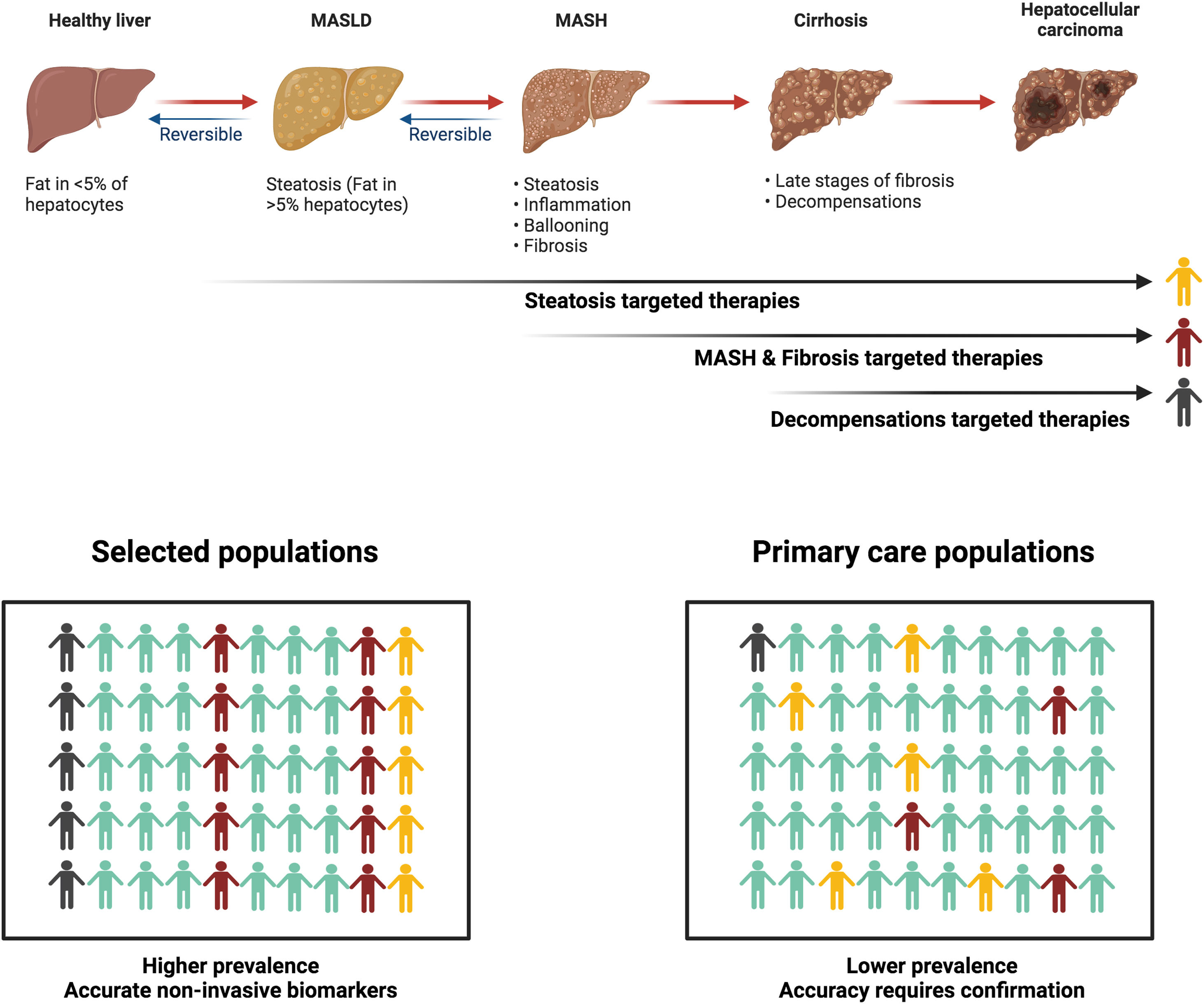
array:23 [ "pii" => "S0210570524000530" "issn" => "02105705" "doi" => "10.1016/j.gastrohep.2024.02.004" "estado" => "S300" "fechaPublicacion" => "2024-04-01" "aid" => "2173" "copyright" => "Elsevier España, S.L.U.. All rights reserved" "copyrightAnyo" => "2024" "documento" => "article" "crossmark" => 1 "subdocumento" => "sco" "cita" => "Gastroenterol Hepatol. 2024;47:393-6" "abierto" => array:3 [ "ES" => false "ES2" => false "LATM" => false ] "gratuito" => false "lecturas" => array:1 [ "total" => 0 ] "itemSiguiente" => array:17 [ "pii" => "S021057052300393X" "issn" => "02105705" "doi" => "10.1016/j.gastrohep.2023.08.006" "estado" => "S300" "fechaPublicacion" => "2024-04-01" "aid" => "2111" "documento" => "article" "crossmark" => 1 "subdocumento" => "ssu" "cita" => "Gastroenterol Hepatol. 2024;47:397-400" "abierto" => array:3 [ "ES" => false "ES2" => false "LATM" => false ] "gratuito" => false "lecturas" => array:1 [ "total" => 0 ] "en" => array:13 [ "idiomaDefecto" => true "cabecera" => "<span class="elsevierStyleTextfn">Special article</span>" "titulo" => "<span class="elsevierStyleItalic">MSH3</span>-related adenomatous polyposis in a patient with the negative family history of colorectal polyps" "tienePdf" => "en" "tieneTextoCompleto" => "en" "tieneResumen" => array:2 [ 0 => "en" 1 => "es" ] "paginas" => array:1 [ 0 => array:2 [ "paginaInicial" => "397" "paginaFinal" => "400" ] ] "titulosAlternativos" => array:1 [ "es" => array:1 [ "titulo" => "Poliposis adenomatosa relacionada con MSH3 en un paciente con antecedentes familiares negativos de pólipos colorrectales" ] ] "contieneResumen" => array:2 [ "en" => true "es" => true ] "contieneTextoCompleto" => array:1 [ "en" => true ] "contienePdf" => array:1 [ "en" => true ] "resumenGrafico" => array:2 [ "original" => 0 "multimedia" => array:7 [ "identificador" => "fig0015" "etiqueta" => "Figure 3" "tipo" => "MULTIMEDIAFIGURA" "mostrarFloat" => true "mostrarDisplay" => false "figura" => array:1 [ 0 => array:4 [ "imagen" => "gr3.jpeg" "Alto" => 841 "Ancho" => 2508 "Tamanyo" => 116500 ] ] "descripcion" => array:1 [ "en" => "<p id="spar0025" class="elsevierStyleSimplePara elsevierViewall">A retrospective chronological phenotypic description of our patient.</p>" ] ] ] "autores" => array:1 [ 0 => array:2 [ "autoresLista" => "Aleksandar Gavric, Mateja Krajc, Luka Strnisa, Ana Ursula Gavric, Samo Plut" "autores" => array:5 [ 0 => array:2 [ "nombre" => "Aleksandar" "apellidos" => "Gavric" ] 1 => array:2 [ "nombre" => "Mateja" "apellidos" => "Krajc" ] 2 => array:2 [ "nombre" => "Luka" "apellidos" => "Strnisa" ] 3 => array:2 [ "nombre" => "Ana Ursula" "apellidos" => "Gavric" ] 4 => array:2 [ "nombre" => "Samo" "apellidos" => "Plut" ] ] ] ] ] "idiomaDefecto" => "en" "EPUB" => "https://multimedia.elsevier.es/PublicationsMultimediaV1/item/epub/S021057052300393X?idApp=UINPBA00004N" "url" => "/02105705/0000004700000004/v2_202406091228/S021057052300393X/v2_202406091228/en/main.assets" ] "itemAnterior" => array:18 [ "pii" => "S0210570523004752" "issn" => "02105705" "doi" => "10.1016/j.gastrohep.2023.11.005" "estado" => "S300" "fechaPublicacion" => "2024-04-01" "aid" => "2142" "copyright" => "Elsevier España, S.L.U." "documento" => "simple-article" "crossmark" => 1 "subdocumento" => "cor" "cita" => "Gastroenterol Hepatol. 2024;47:391-2" "abierto" => array:3 [ "ES" => false "ES2" => false "LATM" => false ] "gratuito" => false "lecturas" => array:1 [ "total" => 0 ] "en" => array:11 [ "idiomaDefecto" => true "cabecera" => "<span class="elsevierStyleTextfn">Letter to the Editor</span>" "titulo" => "Ischemic colitis due to Kayexalate crystals" "tienePdf" => "en" "tieneTextoCompleto" => "en" "paginas" => array:1 [ 0 => array:2 [ "paginaInicial" => "391" "paginaFinal" => "392" ] ] "titulosAlternativos" => array:1 [ "es" => array:1 [ "titulo" => "Colitis isquémica por cristales de kayexalato" ] ] "contieneTextoCompleto" => array:1 [ "en" => true ] "contienePdf" => array:1 [ "en" => true ] "resumenGrafico" => array:2 [ "original" => 0 "multimedia" => array:7 [ "identificador" => "fig0005" "etiqueta" => "Figure 1" "tipo" => "MULTIMEDIAFIGURA" "mostrarFloat" => true "mostrarDisplay" => false "figura" => array:1 [ 0 => array:4 [ "imagen" => "gr1.jpeg" "Alto" => 1862 "Ancho" => 2417 "Tamanyo" => 748598 ] ] "descripcion" => array:1 [ "en" => "<p id="spar0005" class="elsevierStyleSimplePara elsevierViewall">(a) Cecum and ascending colon wall thickening in CT scan. (b) Endoscopic ulcers at cecum, ileocecal valve. (c) Ulcerated areas, observing fibrinoleukocyte material and granulation tissue with Kayexalate crystals. (d) Kayexalate crystals amplified in size.</p>" ] ] ] "autores" => array:1 [ 0 => array:2 [ "autoresLista" => "Berta Lopez-Saez, Alex Casalots, Eva Ballesteros, Eduard Brunet-Mas" "autores" => array:4 [ 0 => array:2 [ "nombre" => "Berta" "apellidos" => "Lopez-Saez" ] 1 => array:2 [ "nombre" => "Alex" "apellidos" => "Casalots" ] 2 => array:2 [ "nombre" => "Eva" "apellidos" => "Ballesteros" ] 3 => array:2 [ "nombre" => "Eduard" "apellidos" => "Brunet-Mas" ] ] ] ] ] "idiomaDefecto" => "en" "EPUB" => "https://multimedia.elsevier.es/PublicationsMultimediaV1/item/epub/S0210570523004752?idApp=UINPBA00004N" "url" => "/02105705/0000004700000004/v2_202406091228/S0210570523004752/v2_202406091228/en/main.assets" ] "en" => array:15 [ "idiomaDefecto" => true "cabecera" => "<span class="elsevierStyleTextfn">Young corner</span>" "titulo" => "MASLD biomarkers: Are we facing a new era?" "tieneTextoCompleto" => true "paginas" => array:1 [ 0 => array:2 [ "paginaInicial" => "393" "paginaFinal" => "396" ] ] "autores" => array:1 [ 0 => array:4 [ "autoresLista" => "Douglas Maya-Miles, Javier Ampuero, David Martí-Aguado, Andrés Conthe, Rocío Gallego-Durán" "autores" => array:5 [ 0 => array:3 [ "nombre" => "Douglas" "apellidos" => "Maya-Miles" "referencia" => array:2 [ 0 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">a</span>" "identificador" => "aff0005" ] 1 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">b</span>" "identificador" => "aff0010" ] ] ] 1 => array:3 [ "nombre" => "Javier" "apellidos" => "Ampuero" "referencia" => array:3 [ 0 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">a</span>" "identificador" => "aff0005" ] 1 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">b</span>" "identificador" => "aff0010" ] 2 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">c</span>" "identificador" => "aff0015" ] ] ] 2 => array:3 [ "nombre" => "David" "apellidos" => "Martí-Aguado" "referencia" => array:1 [ 0 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">d</span>" "identificador" => "aff0020" ] ] ] 3 => array:3 [ "nombre" => "Andrés" "apellidos" => "Conthe" "referencia" => array:2 [ 0 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">b</span>" "identificador" => "aff0010" ] 1 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">e</span>" "identificador" => "aff0025" ] ] ] 4 => array:4 [ "nombre" => "Rocío" "apellidos" => "Gallego-Durán" "email" => array:1 [ 0 => "rgallego-ibis@us.es" ] "referencia" => array:3 [ 0 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">a</span>" "identificador" => "aff0005" ] 1 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">b</span>" "identificador" => "aff0010" ] 2 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">*</span>" "identificador" => "cor0005" ] ] ] ] "afiliaciones" => array:5 [ 0 => array:3 [ "entidad" => "SeLiver Group, Instituto de Biomedicina de Sevilla/CSIC/Universidad de Sevilla, Sevilla, Spain" "etiqueta" => "a" "identificador" => "aff0005" ] 1 => array:3 [ "entidad" => "Hepatic and Digestive Diseases Networking Biomedical Research Centre (CIBERehd), Madrid, Spain" "etiqueta" => "b" "identificador" => "aff0010" ] 2 => array:3 [ "entidad" => "Digestive Diseases Unit, Hospital Universitario Virgen del Rocío, Sevilla, Spain" "etiqueta" => "c" "identificador" => "aff0015" ] 3 => array:3 [ "entidad" => "Servicio de Aparato Digestivo, Hospital Clínico Universitario de Valencia, INCLIVA Instituto de Investigación Sanitaria, Valencia, Spain" "etiqueta" => "d" "identificador" => "aff0020" ] 4 => array:3 [ "entidad" => "Sección de Hepatología, Servicio de Aparato Digestivo, Hospital General Universitario Gregorio Marañón, Madrid, Spain" "etiqueta" => "e" "identificador" => "aff0025" ] ] "correspondencia" => array:1 [ 0 => array:3 [ "identificador" => "cor0005" "etiqueta" => "⁎" "correspondencia" => "Corresponding author." ] ] ] ] "titulosAlternativos" => array:1 [ "es" => array:1 [ "titulo" => "Biomarcadores en MASLD, ¿estamos ante una nueva era?" ] ] "resumenGrafico" => array:2 [ "original" => 0 "multimedia" => array:7 [ "identificador" => "fig0005" "etiqueta" => "Figure 1" "tipo" => "MULTIMEDIAFIGURA" "mostrarFloat" => true "mostrarDisplay" => false "figura" => array:1 [ 0 => array:4 [ "imagen" => "gr1.jpeg" "Alto" => 2439 "Ancho" => 2925 "Tamanyo" => 500218 ] ] "descripcion" => array:1 [ "en" => "<p id="spar0005" class="elsevierStyleSimplePara elsevierViewall">Facing a new era in MASLD biomarkers. Progressive forms of liver disease require the use of targeted therapies adapted to stratified groups of patients, considering the main drivers of the disease to define patients’ subphenotypes. Biomarkers have proven diagnostic accuracy in targeted populations (higher prevalence), but require further validation in primary care settings (lower prevalence) for screening purposes.</p>" ] ] ] "textoCompleto" => "<span class="elsevierStyleSections"><span id="sec0005" class="elsevierStyleSection elsevierViewall"><span class="elsevierStyleSectionTitle" id="sect0005">Introduction</span><p id="par0005" class="elsevierStylePara elsevierViewall">During the past decades, dramatic changes in lifestyle have paralleled the worldwide epidemic of obesity, metabolic disease and chronic liver disorders. This is currently considered a major health concern, in which aberrant metabolism is the root cause for a number of severe health conditions that are currently placing a heavy burden on individuals, families, governments, and healthcare systems. In this context, metabolic dysfunction-associated steatotic liver disease (MASLD,<a class="elsevierStyleCrossRef" href="#bib0125"><span class="elsevierStyleSup">1</span></a> formerly known as NAFLD), has emerged as the most predominant chronic liver disorder worldwide. The global prevalence among adults is currently estimated to be 25% in the general population, and is expected to become the leading cause of end-stages liver diseases worldwide.<a class="elsevierStyleCrossRef" href="#bib0130"><span class="elsevierStyleSup">2</span></a> MASLD is considered the hepatic manifestation of metabolic syndrome and is linked to extrahepatic complications including malignancies, chronic kidney disease, endocrinopathies, brain ageing and cognitive impairment.<a class="elsevierStyleCrossRefs" href="#bib0135"><span class="elsevierStyleSup">3–5</span></a> MASLD encompasses a large spectrum of clinical and metabolic phenotypes. Patients can suffer from steatosis, characterized by the accumulation of lipids in the hepatocytes, or a progressive form of the disease so-called metabolic dysfunction associated steatohepatitis (MASH), identified by steatosis, inflammation and ballooning, a form of liver degeneration with cell swelling. MASH is associated with liver fibrosis and progression of liver disease to cirrhosis and hepatocellular carcinoma in ∼3–9% of patients, and consequently has become one of the most prevalent indications for liver transplantation worldwide.