To calculate the incidence of invasive pneumococcal disease (IPD) in the paediatric population of Gran Canaria (Spain), its clinical and epidemiological characteristics, serotype distribution, antibiotic resistance, and variations in these variables before and after the introduction of the PCV13 vaccine.
MethodsProspective hospital-based study including all patients (190) aged 0–14 years admitted with confirmed IPD between January 2001–May 2010 (152 cases) and June 2010–December 2016 (38 cases). Patients were divided into 3 age groups (<2 years; 2–5 years; and >5 years). Clinical symptoms were mutually-exclusively classified as meningitis, bacteraemic pneumonia, pleural effusion (PE), empyema or bacteraemia without a focus.
ResultsMost cases occurred in boys (59.47%), during autumn-winter (65.79%), in children aged <2 years (55.79%) and with mean age increasing from the pre-PCV13 to the post-PCV13 period (2.5 vs 3.1 years). Incidence between periods reduced by 66.4% (p<0.001): from 13.1/100,000 to 4.4/100,000. PEs (3.9% vs 18.4%, p<0.005) and empyemas (1.5% vs 16.7%, p=NS) increased in the post-PCV13 period whereas all other symptoms decreased, although this was not statistically significant. Vaccine serotypes (77% vs 40.6%, p=0.000), particularly serotypes 19A (23.9% vs 12.5%) and 14 (14.2% vs 9.4%), as well as erythromycin resistance (57.2% vs 7.9%, p=0.000) decreased in the post-PCV13 period.
ConclusionIPD incidence, vaccine serotypes and erythromycin resistance decreased in the post-PCV13 period whereas PEs increased.
Calcular la incidencia de enfermedad neumocócica invasiva (ENI) pediátrica en Gran Canaria (España), sus características clínicas y epidemiológicas, distribución de serotipos, resistencias antibióticas, y las variaciones en estas variables antes y después de la introducción de la vacuna PCV13.
MétodosEstudio hospitalario prospectivo que incluye todos los pacientes con edades entre 0-14 años (190) ingresados con un diagnóstico confirmado de ENI entre enero 2001 – mayo 2010 (152 casos) y junio 2010 – diciembre 2016 (38 casos). Los pacientes se dividieron en 3 grupos de edad (<2 años; 2-5 años; y >5 años). Los síntomas clínicos se clasificaron de manera mutuamente exclusiva en meningitis; neumonía bacterémica, derrame pleural (DP), empiema, o bacteremia sin foco.
ResultadosLa mayoría de los casos se concentró en en varones (59.47%), durante otoño-invierno (65.79%), en <2 años (55.79%) y con un aumento de la edad media entre periodos (2.5vs3.1 años). La incidencia entre periodos se redujo un 66.4% (P<0.001): de 13.1/100,000 a 4.4/100,000. Los DPs (3.9%vs18.4%, P<0.005) y empiemas (1.5%vs16.7%, P=NS) aumentaron durante el periodo post-PCV13 mientras que el resto de síntomas se redujeron pero de manera no estadísticamente significativa. Los serotipos vacunales (77%vs40.6%, P=0.000), especialmente los serotipos 19A (23.9%vs12.5%) y 14 (14.2%vs9.4%), así como la resistencia a la eritromicina (57.2%vs7.9%, P=0.000) se redujeron en el periodo post-PCV13.
ConclusiónEn el periodo post-PCV13 se redujo la incidencia de ENI, los serotipos vacunales, y la resistencia a la eritromicina mientras que aumentaron los DPs.
Streptococcus pneumoniae is responsible for a large variety of infectious diseases, the most serious of which constitute the group called invasive pneumococcal disease (IPD).
In our country, marketing of paediatric pneumococcal vaccines began in June 2001 with a vaccine (PCV7, Wyeth/Pfizer, Prevenar®), that immunized against 7 serotypes (ST), followed by a new vaccine adding 3 more STs in March 2009 (PCV10, GSK, Synflorix®) and a last one in June 2010 adding other 3 STs (PCV13 Wyeth/Pfizer, Prevenar13®). However, the immunization schedule in our region did not publicly fund pneumococcal vaccination until March 2015 (in a 2+1 schedule).
