p=0.220, Fig. 3K). There was no correlation between levels of anti-FH and anti-FP in LN patients (r=0.013, p=0.924, Fig. 3N).
We observed that patients with a more severe category A LN, as determined by the BILAG renal score, tended to have higher levels of anti-FH (median – 0.110) compared to those in other categories (B–D) indicating milder LN severity (median – 0.060), although this difference was not statistically significant (p=0.149, Fig. 4A). When comparing the levels of anti-FH among patients in different categories according to the BILAG renal score – A (median – 0.110), B (median – 0.060), C (median – 0.041), and D (median – 0.093), we did not find statistically significant variations in the medians across the individual groups (p=0.366). Furthermore, no significant differences in the levels of anti-FH were observed between the individual categories (Dunn's multiple comparison test, p>0.05, n.s.) (Fig. 4B).
Comparison of distribution of anti-FH between patients with active LN (category A according to the BILAG renal score) and those with milder active LN (categories B–D) (A). Distribution of anti-FH in all four BILAG renal score categories (B). Kruskal–Wallis test, Dunn's multiple comparison test, n.s. – p>0.05 were used (B). Correlation between anti-FH and histology activity index (C). Correlation between anti-FH and histology chronicity index (D).
Due to the importance of histological changes as indicators for the severity and the prognosis of the disease, the relationship between anti-FH and histological markers of activity (endocapillary hypercellularity, glomerular leukocyte infiltration, subendothelial immune deposits – “wire loops”, fibrinoid necrosis, cellular crescents, and interstitial inflammation) and chronicity (glomerular sclerosis, fibrous crescents, tubular atrophy, and interstitial fibrosis) was also studied (Table 2). A statistically significant higher level of anti-FH was observed in the presence of endocapillary proliferation compared to all other histological markers of activity (p=0.004, Table 2). There were no significant differences in anti-FH levels among the histological signs of chronicity, including glomerular sclerosis, fibrous crescent, tubular atrophy, and interstitial fibrosis. We found a strong significant correlation between anti-FH and histology activity index (HAI) (r=0.545, p=0.006, Fig. 4C), but not with histology chronicity index (HCI) (Fig. 4D).
Comparative analysis between levels of anti-FH in groups of patients with and without histological signs of LN activity and chronicity.*
| Histological features | Anti-FH, median | ||
|---|---|---|---|
| Presence | Absence | p-Value | |
| Endocapillary proliferation | 0.114 | 0.039 | 0.004 |
| Glomerular leukocyte infiltration | 0.174 | 0.076 | 0.068 |
| “Wire loop” deposits | 0.111 | 0.068 | 0.522 |
| Fibrinoid necrosis/karyorrhexis | 0.114 | 0.053 | 0.152 |
| Cellular crescents | 0.117 | 0.084 | 0.670 |
| Interstitial inflammation | 0.117 | 0.053 | 0.170 |
| Glomerular sclerosis | 0.042 | 0.111 | 0.064 |
| Fibrous crescents | 0.042 | 0.094 | 0.320 |
| Tubular atrophy | 0.063 | 0.101 | 0.561 |
| Interstitial fibrosis | 0.114 | 0.053 | 0.228 |
Factor H is a negative regulator of complement alternative pathway, highly abundant in plasma, inhibiting C3 activation in the fluid phase as well as on cell surfaces.18 Recently, it was shown that factor H protects against glomerular endothelial injury by interacting with neutrophils in patients with anti-neutrophil cytoplasmic autoantibody (ANCA)-associated vasculitis (AAV).19 Serum factor H levels in active LN were reported to be lower than in SLE without nephritis or in the control group.20 A possible explanation of this observation is the presence of autoantibodies against factor H. These autoantibodies are well studied in the autoimmune form of HUS.8,10,21 In aHUS anti-FH autoantibodies form complexes with the antigen, resulting in a functional factor H deficiency.10,22 In C3G anti-FH autoantibodies also inhibit the regulatory factor H activity, but unlike in aHUS, where these antibodies are more specific for C-terminal part of factor H, here they bind to the molecule at different epitopes.10 Therefore, anti-FH antibodies may predispose to renal diseases.
