Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the causal agent of coronavirus disease 2019 (COVID-19). Acute respiratory distress syndrome is the main cause of death from COVID-19 and occurs due to an exaggerated inflammatory response that causes the release of pro-inflammatory cytokines such as interleukins and tumor necrosis factor-alpha (TNF-α). Statins are lipid lowering drugs with pleiotropic effects. They have shown benefit in the management of inflammatory and autoimmune diseases such as systemic lupus erythematosus, rheumatoid arthritis and multiple sclerosis. Furthermore, due to their immunomodulatory properties, they have been used in the treatment of various infectious diseases such as community-acquired pneumonia and influenza. In this review we analyze the pathophysiological foundations that support the use of statins as an adjunctive treatment in patients with COVID-19.
El coronavirus tipo 2 del síndrome respiratorio agudo grave (SARS-CoV-2) es el agente causal de la enfermedad por coronavirus 2019 (COVID-19). El síndrome de distress respiratorio agudo constituye la principal causa de muerte por COVID-19 y ocurre por una respuesta inflamatoria exagerada que provoca la liberación de citocinas proinflamatorias como interleucinas y factor de necrosis tumoral alfa (TNF-α). Las estatinas son fármacos hipolipemiantes con efectos pleiotrópicos. Han demostrado beneficio en el manejo de enfermedades inflamatorias y autoinmunes como el lupus eritematoso sistémico, la artritis reumatoide y la esclerosis múltiple. Además, debido a sus propiedades inmunomoduladoras se han utilizado en el tratamiento de diversas enfermedades infecciosas como neumonía adquirida en la comunidad e influenza. En esta revisión analizamos los fundamentos fisiopatológicos que apoyan el uso de estatinas como tratamiento coadyuvante en pacientes con COVID-19.
In December 2019, an outbreak of acute respiratory disease characterized by fever, dry cough and shortness of breath occurred in China. A new coronavirus was later identified, termed severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2), which causes coronavirus disease 2019 (COVID-19).1
Acute respiratory distress syndrome (ARDS) is the leading cause of death from COVID-19 and occurs as a consequence of an exaggerated inflammatory response that causes the release of pro-inflammatory cytokines such as interleukins and tumour necrosis factor-alpha (TNF-α). Therefore, the use of anti-inflammatory agents has been proposed in the treatment of these patients.2