The aim of our study was to calculate the incidence of paediatric IPD in our population, its clinical and epidemiological characteristics, ST distribution, antibiotic resistance, and the evolution in these variables among the years preceeding PCV13 introduction (pre-PCV13 period) and the post-PCV13 period.
MethodsDesignProspective hospital-based study including the 190 patients aged 0–14 years admitted to the only paediatric tertiary referral hospital in the island of Gran Canaria (Hospital Universitario Materno-Infantil de Canarias, Gran Canaria, Spain) between January 2001–December 2016 with confirmed IPD by culture or PCR.
Patients were divided into 3 age groups (<2 years; 2–5 years; and >5 years) and 2 different periods for comparison purposes, January 2001–May 2010 (pre-PCV13 period) and June 2010–March 2016 (post-PCV13 period).
Vaccination status, basic demographic and clinical data were collected. Clinical syndromes were mutually exclusive classified as meningitis, bacteremic pneumonia (BP), pleural effusion (PE), empyema or bacteremia without a focus. The study was approved by the local Ethical Committee and informed consent was not necessary.
Identification and serotypingSterile samples were collected from all patients: blood (71.1%), cerebrospinal fluid (15.8%) or pleural fluid (13.1%). Identification of S. pneumoniae was performed at the local Service of Microbiology by observing umbilicated α-hemolytic colonies or mucosa on blood agar and sensitive to optochin. In doubtful cases, species was identified by determining the solubility in bile and the agglutination with latex particles coated with specific antiserum (Pneumo-Slidex Test Kit, Biomerieux, Marcy L’Etoile, France). When cerebrospinal or pleural fluid samples did not yield a positive culture, samples were sent to the reference laboratory (National Centre for Microbiology, Majadahonda, Madrid) for identification by a quantitative real time PCR detecting pneumolysin (ply) gene.1
Serotyping was performed at the reference laboratory using the Quelling method or PCR methodology2 when culture was negative. ST was available in 145 cases.
Antibiotic susceptibility testsAntibiotic susceptibility testing was performed at the local Service of Microbiology by E-test (ABbiodisk, Solna, Sweden) on Müller-Hinton agar with blood for the following antibiotics: penicillin, cefotaxime and erythromycin. The results were reported according to CLSI criteria in force at every time.
Statistical analysisTo determine the incidences of each period, incidence was considered as a random variable distributed according to a Poisson law. This model assumed that expected incidences were constants at each period. Incidences for each period/year were deduced from the model, which was estimated by the maximum likelihood method, and the results were summarized as number of cases by 100,000 persons. Population data were obtained from the Canary Islands Statistics Institute (ISTAC).
Data were analyzed using SPSS® v23 (SPSS, Chicago, Illinois, USA). Differences were assessed using X2 or Fisher's exact test when appropriate. Statistical significance was established at α=0.05 and confidence intervals were calculated.
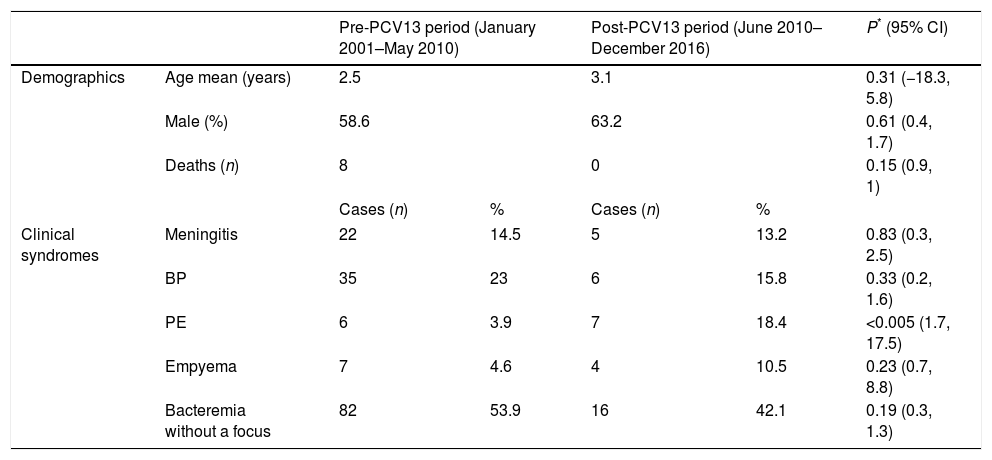
ResultsDemographic characteristics of the study population and distribution of clinical syndromes among study periods are showed in Table 1. During pre-PCV13 period, 152 cases were registered with an average age of 2.5 years, mostly in males (58.6%) and in winter (36.2%). During post-PCV13 period, 38 cases were registered with an average age of 3.1 years and, again, mostly in males (63.2%) and in winter (34.2%).