<a class="elsevierStyleCrossRef" href="#bib0150"><span class="elsevierStyleSup">6</span></a> Further, hepatic fibrosis strongly correlates with morbidity and has been reported as the best predictor of mortality in MASLD patients.<a class="elsevierStyleCrossRef" href="#bib0155"><span class="elsevierStyleSup">7</span></a> Despite its global burden, there are no effective drugs approved for MASLD treatment other than changes in lifestyle that lead to weight loss.<a class="elsevierStyleCrossRef" href="#bib0160"><span class="elsevierStyleSup">8</span></a> Finally, histological evaluation of the liver is the reference method for diagnosis and disease activity, but requires an invasive liver biopsy that has associated risks and limitations, and therefore, is not adequate for follow-up studies or to monitor disease progression. Hence, improving diagnosis and therapeutic approaches for MASLD is a key health objective.</p></span><span id="sec0010" class="elsevierStyleSection elsevierViewall"><span class="elsevierStyleSectionTitle" id="sect0010">MASLD, a multi-layered disease</span><p id="par0010" class="elsevierStylePara elsevierViewall">Currently, in order to conceive new therapies, there is an urgent need to better understand the pathophysiology behind this disease. Since the term MASLD groups a heterogenous population of patients, it is essential to stratify them following the main driver/drivers of the disease, creating patients’ sub-phenotypes based on personalized medicine strategies. MASLD is the result from a complex and dynamic interaction of several modifiable and non-modifiable factors, including sex, ethnicity, diet patterns and physical activity, comorbidities, genetic polymorphisms, alcohol intake, microbiota and socioeconomic status, and this heterogeneity will lead to different natural history, prognosis and eventually, response to therapy.<a class="elsevierStyleCrossRef" href="#bib0165"><span class="elsevierStyleSup">9</span></a> Thus, it has been suggested that the main factors that shape heterogeneity in MASLD need to be considered when devising and applying risk-stratification scores and algorithms to diagnose MASLD.<a class="elsevierStyleCrossRef" href="#bib0170"><span class="elsevierStyleSup">10</span></a> Further, availability and cost of the techniques are equally important, since the pragmatic utilization is key to avoid ineffective referrals, long waiting lists and the collapse of health resources.</p></span><span id="sec0015" class="elsevierStyleSection elsevierViewall"><span class="elsevierStyleSectionTitle" id="sect0015">MASLD, the search for non-invasive approaches</span><p id="par0015" class="elsevierStylePara elsevierViewall">Current available non-invasive tools and scores are quite efficient in detecting patients with advanced liver fibrosis, which is a good indicator of disease severity, as it will not only increase the odds of promoting liver-related complications (i.e. cirrhosis, hepatocellular carcinoma and liver failure), but also the chances of deaths that cannot be directly attributed to liver dysfunction.<a class="elsevierStyleCrossRef" href="#bib0175"><span class="elsevierStyleSup">11</span></a> These tools, however, leave a large proportion of individuals with moderate disease undetected, because no simple and efficient strategy is available for early screen in the general population. These patients in turn only get diagnosed when advanced liver disease is present and complications are more likely to take place, limiting the potential benefits of treatment and lifestyle advice, so early detection tools are necessary. The identification of significant fibrosis and MASH (≥F2 fibrosis, NAFLD activity score [NAS]<span class="elsevierStyleHsp" style=""></span>><span class="elsevierStyleHsp" style=""></span>4) in high-risk patients is also of utmost importance for given prognosis, correlation with progression to cirrhosis and major adverse liver outcomes.<a class="elsevierStyleCrossRefs" href="#bib0180"><span class="elsevierStyleSup">12,13</span></a></p></span><span id="sec0020" class="elsevierStyleSection elsevierViewall"><span class="elsevierStyleSectionTitle" id="sect0020">Comparative study of the new horizons</span><p id="par0020" class="elsevierStylePara elsevierViewall">Two of the biggest Consortia aiming to find new biomarkers for MASLD diagnosis and progression, the Liver Investigation: Testing Marker Utility in Steatohepatitis (LITMUS) and the Non-Invasive Biomarkers of Metabolic Liver Disease (NIMBLE), have recently published their primary project results. LITMUS has evaluated in approximately 1000 patients coming from 13 European countries, a panel of 17 non-invasive biomarkers, including serological and imaging approaches, together with multimarker scores chosen based on their performance in identifying either fibrosis or MASH.<a class="elsevierStyleCrossRef" href="#bib0190"><span class="elsevierStyleSup">14</span></a> On the other hand, the NIMBLE study tested the efficacy of five serological biomarker panels’ diagnostic performance (NIS4, OWLiver, PROC3, ELF and FibroMeter VCTE) for the diagnosis of MASH, high NAS Score rates and severity of liver fibrosis in MASLD patients in USA (<span class="elsevierStyleItalic">n</span><span class="elsevierStyleHsp" style=""></span>=<span class="elsevierStyleHsp" style=""></span>1.073).<a class="elsevierStyleCrossRef" href="#bib0195"><span class="elsevierStyleSup">15</span></a></p><p id="par0025" class="elsevierStylePara elsevierViewall">Analyses in these large multinational cohorts indicated that <span class="elsevierStyleBold">NIS4,</span><span class="elsevierStyleBold">SomaSignal</span> and the <span class="elsevierStyleBold">FAST algorithm</span> are good tools to discriminate patients with at-risk MASH, which is logical if we consider that all of them were specifically designed with this purpose. <span class="elsevierStyleBold">NIS4</span> is a proprietary technology that utilizes a panel of four independent biomarkers (miR-34a-5p, alpha-2 macroglobulin [A2M], glycated haemoglobin [HbA1c], and chitinase-3-like protein 1 [CHI3L1; also known as YKL-40]) specifically designed for detecting at-risk MASH in patients with metabolic risk factors. NIS4 value lower than 0.36 ruled out at-risk MASH with 81.5% sensitivity and 63.0% specificity and a negative predictive value (NPV) of 77.9%. On the other hand, NIS4 values higher than 0.63 ruled-in at-risk MASH patients with 87.1% specificity, and 50.7% sensitivity, and a positive predictive value (PPV) of 79.2%.