Statins are lipid-lowering drugs but are also known for their anti-inflammatory and antioxidant effects.3 As yet, there are no randomised controlled clinical trials that have demonstrated the benefit of statins in patients with COVID-19. However, a multinational observational study involving over 8000 hospitalised patients with COVID-19 showed that the use of statins was associated with improved survival rate in these patients.4 In addition, statins have shown benefit as immunomodulatory drugs in several infectious diseases, including community-acquired pneumonia and influenza.5,6
In this review we will address the rationale behind the judicious use of statins in COVID-19 patients.
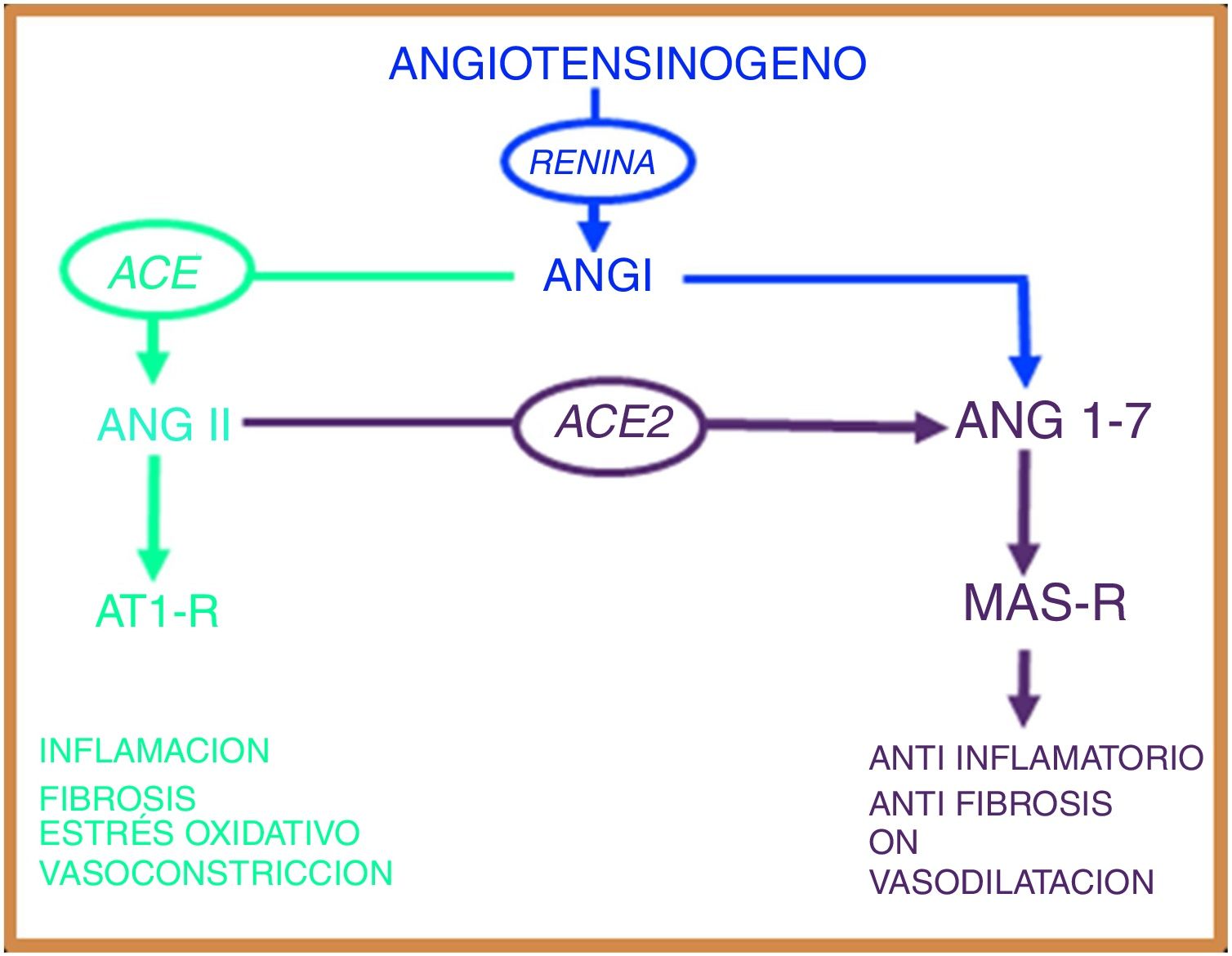
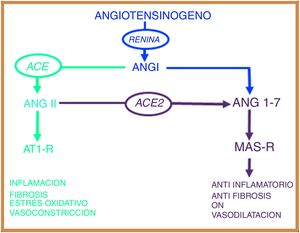
The renin-angiotensin systemThe renin-angiotensin system (RAS) is an elegant cascade of vasoactive peptides that orchestrates key processes in human physiology. Angiotensinogen is a glycoprotein of 452 amino acids produced in the liver and circulates as a biologically inactive peptide. Through the action of renin, angiotensinogen is split into a decapeptide called angiotensin (Ang) I and this in turn is converted by the angiotensin converting enzyme (ACE) into an octapeptide called Ang II, which, by acting on the Ang type 1 receptor (AT1-R), exerts vasoconstrictive and oxidative effects and at lung level induces bronchial smooth muscle contraction, fibroblast proliferation, alveolar epithelial cell apoptosis and increased vascular permeability.7,8
In 2000, two independent groups identified a new ACE homologous enzyme, which they named angiotensin-converting enzyme 2 (ACE2).9,10 This enzyme is 42% homologous with ACE, but with different biochemical activities. ACE2 when hydrolysed to Ang I generates Ang (1–9);9 however, the catalytic activity of ACE2 is 400 times higher over Ang II than over Ang I and leads to the formation of Ang (1–7) with vasodilatory properties through the Mas receptor (MAS-R).7 Thus, the RAS works as a dual endocrine system in which vasoconstrictive/proliferative actions and vasodilatory/antiproliferative actions are regulated by a balance between ACE and ACE2 (Fig. 1).
The renin angiotensin system has two opposing biological pathways: the pro-inflammatory pathway regulated by the angiotensin converting enzyme (ACE) that modulates the angiotensin II peptide (Ang II) and the AT1 receptor (AT1-R). The anti-inflammatory pathway regulated by the angiotensin converting enzyme 2 (ACE2) that modulates the angiotensin peptide (1–7) and the Mas receptor (MAS-r).