Demographic characteristics and clinical syndromes of the study population.
| Pre-PCV13 period (January 2001–May 2010) | Post-PCV13 period (June 2010–December 2016) | P* (95% CI) | ||||
|---|---|---|---|---|---|---|
| Demographics | Age mean (years) | 2.5 | 3.1 | 0.31 (−18.3, 5.8) | ||
| Male (%) | 58.6 | 63.2 | 0.61 (0.4, 1.7) | |||
| Deaths (n) | 8 | 0 | 0.15 (0.9, 1) | |||
| Cases (n) | % | Cases (n) | % | |||
| Clinical syndromes | Meningitis | 22 | 14.5 | 5 | 13.2 | 0.83 (0.3, 2.5) |
| BP | 35 | 23 | 6 | 15.8 | 0.33 (0.2, 1.6) | |
| PE | 6 | 3.9 | 7 | 18.4 | <0.005 (1.7, 17.5) | |
| Empyema | 7 | 4.6 | 4 | 10.5 | 0.23 (0.7, 8.8) | |
| Bacteremia without a focus | 82 | 53.9 | 16 | 42.1 | 0.19 (0.3, 1.3) | |
Patients were stratified by age group: aged <2 years (59.2% in pre-PCV13 period vs 42.1% in post-PCV13 period); aged 2–5 years (28.9% vs 44.7%); and aged >5 years (11.8% vs 13.2%). The 8 deaths registered occurred during the pre-PCV13 period and mostly in females (60%): a 14 months female, a 10 months male, a 43 months female, and a 119 months female (having an underlying Mondini malformation) died from meningitis; a 11 months female (having an underlying bronchopulmonary dysplasia) and a 10 months female (having an underlying encephalopathy with a non-filiated genetic syndrome) died from BP; a 33 months female died from PE and a 127 months male (having an underlying leukaemia) died from bacteremia.
We found a 66.4% reduction (p<0.001) in the overall incidence rate: from 13.1 (95% CI: 11.2, 15.2)/100,000 in pre-PCV13 period to 4.4 (95% CI: 3.2, 6)/100,000 in post-PCV13 period. This decrease was observed in every age group: from 59.9 (95% CI: 48.8, 73.5)/100,000 to 18.9 (95% CI: 11.8, 30.4)/100,000 (68.4% reduction, p<0.001) in aged <2 years; from 20.4 (95% CI: 15.5, 26.9)/100,000 to 9.9 (95% CI: 6.1, 16.2)/100,000 (51.5% reduction, p=0.012) in aged 2–5 years; and from 2.4 (95% CI: 1.5, 3.7)/100,000 to 0.8 (95% CI: 0.3, 1.9)/100,000 (66.6% reduction, p=0.031) in aged >5 years.
The distribution of clinical syndromes among periods was as follows: meningitis (14.5% vs 13.2%), BP (23% vs 15.8%), PE (3.9% vs 18.4%), empyema (4.6% vs 10.5%) and bacteremia without a focus (53.9% vs 42.1%). Variations in BP, empyemas, bacteremia without a focus, and meningitis were not statistically significant. On the other hand, increase of PEs was statistically significant (p<0.005; 95% CI: 1.7, 17.5) but, when studying cases according to group age, this increase was only observed in children aged <2 years (p=0.002; 95% CI: 3, 287.9).