Our study found the presence of anti-FH antibodies in only 11.7% (7/60) of the studied LN patients. Since the occurrence of anti-FH is strongly associated with deletion of the CFHR1/CFHR3 genes in aHUS,23 we checked if this is cause for our cohort. We can conclude that among Bulgarian LN patients the complete deletion of CHFR1 occurs in ∼2% or less. Indeed, the frequency of the complete deficiency of CFHR1 and CFHR3 had been found in approximately 4% of the European population and there is a high population difference that ranges from 0 to ∼30%.23 In our LN cohort, as in C3G, anti-FH are not related to CFHR1/CFHR3 deletion.10
The frequency of 11.7% of positive patients found here is in agreement with the study of Li et al. where 8.3% (10/120) of the LN patients were found positive for anti-FH autoantibodies.12 Foltyn et al. found 6.8% (4/60) anti-FH autoantibodies. Notably, these were observed in SLE patients without renal involvement and during periods of heightened disease activity.11 Pradhan et al. reported the largest frequency of anti-FH positive patients – 19.3% (11/57), clinically diagnosed with SLE without LN.24 However, in a larger cohort of 241 LN patients anti-FH autoantibodies were not detected.20 Taken together, these findings suggest that anti-FH autoantibodies are not common in LN. On the other hand, the prevalence in LN patients is similar to the prevalence in aHUS and C3G, where these antibodies are pathologically relevant and hamper the regulation of the alternative complement pathway.9 In the recent years, a role of the alternative pathway in LN pathogenesis was considered.25 There are several studies that describe the functional role of autoantibodies against alternative pathway components in LN.4–6 Therefore, it was tempting to speculate that anti-FH autoantibodies may contribute to the dysregulation of the alternative pathway in the patients with LN.
Surprisingly, the study of Li et al. suggested a protective role for anti-FH in a Chinese cohort with LN.12 They claims that LN patients with anti-FH autoantibodies had lower serum creatinine levels and a lower prevalence of acute kidney injury than patients who were negative for these autoantibodies.12 The same group also found that anti-FH autoantibodies could attenuate pristane-induced lupus nephritis in mice.26 In our Caucasian cohort, we found that anti-FH levels were not correlated with ANA titers, anti-dsDNA levels, presence of C3 and C4 hypocomplementemia, eGFR, proteinuria, or the presence of active urinary sediment. We only observed a weak positive correlation between the levels of anti-FH and anti-C3. The presence of elevated anti-FH antibodies was significantly associated only with the presence of endocapillary proliferation and correlated with the histological activity index. Moreover, four patients seropositive for anti-FH (4/7) had severe or moderate LN according to BILAG renal score (category A and category B). Taken together, our findings suggest that anti-FH autoantibodies are rather related to active phase of the disease, than to have a protective role as suggested by Li et al.12 In confirmation to our hypothesis comes a case study of a young LN (Class IV) patient with severe lupus flare, who developed thrombotic microangiopathy (TMA).27 Figueiredo et al. detected high levels of anti-FH autoantibodies in the patient.27 Since there is an evidence for alternative pathway involvement in TMA development,28 the authors suggested the large amounts of anti-FH, likely triggered by a flare, may be one of the factors contributing to this serious complication.
To clarify the differences in results between our study and that of Li et al., we conducted a thorough comparison of our protocol with theirs, which was used for detecting anti-FH autoantibodies in plasma. We identified only one significant methodological difference in the immobilization of FH on an ELISA plate. Specifically, Li et al. biotinylated FH prior to analysis, allowing it to be immobilized on a streptavidin-coated plate. Given the abundance of lysines on protein surfaces, biotin can be introduced at various positions on the protein. Consequently, the target protein, in this case, FH, can be immobilized in multiple orientations, thereby probably exposing different potential epitopes. It is important to note that protein biotinylation can alter and potentially obscure epitopes, which can affect the three-dimensional conformation or specific properties of the target molecule.29
In Li et al.’s experiments, the biotinylation of FH may have effects similar to those of FH reduction, leading to its recognition by various anti-reduced FH antibodies. These antibodies have been identified in patients with lung and renal cancer.30–32 It is assumed that they recognize an epitope on FH, which is revealed after its reduction only on the cancer cell surface.31 Anti-reduced FH antibodies have been found in both cancer patients and some healthy donors. They have been associated with favorable outcomes and are believed to play a role in protective autoimmunity against tumor FH neoantigens.30
Factor H appears to be a paradoxical molecule regarding two extremely opposite categories of diseases – autoimmune diseases and cancer. On one hand, it may act as an inhibitor of the alternative pathway involved in lupus pathology. On the other hand, its immunosuppressive properties favor the tumor microenvironment. This contrast raises questions about the role of anti-factor H antibodies in both autoimmunity and cancer – are they pathological, protective, or simply neutral findings? In cancer, they may play a protective role. Binding to Factor H leads to complement-mediated lysis of tumor cells.31 While anti-FH antibody therapy in cancer shows promise, a major concern is the potential for triggering an unwanted autoimmune response. In terms of autoimmune diseases, anti-FH are definitively associated with the development of aHUS. However, the role of these antibodies in LN remains unclear. The main problem with the anti-FH in LN, though, is that their levels are significantly lower compared to the high titres observed in aHUS and C3G, making them challenging targets for functional analyses.
