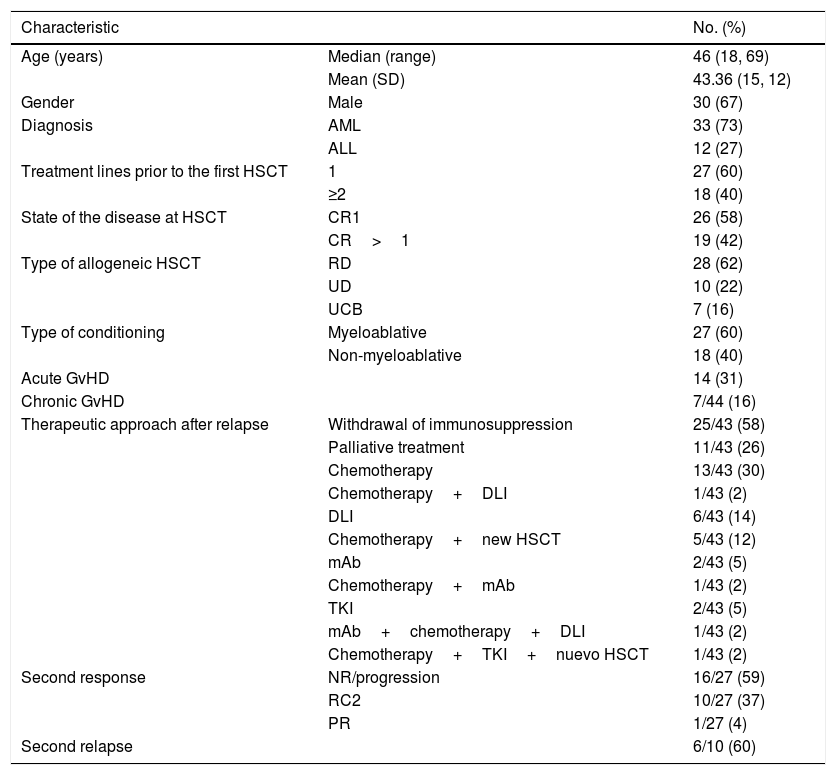
array:24 [ "pii" => "S2387020618303073" "issn" => "23870206" "doi" => "10.1016/j.medcle.2017.11.054" "estado" => "S300" "fechaPublicacion" => "2018-11-09" "aid" => "4379" "copyright" => "Elsevier España, S.L.U.. All rights reserved" "copyrightAnyo" => "2017" "documento" => "simple-article" "crossmark" => 0 "subdocumento" => "crp" "cita" => "Med Clin. 2018;151:376-7" "abierto" => array:3 [ "ES" => false "ES2" => false "LATM" => false ] "gratuito" => false "lecturas" => array:1 [ "total" => 0 ] "Traduccion" => array:1 [ "es" => array:19 [ "pii" => "S0025775317309284" "issn" => "00257753" "doi" => "10.1016/j.medcli.2017.11.044" "estado" => "S300" "fechaPublicacion" => "2018-11-09" "aid" => "4379" "copyright" => "Elsevier España, S.L.U." "documento" => "simple-article" "crossmark" => 1 "subdocumento" => "crp" "cita" => "Med Clin. 2018;151:376-7" "abierto" => array:3 [ "ES" => false "ES2" => false "LATM" => false ] "gratuito" => false "lecturas" => array:2 [ "total" => 10 "formatos" => array:2 [ "HTML" => 3 "PDF" => 7 ] ] "es" => array:10 [ "idiomaDefecto" => true "cabecera" => "<span class="elsevierStyleTextfn">Carta científica</span>" "titulo" => "Tratamiento de rescate de pacientes con leucemia aguda en recaída después de un primer trasplante alogénico de progenitores hematopoyéticos" "tienePdf" => "es" "tieneTextoCompleto" => "es" "paginas" => array:1 [ 0 => array:2 [ "paginaInicial" => "376" "paginaFinal" => "377" ] ] "titulosAlternativos" => array:1 [ "en" => array:1 [ "titulo" => "Rescue treatment of patients with relapsed acute leukemia after first allogeneic hematopoietic stem cell transplantation" ] ] "contieneTextoCompleto" => array:1 [ "es" => true ] "contienePdf" => array:1 [ "es" => true ] "autores" => array:1 [ 0 => array:2 [ "autoresLista" => "Anna Torrent, Christelle Ferrá, Josep-Maria Ribera" "autores" => array:3 [ 0 => array:2 [ "nombre" => "Anna" "apellidos" => "Torrent" ] 1 => array:2 [ "nombre" => "Christelle" "apellidos" => "Ferrá" ] 2 => array:2 [ "nombre" => "Josep-Maria" "apellidos" => "Ribera" ] ] ] ] ] "idiomaDefecto" => "es" "Traduccion" => array:1 [ "en" => array:9 [ "pii" => "S2387020618303073" "doi" => "10.1016/j.medcle.2017.11.054" "estado" => "S300" "subdocumento" => "" "abierto" => array:3 [ "ES" => false "ES2" => false "LATM" => false ] "gratuito" => false "lecturas" => array:1 [ "total" => 0 ] "idiomaDefecto" => "en" "EPUB" => "https://multimedia.elsevier.es/PublicationsMultimediaV1/item/epub/S2387020618303073?idApp=UINPBA00004N" ] ] "EPUB" => "https://multimedia.elsevier.es/PublicationsMultimediaV1/item/epub/S0025775317309284?idApp=UINPBA00004N" "url" => "/00257753/0000015100000009/v1_201810220613/S0025775317309284/v1_201810220613/es/main.assets" ] ] "itemSiguiente" => array:18 [ "pii" => "S238702061830398X" "issn" => "23870206" "doi" => "10.1016/j.medcle.2017.11.062" "estado" => "S300" "fechaPublicacion" => "2018-11-09" "aid" => "4378" "copyright" => "Elsevier España, S.L.U." "documento" => "simple-article" "crossmark" => 1 "subdocumento" => "cor" "cita" => "Med