5 vaccine failures were observed in pre-PCV13 period (by ST6B, 9V, 14, 18C and 23F) vs 1 (by ST3) in post-PCV13 period. 52 patients did not have the appropriate age for vaccination or there was no vaccine available at that time. From the remaining 138, 80 (58%) were immunized (51 with PCV7 and 29 with PCV13) but only 27 (33.7%) had their age-appropriate doses. In a previous study,3 PCV7 vaccination coverage in Gran Canaria was reported to be only 25% in 2003 and 48% in 2006. On the other hand, the estimated PCV13 vaccination coverage in 2014 was 49.15% (according to sales data supplied by Wyeth) but after introduction of PCV13 in the publicly funded immunization schedule in our region, it reached 82.2% in 2015 and 96.2% in 2016 (data supplied by the Public Health Directorate of the Canary Islands Health Service).
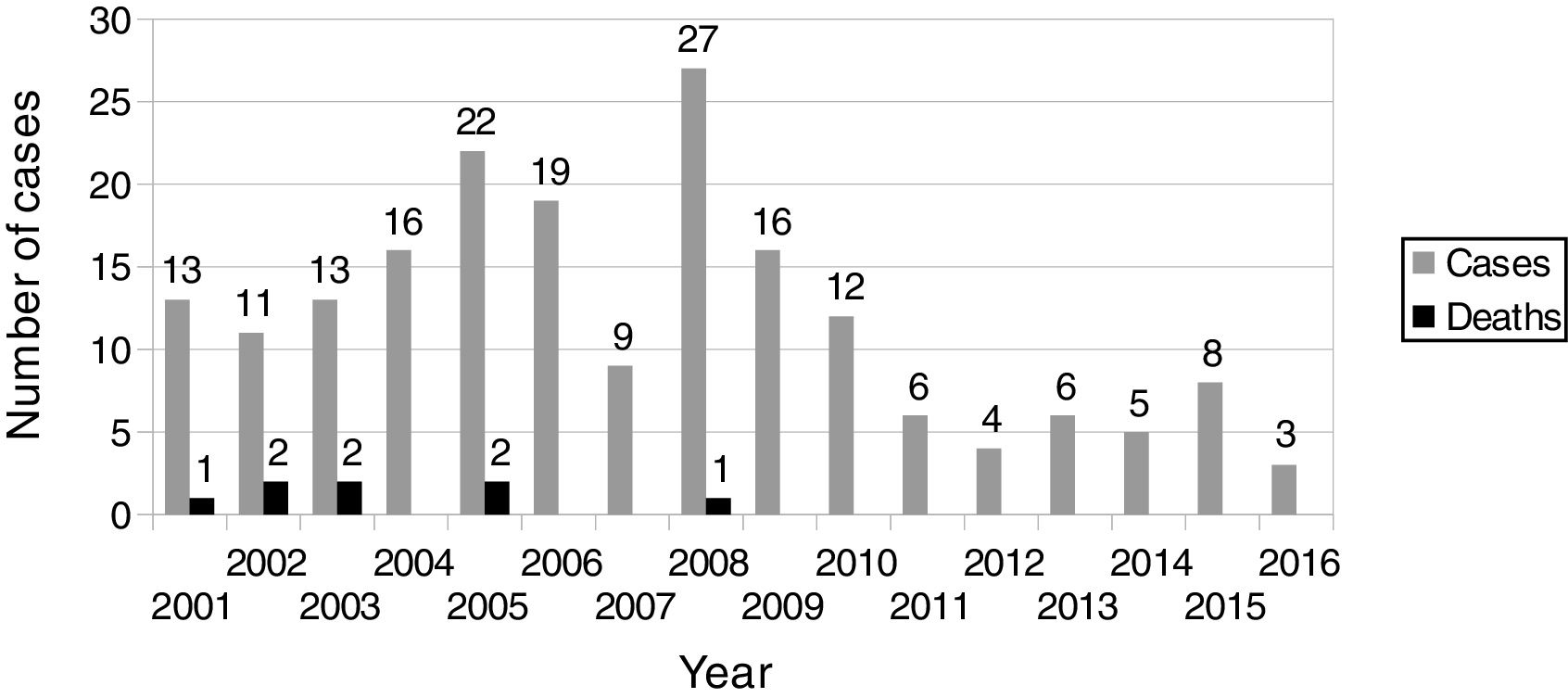
Annual distribution of IPD cases is showed in Fig. 1. When analysing data year by year, we found that in 2007 none of the 9 IPD cases in that year had a PCV7-included ST and, in 2008, when cases were three times the figure of the previous year, only 33% were PCV7-included STs. Moreover, there was a dramatic increase in the number of ST19A cases (10) but, since then, a decline in both ST19A and ST1 was observed, specially after introduction of PCV13 in June 2010. In 2011, the number of recorded IPD cases was the half of that in 2010 (6 vs 12) and the lowest number of cases in our series (4) occurred in 2012.