Viral infections depend on the virus entering the cell and using the host's cellular machinery to replicate multiple copies that will subsequently infect more cells. The coronaviruses SARS-CoV-1 and SARS-CoV-2 enter host cells using ACE2 as a receptor. ACE2 is expressed in type 1 and type 2 alveolar epithelial cells and has two fractions: a soluble and a membrane-bound fraction.11
SARS-CoV-1 and SARS-CoV-2 express in their shells a protein called S-protein that contains a receptor-binding region that binds with high affinity to the extracellular domain of ACE2, causing membrane fusion and internalization of the virus through endocytosis.12 The internalization of ACE2 by SARS-CoV-2 results in a loss of ACE2 on the cell surface and thus prevents the degradation of Ang II into Ang (1–7), which could contribute to the lung damage and fibrosis associated with COVID-19.13
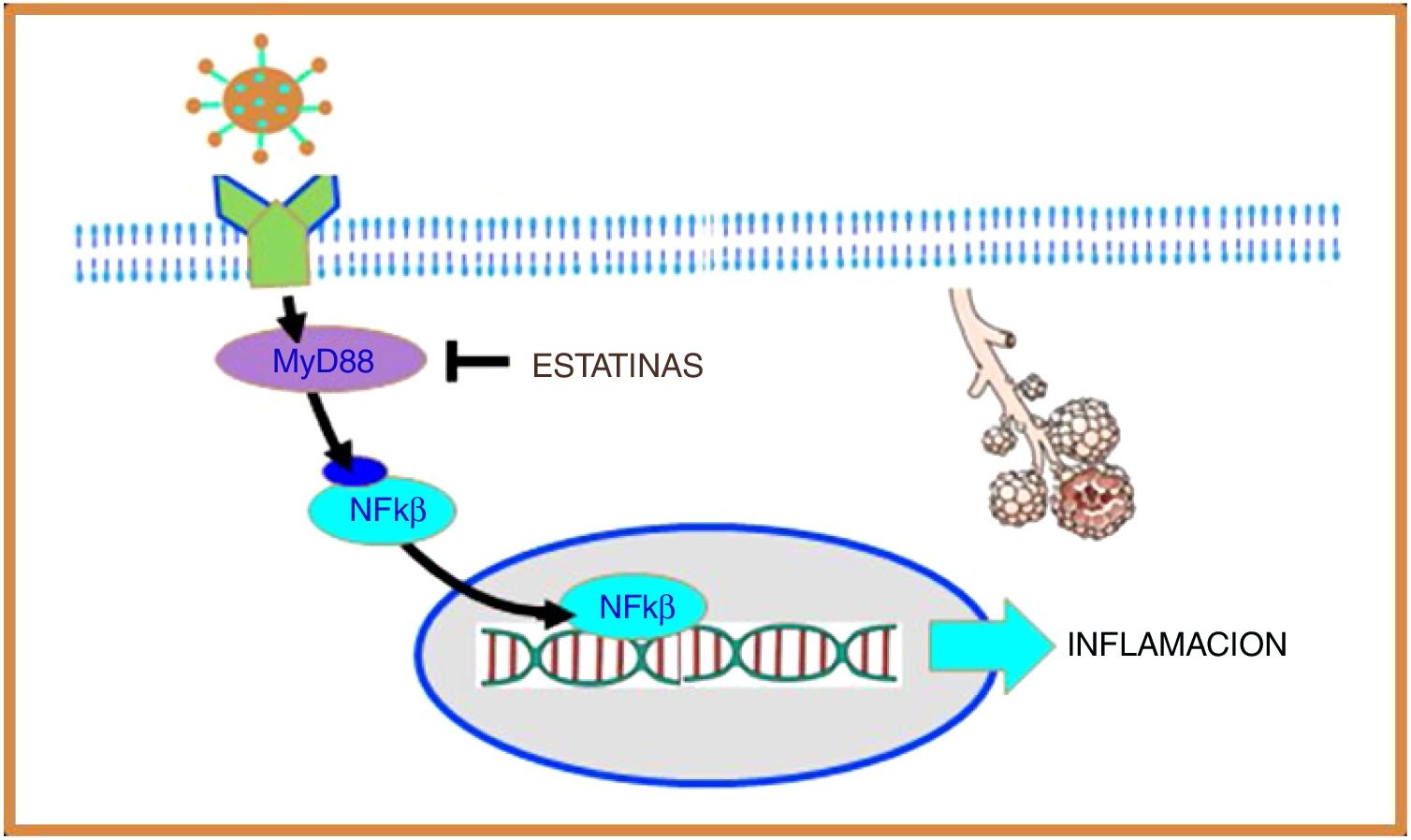
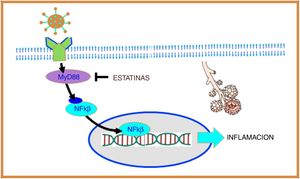
Coronavirus and inflammationToll-like receptors (TLRs) are a family of proteins that act as sensors and help the immune system discriminate between foreign and domestic elements. SARS-CoV-1 and, presumably, SARS-CoV-2 interact with TLRs on the host cell membrane and increase the expression of the primary myeloid differentiation response gene 88 (MyD88), which in turn activates nuclear factor kappa B (NF-κB), eventually promoting an inflammatory cascade that increases lung damage14 (Fig. 2).