<a class="elsevierStyleCrossRef" href="#bib0200"><span class="elsevierStyleSup">16</span></a> NIS4 ability to detect at-risk MASH has been recently validated in an additional cohort from the RESOLVE-IT trial (0.792 [0.772–0.811], <span class="elsevierStyleItalic">N</span><span class="elsevierStyleHsp" style=""></span>=<span class="elsevierStyleHsp" style=""></span>2.035) in which a simpler where A2M and HbA1c are no longer required, and sex was incorporated into the equation. This NIS2+, demonstrated a similar ability to discriminate at-risk NASH (0.813 [0.795, 0.832]).<a class="elsevierStyleCrossRef" href="#bib0205"><span class="elsevierStyleSup">17</span></a> In NIMBLE report, NIS4 was the only panel with an intended use to diagnose the underlying composite phenotype of at-risk MASH (<span class="elsevierStyleItalic">n</span><span class="elsevierStyleHsp" style=""></span>=<span class="elsevierStyleHsp" style=""></span>539 within the full cohort). The sensitivity and specificity of NIS4 were 78.1% and 73.6%, respectively, with an AUROC of 0.815, which was superior to other two scores, ALT (AUROC<span class="elsevierStyleHsp" style=""></span>=<span class="elsevierStyleHsp" style=""></span>0.726) and FIB-4 (AUROC<span class="elsevierStyleHsp" style=""></span>=<span class="elsevierStyleHsp" style=""></span>0.704). NIS4 also obtained the best ability to detect significant fibrosis [0.874 (0.848, 0.899)] but not advanced fibrosis or cirrhosis, in which Fibrometer VCTE achieved the highest performance [F3: AUC 0.858 (0.814–0.902) and F4 AUC 0.897 (0.843–0.951)] followed by ELF [F3: AUC: 0.835 (0.807–0.863) and F4: AUC 0.855 (0.818–0.892)]. In this cohort, and since OWLiver provides the results in categorical format, not permitting the generation of an AUROC; it was reported a sensitivity of 77.3% and a specificity of 66.8% for at-risk MASH. The AUROCs of the ELF test, PROC3 and FibroMeterVCTE for significant fibrosis, advanced fibrosis or cirrhosis were all ≥0.8.</p><p id="par0030" class="elsevierStylePara elsevierViewall"><span class="elsevierStyleBold">SomaSignal</span> is a proprietary test that relies on chemically modified nucleotides able to bind to proteins for the “identification” and the amplification of reporter sequences by quantitative real-time PCR. Proteins included for at-risk MASH detection have been identified through a training and validation of whole arrays of proteins against biopsy results for each component (steatosis [12 protein analytes], lobular inflammation [14 protein analytes], hepatocellular ballooning [five protein analytes], and fibrosis [eight protein analysts]).<a class="elsevierStyleCrossRefs" href="#bib0210"><span class="elsevierStyleSup">18,19</span></a> LITMUS identified SomaSignal as the best method to diagnose MASH [AUC 0.81 [0.75–0.86)] and advanced fibrosis [AUC 0.90 [0.86–0.94)], but this was analyzed in a relatively small subset of patients (<span class="elsevierStyleItalic">n</span><span class="elsevierStyleHsp" style=""></span>=<span class="elsevierStyleHsp" style=""></span>264). This test, however, achieved better results during its original training and validation for at-risk MASH detection (training AUC: 0.86; validation AUC: 0.91) in a larger population (<span class="elsevierStyleItalic">n</span><span class="elsevierStyleHsp" style=""></span>=<span class="elsevierStyleHsp" style=""></span>636). ADAPT also reached a good accuracy for advanced fibrosis detection [AUC 0.85 (0.81–0.89)] together with FibroScan [AUC 0.83 (0.80–0.86)]. <span class="elsevierStyleBold">FibroScan-AST (FAST</span>) algorithm is a combination of VCTE with AST which originally obtained a first <span class="elsevierStyleItalic">C</span>-statistic of 0.80 [0.76–0.85) for at-risk MASH detection, quite consistent across the validation cohorts, in which it was assessed (<span class="elsevierStyleItalic">C</span>-statistic range 0.74–0.95, 0.85; 95% CI 0.83–0.87, <span class="elsevierStyleItalic">N</span><span class="elsevierStyleHsp" style=""></span>=<span class="elsevierStyleHsp" style=""></span>1026)<a class="elsevierStyleCrossRef" href="#bib0220"><span class="elsevierStyleSup">20</span></a> and when was validated in the NASH-CRN cohort in an independent study (AUC: 0.81 [0.77–0.84]). Usual cutoffs include 0.35 for high sensitivity and 0.67 for high specificity. A recent comprehensive analysis of screening data from seven non-cirrhotic phase 2 trials for MASH including 2173 individuals with centrally assessed liver biopsies revealed as AST levels, FAST-score, FIB-4 and HbA1c levels key independent predictors. This study recommended the use of the FAST-score, with a suggested cutoff point of 0.5 for patients with HbA1c<span class="elsevierStyleHsp" style=""></span>≥<span class="elsevierStyleHsp" style=""></span>6.5% and 0.67 for patients with HbA1c<span class="elsevierStyleHsp" style=""></span><<span class="elsevierStyleHsp" style=""></span>6.5%, as a simple and effective tool for stratifying at-risk MASH patients.