The most common STs in pre-PCV13 period were ST19A (23.9% of serotyped cases) and ST14 (14.2%); and ST19A, ST23B and ST24F (12.5% each) in post-PCV13 period. STs showing higher increases between periods were ST24F (0.9% vs 12.5%), ST23B (5.3% vs 12.5%) and ST3 (2.7% vs 9.4%) whereas STs showing higher decreases were ST19A (23.9% vs 12.5%), ST14 (14.2% vs 9.4%) and ST19F (6.2% vs 0%).
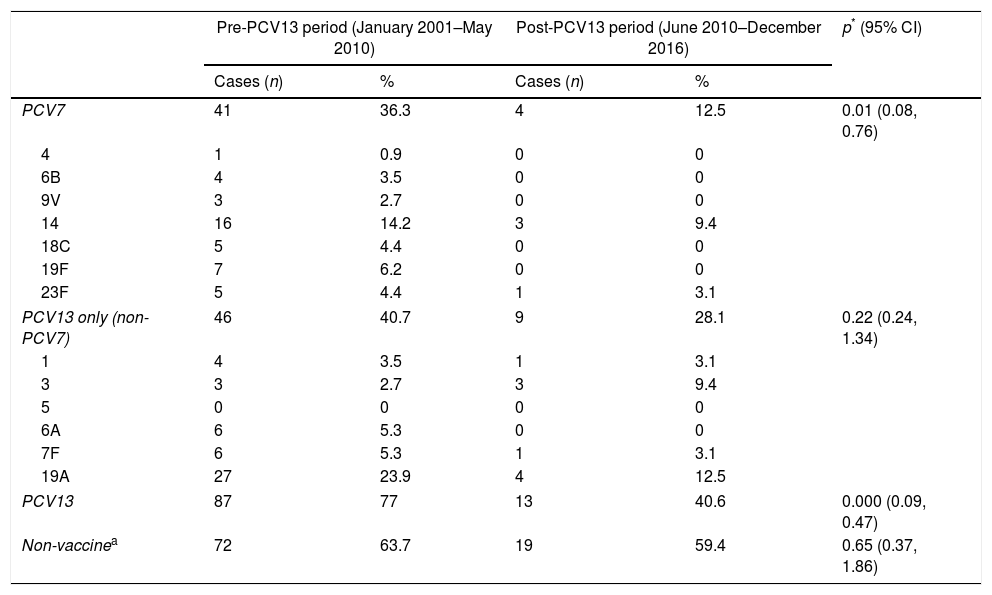
Vaccine ST distribution among periods is showed in Table 2. PCV7-included STs were responsible of 36.3% of IPD cases in pre-PCV13 period vs 12.5% in post-PCV13 period (p<0.001; 95% CI: 0.1, 0.8) whereas PCV13-included STs were responsible of 77% of IPD cases in pre-PCV13 period vs 40.6% in post-PCV13 period (p=0.000; 95% CI: 0.1, 0.7). Non-vaccine STs also decreased from 63.7% in pre-PCV13 period to 59.4% in post-PCV13 period.