SARS-CoV-2 interacts with toll-like receptors (TLRs) on the host cell membrane and increases the expression of the primary myeloid differentiation response gene 88 (MyD88), which in turn activates nuclear factor kappa B (NF-B), promoting inflammation. Statins suppress the expression of MyD88, thereby suppressing the activation of NF-B, exerting an anti-inflammatory effect.
In the coronavirus causing Middle East acute respiratory syndrome (MERS-CoV) it was observed that increased expression of the MyD88 gene was associated with increased mortality.15 Moreover, in a murine model of SARS-CoV-1 it was shown that inhibition of NF-κB resulted in less lung damage and increased survival, and therefore this mechanism appears to be of particular importance in coronavirus infection.16
Pathophysiological basis for the use of statins in COVID-19Statins exert pleiotropic effects and there are at least 4 reasons that support the use of statins in patients with COVID-19:
- 1)
Experimental models have shown that statins preserve normal levels of MyD88 during hypoxia, thereby inhibiting NF-κB activation, exerting an anti-inflammatory effect17 (Fig. 2).
- 2)
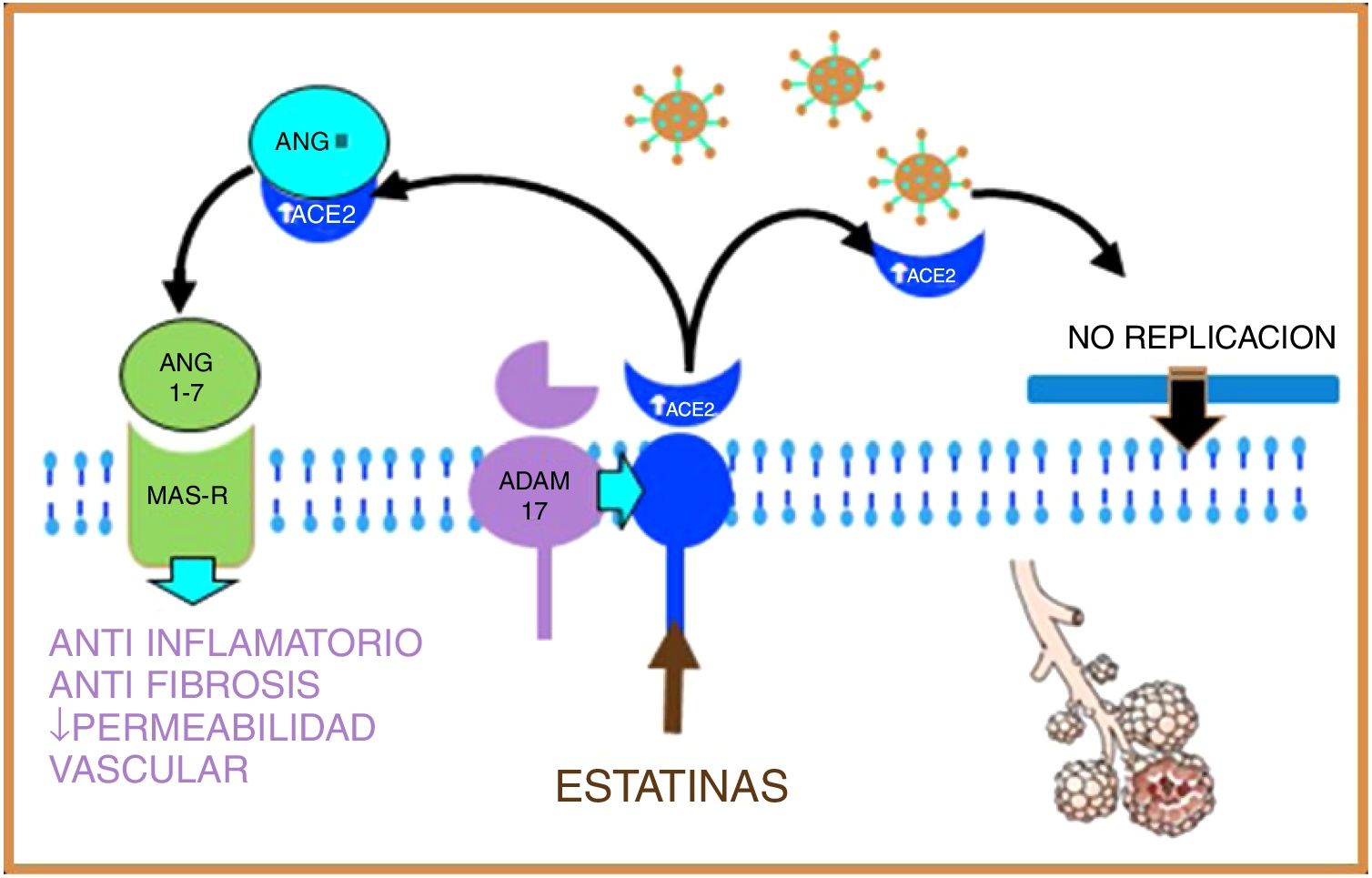
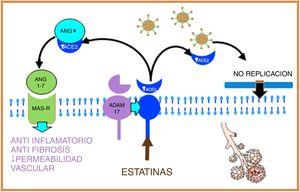
Statins are able to induce positive regulation of ADAM17 through epigenetic pathways, thus increasing levels of soluble ACE2, stimulating on the one hand the protective system of Ang (1–7), and MAS-R, thus decreasing pulmonary fibrosis and exerting a cardioprotective effect; on the other hand, soluble ACE2 could bind to SARS-CoV-2, preventing its fusion with the host cell membrane and therefore inhibiting viral replication18,19 (Fig. 3).
Figure 3.Statins, by increasing the production of soluble ACE2 through epigenetic pathways, will stimulate, on the one hand, the protective system of Ang (1–7) and MAS-r and on the other hand, soluble ACE2 binds to SARS-CoV-2, preventing its fusion with the membrane and, therefore, inhibiting viral replication.