</p><p id="par0035" class="elsevierStylePara elsevierViewall">Other promising new biomarkers for at-risk MASH including the promising MRI-aspartate aminotransferase score (MAST)<a class="elsevierStyleCrossRef" href="#bib0225"><span class="elsevierStyleSup">21</span></a>(AUC: 0.93 [0.88–0.97]), or alternative combinations of routine analytics obtained with circulating proteins<a class="elsevierStyleCrossRefs" href="#bib0230"><span class="elsevierStyleSup">22,23</span></a> or metabolites<a class="elsevierStyleCrossRef" href="#bib0240"><span class="elsevierStyleSup">24</span></a> are emerging. Although still none of the markers achieved AUCs to be considered as alternative of biopsy, these biomarkers could be used for pre-screening strategies in randomized clinical trials recruitment.</p></span><span id="sec0025" class="elsevierStyleSection elsevierViewall"><span class="elsevierStyleSectionTitle" id="sect0025">Concluding remarks</span><p id="par0040" class="elsevierStylePara elsevierViewall">In MASLD, early disease detection has arisen as a cornerstone in personalized medicine. Understanding disease prevalence, addressing NAFLD heterogeneity and contributing factors is essential for effective resource allocation in healthcare. The heterogeneity in MASLD sub-phenotypes has presumably hampered the progress towards effective non-invasive tools. On one hand, efficient referral pathways are needed at detecting fibrosis from primary care or targeted groups, without resulting in futile referrals and preferably at low costs. The current strategies are quite efficient in detecting advanced stages of the disease, but offer modest sensitivity in patients with intermediate risk (<a class="elsevierStyleCrossRef" href="#fig0005">Fig. 1</a>). Further, current recruitment for clinical trials is based on liver biopsy. Screening failure rates could be reduced in trials if only those patients positive for the proposed biomarkers underwent a biopsy for eligibility. Thus, a pre-selection would be performed to substantially reduce the number of liver biopsies. Although great progress has been recently made, some gaps need to be filled for qualification, so further studies will be required to validate their diagnostic accuracy and utility.</p><elsevierMultimedia ident="fig0005"></elsevierMultimedia></span></span>" "textoCompletoSecciones" => array:1 [ "secciones" => array:7 [ 0 => array:2 [ "identificador" => "sec0005" "titulo" => "Introduction" ] 1 => array:2 [ "identificador" => "sec0010" "titulo" => "MASLD, a multi-layered disease" ] 2 => array:2 [ "identificador" => "sec0015" "titulo" => "MASLD, the search for non-invasive approaches" ] 3 => array:2 [ "identificador" => "sec0020" "titulo" => "Comparative study of the new horizons" ] 4 => array:2 [ "identificador" => "sec0025" "titulo" => "Concluding remarks" ] 5 => array:2 [ "identificador" => "xack750365" "titulo" => "Acknowledgements" ] 6 => array:1 [ "titulo" => "References" ] ] ] "pdfFichero" => "main.pdf" "tienePdf" => true "multimedia" => array:1 [ 0 => array:7 [ "identificador" => "fig0005" "etiqueta" => "Figure 1" "tipo" => "MULTIMEDIAFIGURA" "mostrarFloat" => true "mostrarDisplay" => false "figura" => array:1 [ 0 => array:4 [ "imagen" => "gr1.jpeg" "Alto" => 2439 "Ancho" => 2925 "Tamanyo" => 500218 ] ] "descripcion" => array:1 [ "en" => "<p id="spar0005" class="elsevierStyleSimplePara elsevierViewall">Facing a new era in MASLD biomarkers. Progressive forms of liver disease require the use of targeted therapies adapted to stratified groups of patients, considering the main drivers of the disease to define patients’ subphenotypes. Biomarkers have proven diagnostic accuracy in targeted populations (higher prevalence), but require further validation in primary care settings (lower prevalence) for screening purposes.</p>" ] ] ] "bibliografia" => array:2 [ "titulo" => "References" "seccion" => array:1 [ 0 => array:2 [ "identificador" => "bibs0015" "bibliografiaReferencia" => array:24 [ 0 => array:3 [ "identificador" => "bib0125" "etiqueta" => "1" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "A multisociety Delphi consensus statement on new fatty liver disease nomenclature" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "M.E. Rinella" 1 => "J.V. Lazarus" 2 => "V. Ratziu" 3 => "S.M. Francque" 4 => "A.J. Sanyal" 5 => "F. Kanwal" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1097/HEP.0000000000000520" "Revista" => array:6 [ "tituloSerie" => "Hepatology" "fecha" => "2023" "volumen" => "78" "paginaInicial" => "1966" "paginaFinal" => "1986" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/37363821" "web" => "Medline" ] ] ] ] ] ] ] ] 1 => array:3 [ "identificador" => "bib0130" "etiqueta" => "2" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Global epidemiology of nonalcoholic fatty liver disease – meta-analytic assessment of prevalence, incidence, and outcomes" "autores" => array:1 [ 0 => array:2 [ "etal" => false "autores" => array:6 [ 0 => "Z.M. Younossi" 1 => "A.B. Koenig" 2 => "D. Abdelatif" 3 => "Y. Fazel" 4 => "L. Henry" 5 => "M. Wymer" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1002/hep.28431" "Revista" => array:7 [ "tituloSerie" => "Hepatology" "fecha" => "2016" "volumen" => "64" "paginaInicial" => "73" "paginaFinal" => "84" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/26707365" "web" => "Medline" ] ] "itemHostRev" => array:3 [ "pii" => "S0883944120303907" "estado" => "S300" "issn" => "08839441" ] ] ] ] ] ] ] 2 => array:3 [ "identificador" => "bib0135" "etiqueta" => "3" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Significant fibrosis predicts new-onset diabetes mellitus and arterial hypertension in patients with NASH" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "J. Ampuero" 1 => "R. Aller" 2 => "R. Gallego-Durán" 3 => "J. Crespo" 4 => "J.L. Calleja" 5 => "C. García-Monzón" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/j.jhep.2020.02.028" "Revista" => array:6 [ "tituloSerie" => "J Hepatol" "fecha" => "2020" "volumen" => "73" "paginaInicial" => "17" "paginaFinal" => "25" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/32147361" "web" => "Medline" ] ] ] ] ] ] ] ] 