Distribution of vaccine serotypes among periods.
| Pre-PCV13 period (January 2001–May 2010) | Post-PCV13 period (June 2010–December 2016) | p* (95% CI) | |||
|---|---|---|---|---|---|
| Cases (n) | % | Cases (n) | % | ||
| PCV7 | 41 | 36.3 | 4 | 12.5 | 0.01 (0.08, 0.76) |
| 4 | 1 | 0.9 | 0 | 0 | |
| 6B | 4 | 3.5 | 0 | 0 | |
| 9V | 3 | 2.7 | 0 | 0 | |
| 14 | 16 | 14.2 | 3 | 9.4 | |
| 18C | 5 | 4.4 | 0 | 0 | |
| 19F | 7 | 6.2 | 0 | 0 | |
| 23F | 5 | 4.4 | 1 | 3.1 | |
| PCV13 only (non-PCV7) | 46 | 40.7 | 9 | 28.1 | 0.22 (0.24, 1.34) |
| 1 | 4 | 3.5 | 1 | 3.1 | |
| 3 | 3 | 2.7 | 3 | 9.4 | |
| 5 | 0 | 0 | 0 | 0 | |
| 6A | 6 | 5.3 | 0 | 0 | |
| 7F | 6 | 5.3 | 1 | 3.1 | |
| 19A | 27 | 23.9 | 4 | 12.5 | |
| PCV13 | 87 | 77 | 13 | 40.6 | 0.000 (0.09, 0.47) |
| Non-vaccinea | 72 | 63.7 | 19 | 59.4 | 0.65 (0.37, 1.86) |
It was observed a decrease in the percentage of serotyped cases caused by vaccine STs for bacteremia without a focus (72.1% vs 40%), PE (100% vs 20%), BP (85.7% vs 33.4%), meningitis (50.2% vs 20%) and empyemas (100% vs 66.6%). However, only 14.2% of empyema cases in pre-PCV13 period were serotyped. The most common STs in bacteremia cases were ST19A (23% vs 13.3%), ST14 (9.8% vs 13.3%) and ST23B (9.8% vs 13.3%). In meningitis, 39.1% of serotyped cases were produced by PCV7-included STs, the most common STs being ST14 and ST18C (13% of serotyped meningitis in both cases). However, they all occurred in pre-PCV13 period whereas none of the serotyped cases in post-PCV13 period were produced by PCV7-included STs. In BP, the overall most common STs were ST19A (29.4% of BP serotyped cases), ST14 (17.6%), ST 19F (8.8%) and ST24F (8.8%) but with ST19A not being responsible of any case in post-PCV13 period.
No resistant cases were found for cefotaxime whereas 9.9% in pre-PCV13 period and 2.6% in post-PCV13 period showed intermediate sensitivity and 90.1% vs 97.4% were sensitive. Overall, 38.88% of all erythromycin resistant cases were attributable to only two STs: ST19A and ST14. Among periods, 57.2% vs 7.9% (p=0.000; 95% CI: 4.6, 53) showed resistance, 17.1% vs 47.4% showed intermediate sensitivity and 25.7% vs 44.7% were sensitive. 5.3% vs 0% were resistant to penicillin, 35.5% vs 5.3% showed intermediate sensitivity and 59.2% vs 94.7% were sensitive.
DiscussionA total of 190 cases were recorded, mostly in males aged <2 years and during autumn-winter. This trend was observed in both periods although the mean age slightly increased in the post-PCV13 period.