Our study has of course several limitations that ought to be addressed in future research attempts. First of all, our cohort is relatively small and the retrospective design does not allow a follow-up of the fluctuations in anti-FH levels during different disease flare-ups. Moreover, we, as well as other similar studies lack a detailed analysis of the extra-renal manifestations of SLE, which can at least partially account for the controversies, existing among the studies. For example, Foltyn et al. report autoantibodies against FH in a significant proportion of patients with primary and secondary antiphospholipid syndrome, as well as in recurrent venous thrombosis sufferers, suggesting their role in disruption of hemostasis, unrelated to autoimmunity per se.33 In this line of thought, future prospective studies can be designed, following bigger cohorts for a longer time, accounting for extra-renal manifestations of SLE, secondary antiphospholipid syndrome and different treatment regimens, which are known to affect autoantibody levels.
ConclusionEven though this study is based on a relatively small, retrospective and cross-sectional cohort, which makes it difficult to follow-up the changes in levels of anti-FH antibodies during the development of LN, our data suggest that the anti-FH autoantibodies are probably an epiphenomenon and a part of strongly deregulated immune repertoires during the disease activity. We confirmed the presence of anti-FH autoantibodies in LN patients. However, due to their low frequency and levels it does not appear to be significant to investigate them in the routine clinical practice for LN patients.
Ethical considerationsThe study had the approval of the Ethics Review Board of Medical University of Varna (protocol no. 62/04.05.2017) and conducted according to the Declaration of Helsinki. All participants in the study signed in an informed consent.
FundingThis work was supported by the Bulgarian National Science Fund, Competition for financial support for bilateral projects – 2016 – Bulgaria-France (project No DNTS/France 01/11, 09.05.2017) and by the European Union-NextGenerationEU program, through the National Recovery and Resilience Plan of the Republic of Bulgaria (project No BG-RRP-2.004-0009-C02).
Authors’ contributionsDesigned the study: MR. Performed research: GM, MR, MK. Takes care for the patients and collected the clinical data: VV, MK and MP. Discussed and analyzed the data: GM, VV, and MR. Wrote the manuscript: VV, GM and MR. Acquired funding: MR. All authors approved the submission.
Conflict of interestThe authors have no conflicts of interest to declare.














 0.05 were used (B). Correlation between anti-FH and histology activity index (C). Correlation between anti-FH and histology chronicity index (D).' title='Comparison of distribution of anti-FH between patients with active LN (category A according to the BILAG renal score) and those with milder active LN (categories B–D) (A). Distribution of anti-FH in all four BILAG renal score categories (B). Kruskal–Wallis test, Dunn's multiple comparison test, n.s. – p>0.05 were used (B). Correlation between anti-FH and histology activity index (C). Correlation between anti-FH and histology chronicity index (D).'/>
0.05 were used (B). Correlation between anti-FH and histology activity index (C). Correlation between anti-FH and histology chronicity index (D).' title='Comparison of distribution of anti-FH between patients with active LN (category A according to the BILAG renal score) and those with milder active LN (categories B–D) (A). Distribution of anti-FH in all four BILAG renal score categories (B). Kruskal–Wallis test, Dunn's multiple comparison test, n.s. – p>0.05 were used (B). Correlation between anti-FH and histology activity index (C). Correlation between anti-FH and histology chronicity index (D).'/>