3 => array:3 [ "identificador" => "bib0140" "etiqueta" => "4" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Non-alcoholic fatty liver disease: a patient guideline" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "S.M. Francque" 1 => "G. Marchesini" 2 => "A. Kautz" 3 => "M. Walmsley" 4 => "R. Dorner" 5 => "J.V. Lazarus" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/j.jhepr.2021.100322" "Revista" => array:5 [ "tituloSerie" => "JHEP Rep" "fecha" => "2021" "volumen" => "3" "paginaInicial" => "100322" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/34693236" "web" => "Medline" ] ] ] ] ] ] ] ] 4 => array:3 [ "identificador" => "bib0145" "etiqueta" => "5" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "a systemic metabolic disorder with cardiovascular and malignant complications" "autores" => array:1 [ 0 => array:2 [ "etal" => false "autores" => array:4 [ 0 => "G. Targher" 1 => "C.D. Byrne" 2 => "H. Tilg" 3 => "MASLD:" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1136/gutjnl-2023-330595" "Revista" => array:2 [ "tituloSerie" => "Gut" "fecha" => "2024" ] ] ] ] ] ] 5 => array:3 [ "identificador" => "bib0150" "etiqueta" => "6" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Nonalcoholic steatohepatitis is the second leading etiology of liver disease among adults awaiting liver transplantation in the United States" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "R.J. Wong" 1 => "M. Aguilar" 2 => "R. Cheung" 3 => "R.B. Perumpail" 4 => "S.A. Harrison" 5 => "Z.M. Younossi" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1053/j.gastro.2014.11.039" "Revista" => array:6 [ "tituloSerie" => "Gastroenterology" "fecha" => "2015" "volumen" => "148" "paginaInicial" => "547" "paginaFinal" => "555" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/25461851" "web" => "Medline" ] ] ] ] ] ] ] ] 6 => array:3 [ "identificador" => "bib0155" "etiqueta" => "7" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Fibrosis stage but not NASH predicts mortality and time to development of severe liver disease in biopsy-proven NAFLD" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "H. Hagström" 1 => "P. Nasr" 2 => "M. Ekstedt" 3 => "U. Hammar" 4 => "P. Stål" 5 => "R. Hultcrantz" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/j.jhep.2017.07.027" "Revista" => array:6 [ "tituloSerie" => "J Hepatol" "fecha" => "2017" "volumen" => "67" "paginaInicial" => "1265" "paginaFinal" => "1273" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/28803953" "web" => "Medline" ] ] ] ] ] ] ] ] 7 => array:3 [ "identificador" => "bib0160" "etiqueta" => "8" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Weight loss through lifestyle modification significantly reduces features of nonalcoholic steatohepatitis" "autores" => array:1 [ 0 => array:2 [ "etal" => false "autores" => array:6 [ 0 => "E. Vilar-Gomez" 1 => "Y. Martinez-Perez" 2 => "L. Calzadilla-Bertot" 3 => "A. Torres-Gonzalez" 4 => "B. Gra-Oramas" 5 => "al. Gonzalez-et" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1053/j.gastro.2015.04.005" "Revista" => array:6 [ "tituloSerie" => "Gastroenterology" "fecha" => "2015" "volumen" => "149" "paginaInicial" => "367" "paginaFinal" => "378" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/25865049" "web" => "Medline" ] ] ] ] ] ] ] ] 8 => array:3 [ "identificador" => "bib0165" "etiqueta" => "9" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Insights into nonalcoholic fatty-liver disease heterogeneity" "autores" => array:1 [ 0 => array:2 [ "etal" => false "autores" => array:6 [ 0 => "M. Arrese" 1 => "J.P. Arab" 2 => "F. Barrera" 3 => "B. Kaufmann" 4 => "L. Valenti" 5 => "A.E. Feldstein" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1055/s-0041-1730927" "Revista" => array:6 [ "tituloSerie" => "Semin Liver Dis" "fecha" => "2021" "volumen" => "41" "paginaInicial" => "421" "paginaFinal" => "434" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/34233370" "web" => "Medline" ] ] ] ] ] ] ] ] 9 => array:3 [ "identificador" => "bib0170" "etiqueta" => "10" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "International consensus panel MAFLD: a consensus-driven proposed nomenclature for metabolic associated fatty liver disease" "autores" => array:1 [ 0 => array:2 [ "etal" => false "autores" => array:3 [ 0 => "M. Eslam" 1 => "A.J. Sanyal" 2 => "J. George" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1053/j.gastro.2019.11.312" "Revista" => array:6 [ "tituloSerie" => "Gastroenterology" "fecha" => "2020" "volumen" => "158" "paginaInicial" => "1999" "paginaFinal" => "2014" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/32044314" "web" => "Medline" ] ] ] ] ] ] ] ] 10 => array:3 [ "identificador" => "bib0175" "etiqueta" => "11" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Hepatocellular and extrahepatic cancer risk in people with non-alcoholic fatty liver disease" "autores" => array:1 [ 0 => array:2 [ "etal" => false "autores" => array:5 [ 0 => "J.A. Thomas" 1 => "B.J. Kendall" 2 => "H.B. El-Serag" 3 => "A.P. Thrift" 4 => "G.A. Macdonald" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/S2468-1253(23)00275-3" "Revista" => array:6 [ "tituloSerie" => "Lancet Gastroenterol Hepatol" "fecha" => "2024" "volumen" => "9" "paginaInicial" => "159" "paginaFinal" => "169" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/38215780" "web" => "Medline" ] ] ] ] ] ] ] ] 11 => array:3 [ "identificador" => "bib0180" "etiqueta" => "12" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Association between fibrosis stage and outcomes of patients with nonalcoholic fatty liver disease: a systematic review and meta-analysis" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "R.S. Taylor" 1 => "R.J. Taylor" 2 => "S. Bayliss" 3 => "H. Hagström" 4 => "P. Nasr" 5 => "J.M. Schattenberg" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1053/j.gastro.2020.01.043" "Revista" => array:6 [ "tituloSerie" => "Gastroenterology" "fecha" => "2020" "volumen" => "158" "paginaInicial" => "1611" "paginaFinal" => "1625" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/32027911" "web" => "Medline" ] ] ] ] ] ] ] ] 12 => array:3 [ "identificador" => "bib0185" "etiqueta" => "13" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Prospective study of outcomes in adults with nonalcoholic fatty liver disease" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "A.J. Sanyal" 1 => "M.L. Van Natta" 2 => "J. Clark" 3 => "B.A. Neuschwander-Tetri" 4 => "A. Diehl" 5 => "S. Dasarathy" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1056/NEJMoa2029349" "Revista" => array:6 [ "tituloSerie" => "N Engl J Med" "fecha" => "2021" "volumen" => "385" "paginaInicial" => "1559" "paginaFinal" => "1569" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/34670043" "web" => "Medline" ] ] ] ] ] ] ] ] 13 => array:3 [ "identificador" => "bib0190" "etiqueta" => "14" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Biomarkers for staging fibrosis and non-alcoholic steatohepatitis in non-alcoholic fatty liver disease (the LITMUS project): a comparative diagnostic accuracy study" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "Y. Vali" 1 => "J. Lee" 2 => "J. Boursier" 3 => "S. Petta" 4 => "K. Wonders" 5 => "D. Tiniakos" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/S2468-1253(23)00017-1" "Revista" => array:6 [ "tituloSerie" => "Lancet Gastroenterol Hepatol" "fecha" => "2023" "volumen" => "8" "paginaInicial" => "714" "paginaFinal" => "725" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/36958367" "web" => "Medline" ] ] ] ] ] ] ] ] 14 => array:3 [ "identificador" => "bib0195" "etiqueta" => "15" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Diagnostic performance of circulating biomarkers for non-alcoholic steatohepatitis" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "A.J. Sanyal" 1 => "S.S. Shankar" 2 => "K.P. Yates" 3 => "J. Bolognese" 4 => "E. Daly" 5 => "C.A. Dehn" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1038/s41591-023-02539-6" "Revista" => array:6 [ "tituloSerie" => "Nat Med" "fecha" => "2023" "volumen" => "29" "paginaInicial" => "2656" "paginaFinal" => "2664" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/37679433" "web" => "Medline" ] ] ] ] ] ] ] ] 15 => array:3 [ "identificador" => "bib0200" "etiqueta" => "16" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "A blood-based biomarker panel (NIS4) for non-invasive diagnosis of non-alcoholic steatohepatitis and liver fibrosis: a prospective derivation and global validation study" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "S.A. Harrison" 1 => "V. Ratziu" 2 => "J. Boursier" 3 => "S. Francque" 4 => "P. Bedossa" 5 => "Z. Majd" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/S2468-1253(20)30252-1" "Revista" => array:6 [ "tituloSerie" => "Lancet Gastroenterol Hepatol" "fecha" => "2020" "volumen" => "5" "paginaInicial" => "970" "paginaFinal" => "985" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/32763196" "web" => "Medline" ] ] ] ] ] ] ] ] 16 => array:3 [ "identificador" => "bib0205" "etiqueta" => "17" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "NIS2+™, an optimisation of the blood-based biomarker NIS4® technology for the detection of at-risk NASH: a prospective derivation and validation study" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "S.A. Harrison" 1 => "V. Ratziu" 2 => "J. Magnanensi" 3 => "Y. Hajji" 4 => "S. Deledicque" 5 => "Z. Majd" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/j.jhep.2023.04.031" "Revista" => array:6 [ "tituloSerie" => "J Hepatol" "fecha" => "2023" "volumen" => "79" "paginaInicial" => "758" "paginaFinal" => "767" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/37224923" "web" => "Medline" ] ] ] ] ] ] ] ] 17 => array:3 [ "identificador" => "bib0210" "etiqueta" => "18" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Defining the serum proteomic signature of hepatic steatosis, inflammation, ballooning and fibrosis in non-alcoholic fatty liver disease" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "A.J. Sanyal" 1 => "S.A. Williams" 2 => "J.E. Lavine" 3 => "B.A. Neuschwander-Tetri" 4 => "L. Alexander" 5 => "R. Ostroff" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/j.jhep.2022.11.029" "Revista" => array:6 [ "tituloSerie" => "J Hepatol" "fecha" => "2023" "volumen" => "78" "paginaInicial" => "693" "paginaFinal" => "703" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/36528237" "web" => "Medline" ] ] ] ] ] ] ] ] 18 => array:3 [ "identificador" => "bib0215" "etiqueta" => "19" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "SOMAscan proteomics identifies serum biomarkers associated with liver fibrosis in patients with NASH" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "Y. Luo" 1 => "S. Wadhawan" 2 => "A. Greenfield" 3 => "B.E. Decato" 4 => "A.M. Oseini" 5 => "R. Collen" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1002/hep4.1670" "Revista" => array:6 [ "tituloSerie" => "Hepatol Commun" "fecha" => "2021" "volumen" => "5" "paginaInicial" => "760" "paginaFinal" => "773" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/34027267" "web" => "Medline" ] ] ] ] ] ] ] ] 19 => array:3 [ "identificador" => "bib0220" "etiqueta" => "20" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "FibroScan-AST (FAST) score for the non-invasive identification of patients with non-alcoholic steatohepatitis with significant activity and fibrosis: a prospective derivation and global validation study" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "P.N. Newsome" 1 => "M. Sasso" 2 => "J.J. Deeks" 3 => "A. Paredes" 4 => "J. Boursier" 5 => "W.K. Chan" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/S2468-1253(19)30383-8" "Revista" => array:6 [ "tituloSerie" => "Lancet Gastroenterol Hepatol" "fecha" => "2020" "volumen" => "5" "paginaInicial" => "362" "paginaFinal" => "373" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/32027858" "web" => "Medline" ] ] ] ] ] ] ] ] 20 => array:3 [ "identificador" => "bib0225" "etiqueta" => "21" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "MRI-based (MAST) score accurately identifies patients with NASH and significant fibrosis" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "M. Noureddin" 1 => "E. Truong" 2 => "J.A. Gornbein" 3 => "R. Saouaf" 4 => "M. Guindi" 5 => "T. Todo" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/j.jhep.2021.11.012" "Revista" => array:6 [ "tituloSerie" => "J Hepatol" "fecha" => "2022" "volumen" => "76" "paginaInicial" => "781" "paginaFinal" => "787" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/34798176" "web" => "Medline" ] ] ] ] ] ] ] ] 21 => array:3 [ "identificador" => "bib0230" "etiqueta" => "22" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "ADAMTSL2 protein and a soluble biomarker signature identify at-risk non-alcoholic steatohepatitis and fibrosis in adults with NAFLD" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "K.E. Corey" 1 => "R. Pitts" 2 => "M. Lai" 3 => "J. Loureiro" 4 => "R. Masia" 5 => "S.A. Osganian" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/j.jhep.2021.09.026" "Revista" => array:5 [ "tituloSerie" => "J Hepatol" "fecha" => "2022" "volumen" => "76" "paginaInicial" => "25" "paginaFinal" => "33" ] ] ] ] ] ] 22 => array:3 [ "identificador" => "bib0235" "etiqueta" => "23" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "A proteo-transcriptomic map of non-alcoholic fatty liver disease signatures" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "O. Govaere" 1 => "M. Hasoon" 2 => "L. Alexander" 3 => "S. Cockell" 4 => "D. Tiniakos" 5 => "M. Ekstedt" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1038/s42255-023-00775-1" "Revista" => array:6 [ "tituloSerie" => "Nat Metab" "fecha" => "2023" "volumen" => "5" "paginaInicial" => "572" "paginaFinal" => "578" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/37037945" "web" => "Medline" ] ] ] ] ] ] ] ] 23 => array:3 [ "identificador" => "bib0240" "etiqueta" => "24" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Serum identification of at-risk MASH: the metabolomics-advanced steatohepatitis fibrosis score (MASEF)" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "M. Noureddin" 1 => "E. Truong" 2 => "R. Mayo" 3 => "I. Martínez-Arranz" 4 => "I. Mincholé" 5 => "J.M. Banales" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1097/HEP.0000000000000542" "Revista" => array:6 [ "tituloSerie" => "Hepatology" "fecha" => "2024" "volumen" => "79" "paginaInicial" => "135" "paginaFinal" => "148" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/37505221" "web" => "Medline" ] ] ] ] ] ] ] ] ] ] ] ] "agradecimientos" => array:1 [ 0 => array:4 [ "identificador" => "xack750365" "titulo" => "Acknowledgements" "texto" => "<p id="par0045" class="elsevierStylePara elsevierViewall">The research leading to these results has received funding from the <span class="elsevierStyleGrantSponsor" id="gs1">Consejería de Salud de la Junta de Andalucía</span> under grant agreement <span class="elsevierStyleGrantNumber" refid="gs1">PC-0148-2016-0148</span> and <span class="elsevierStyleGrantNumber" refid="gs1">PE-0451-2018</span>, and from <span class="elsevierStyleGrantSponsor" id="gs2">Instituto de Salud Carlos III</span> under grant agreements <span class="elsevierStyleGrantNumber" refid="gs2">CD21/00095</span>, <span class="elsevierStyleGrantNumber" refid="gs2">PI19/01404</span>, <span class="elsevierStyleGrantNumber" refid="gs2">PI19/00589</span>, <span class="elsevierStyleGrantNumber" refid="gs2">PI22/01342</span>, <span class="elsevierStyleGrantNumber" refid="gs2">PI22/01345</span>.</p>" "vista" => "all" ] ] ] "idiomaDefecto" => "en" "url" => "/02105705/0000004700000004/v2_202406091228/S0210570524000530/v2_202406091228/en/main.assets" "Apartado" => array:4 [ "identificador" => "96400" "tipo" => "SECCION" "es" => array:2 [ "titulo" => "Rincón Joven" "idiomaDefecto" => true ] "idiomaDefecto" => "es" ] "PDF" => "https://static.elsevier.es/multimedia/02105705/0000004700000004/v2_202406091228/S0210570524000530/v2_202406091228/en/main.pdf?idApp=UINPBA00004N&text.app=https://www.elsevier.es/" "EPUB" => "https://multimedia.elsevier.es/PublicationsMultimediaV1/item/epub/S0210570524000530?idApp=UINPBA00004N" ]
Journal Information
Vol. 47. Issue 4.
Pages 393-396 (April 2024)
Vol. 47. Issue 4.
Pages 393-396 (April 2024)
Young corner
MASLD biomarkers: Are we facing a new era?
Biomarcadores en MASLD, ¿estamos ante una nueva era?
Visits
522
Douglas Maya-Milesa,b, Javier Ampueroa,b,c, David Martí-Aguadod, Andrés Contheb,e, Rocío Gallego-Durána,b,
Corresponding author
a SeLiver Group, Instituto de Biomedicina de Sevilla/CSIC/Universidad de Sevilla, Sevilla, Spain
b Hepatic and Digestive Diseases Networking Biomedical Research Centre (CIBERehd), Madrid, Spain
c Digestive Diseases Unit, Hospital Universitario Virgen del Rocío, Sevilla, Spain
d Servicio de Aparato Digestivo, Hospital Clínico Universitario de Valencia, INCLIVA Instituto de Investigación Sanitaria, Valencia, Spain
e Sección de Hepatología, Servicio de Aparato Digestivo, Hospital General Universitario Gregorio Marañón, Madrid, Spain
Article information
These are the options to access the full texts of the publication Gastroenterología y Hepatología
Subscriber
Subscribe
Purchase
Contact
Phone for subscriptions and reporting of errors
From Monday to Friday from 9 a.m. to 6 p.m. (GMT + 1) except for the months of July and August which will be from 9 a.m. to 3 p.m.
Calls from Spain
932 415 960
Calls from outside Spain
+34 932 415 960
E-mail