Since IPD was not a “reportable disease” in Spain prior to the introduction of the different vaccines, no precise data are available on its incidence at that time but some hospital studies estimated it among 31.1–58.8/100,000 in children aged <5 years4–7 whereas a study in the island of Gran Canaria placed the incidence in 52.7/100,000 in children aged <2 years in 2000.3
Two years after PCV7 introduction in the Spanish regions of the Basque Country and Navarre, with an estimated coverage of 28–45%, the incidence of IPD was reduced by 40% in children aged <2 years and by 37% in children aged <5 years.8 In Gran Canaria, with an estimated coverage of 25%, incidence of IPD reduced by 42.5% in children aged <2 years but increased by 6.3% in children aged <14 years. However, when the incidence in the pre-vaccination period was compared to that in the post-vaccination period until 2006 (with an estimated coverage of 48%), an increase was observed both in children aged <2 years (7.4%) and aged <14 years (27.9%).3 In other Spanish regions, however, there were few or no differences between the pre-vaccination period and the first post-vaccination years due to the increase in non-PCV7 STs, specially 7F and 19A, although vaccination rates were as low as 30% in some of these studies.9,10
Following introduction of PCV13, generalized reductions in the incidence of IPD in Spain started to be observed.10,11 In this study, we found an incidence in the island of Gran Canaria of 13.17/100,000 in pre-PCV13 period (59.9 in <2 years, 20.4 in 2–5 years, 2.4 in >5 years) vs 4.4/100,000 in post-PCV13 period (18.9 in <2 years, 9.9 in 2–5 years, 0.8 in >5 years). Incidence therefore reduced among periods both globally (66.4%) and in every age group (68.4% in <2 years, 51.1% in 2–5 years and 66.6% in >5 years). These data are in line with those reported in other countries where a reduction in the incidence of IPD has been observed when compared with the average incidence in the years preceding PCV13 introduction.12–16
Bacteremias without a focus, BP and meningitis decreased from pre-PCV13 period to post-PCV13 period whereas empyemas increased but these variations did not reach statistical significance. On the other hand, there was an increase of PEs, specially in children aged <2 years (p=0.002; 95% CI: 3, 287.9). In other countries, however, empyema and PE incidences decreased after PCV13 introduction.17
During the study, there were different vaccines licensed in our region. However, they were only available for private purchase during most of the study (until March 2015) and publicly funded only onwards. In a previous study,3 PCV7 vaccination coverage in Gran Canaria was reported to be only 25% in 2003 and 48% in 2006. According to sales data supplied by Wyeth, the estimated PCV13 vaccination coverage in 2014 had only reached 49.15%. A possible explanation to the increase in PEs and empyemas may be a low vaccination rate in our island which would have maintained the secular trend of increasing cases in the last decades.18,19 A total of 6 vaccine failures (ST3, 6B, 9V, 14, 18C, 23F) were observed, only one in post-PCV13 period due to ST3 for which a lower effectiveness of PCV13 has been suggested.16
PCV7-included STs have clearly decreased from pre-PCV13 period to post-PCV13 period (36.3% vs 12.5%) and STs added in PCV13 also decreased, but this reduction did not reach statistical significance (40.7% vs 28.1%). STs not included in any of the available vaccines at the time also decreased from 63.1% in pre-PCV13 period to 52.4% in post-PCV13 period. Changes in ST distribution between periods and among clinical syndromes have also been observed although the number of serotyped cases is too low to form a judgement.
Finally, a decrease in resistance to erythromycin (p=0.000; 95% CI: 4.6, 53) was observed. These differences in erythromycin resistance are probably explained by the decrease of ST19A and ST14 during post-PCV13 period.
Since the Spanish health system offers universal and free coverage and our hospital is the only paediatric tertiary referral hospital in the island, it is to be expected that virtually every paediatric IPD case occurring in our island during the study has been included. Therefore, it is quite likely that the incidence figures and the other results presented here are not just “estimations” but a true picture of the actual evolution of paediatric IPD in our island.
On the other hand, the relatively low vaccination coverage and the small number of STs, make difficult to determine to what extent the introduction of vaccination is responsible of these changes and to interpret changes in the distribution of individual STs. However, from March 2015, pneumococcal vaccination is included in the publicly-funded, universal immunization schedule in our region. This will highly increase vaccination coverage in subsequent years.
As far as we know, this is the first study to calculate the incidence of paediatric IPD in the island of Gran Canaria after PCV13 introduction and to describe its clinical and epidemiological characteristics, ST distribution, and antibiotic resistance. Additionally, it evaluates changes in these variables among the different study periods and it will conform a baseline to keep on monitoring these variables in subsequent years when vaccination coverage will increase.
FundingNone.
Conflicts of interestThe authors declare no conflicts of interest.