(0.11MB). - 3)
Statins could directly affect SARS-CoV-2 by binding to the main protease of the virus, in a similar way to protease inhibitors.20
- 4)
Statins are effective in the treatment of hyperlipidaemia associated with the use of antiretrovirals as protease inhibitors or immunosuppressive drugs that could be used in the management of patients with COVID-19.19
There are insufficient studies to date on the effect of SARS-CoV-2 on the liver, but it is plausible that it also uses ACE2 to enter these cells. In fact, COVID-19 is associated with abnormal liver tests in 15%–53% of patients, the most common abnormality being elevated aminotransferases (AST and ALT), with occasional increases in alkaline phosphatase and total bilirubin.21
Statins are associated with mild and transient elevation of aminotransferases in 1%–3% of patients and increases of three times the upper normal limit only in 0.7% of cases.22 We highlight one study that compared 342 patients with liver profile abnormalities and 1437 patients with no abnormalities who started treatment with statins, and observed no statistically significant differences between the groups in the development of severe liver disease,23 and therefore use of statins in patients with COVID-19 could be safe.
ConclusionsStatins are well-studied, low-cost, and generally well-tolerated drugs that could be used as adjuvant treatment in patients with COVID-19 due to their immunomodulatory, anti-inflammatory and cardioprotective effects. Randomised controlled clinical trials are required to confirm these hypotheses.
FundingThis research study has received no specific support from public sector agencies, commercial sector, or non-profit entities.
Conflict of interestsThe authors have no conflict of interests to declare.
Please cite this article as: Lima Martínez MM, Contreras MA, Marín W, D’Marco L. Estatinas en COVID-19: ¿existe algún fundamento? Clin Investig Arterioscler. 2020. https://doi.org/10.1016/j.arteri.2020.06.003