Clin. 2018;151:378-9" "abierto" => array:3 [ "ES" => false "ES2" => false "LATM" => false ] "gratuito" => false "lecturas" => array:1 [ "total" => 0 ] "en" => array:11 [ "idiomaDefecto" => true "cabecera" => "<span class="elsevierStyleTextfn">Letter to the Editor</span>" "titulo" => "Polyneuropathy by statins: case report" "tienePdf" => "en" "tieneTextoCompleto" => "en" "paginas" => array:1 [ 0 => array:2 [ "paginaInicial" => "378" "paginaFinal" => "379" ] ] "titulosAlternativos" => array:1 [ "es" => array:1 [ "titulo" => "Polineuropatía por estatinas: descripción" ] ] "contieneTextoCompleto" => array:1 [ "en" => true ] "contienePdf" => array:1 [ "en" => true ] "resumenGrafico" => array:2 [ "original" => 0 "multimedia" => array:7 [ "identificador" => "fig0005" "etiqueta" => "Fig. 1" "tipo" => "MULTIMEDIAFIGURA" "mostrarFloat" => true "mostrarDisplay" => false "figura" => array:1 [ 0 => array:4 [ "imagen" => "gr1.jpeg" "Alto" => 1524 "Ancho" => 1250 "Tamanyo" => 103532 ] ] "descripcion" => array:1 [ "en" => "<p id="spar0005" class="elsevierStyleSimplePara elsevierViewall">Electromyography of the lower limbs: axonal peripheral polyneuropathy with marked sensorial predominance.</p>" ] ] ] "autores" => array:1 [ 0 => array:2 [ "autoresLista" => "Yenny Shirley Ruiz Anzola, Liliana Simões, Sérgio Borges" "autores" => array:3 [ 0 => array:2 [ "nombre" => "Yenny Shirley" "apellidos" => "Ruiz Anzola" ] 1 => array:2 [ "nombre" => "Liliana" "apellidos" => "Simões" ] 2 => array:2 [ "nombre" => "Sérgio" "apellidos" => "Borges" ] ] ] ] ] "idiomaDefecto" => "en" "EPUB" => "https://multimedia.elsevier.es/PublicationsMultimediaV1/item/epub/S238702061830398X?idApp=UINPBA00004N" "url" => "/23870206/0000015100000009/v1_201811220618/S238702061830398X/v1_201811220618/en/main.assets" ] "itemAnterior" => array:19 [ "pii" => "S2387020618303036" "issn" => "23870206" "doi" => "10.1016/j.medcle.2018.07.002" "estado" => "S300" "fechaPublicacion" => "2018-11-09" "aid" => "4334" "copyright" => "Elsevier España, S.L.U." "documento" => "simple-article" "crossmark" => 1 "subdocumento" => "crp" "cita" => "Med Clin. 2018;151:374-5" "abierto" => array:3 [ "ES" => false "ES2" => false "LATM" => false ] "gratuito" => false "lecturas" => array:1 [ "total" => 0 ] "en" => array:11 [ "idiomaDefecto" => true "cabecera" => "<span class="elsevierStyleTextfn">Scientific letter</span>" "titulo" => "Feohifomicosis in a renal and pulmonary transplant patient" "tienePdf" => "en" "tieneTextoCompleto" => "en" "paginas" => array:1 [ 0 => array:2 [ "paginaInicial" => "374" "paginaFinal" => "375" ] ] "titulosAlternativos" => array:1 [ "es" => array:1 [ "titulo" => "Feohifomicosis en paciente receptor de trasplante renal y pulmonar" ] ] "contieneTextoCompleto" => array:1 [ "en" => true ] "contienePdf" => array:1 [ "en" => true ] "resumenGrafico" => array:2 [ "original" => 0 "multimedia" => array:7 [ "identificador" => "fig0005" "etiqueta" => "Fig. 1" "tipo" => "MULTIMEDIAFIGURA" "mostrarFloat" => true "mostrarDisplay" => false "figura" => array:1 [ 0 => array:4 [ "imagen" => "gr1.jpeg" "Alto" => 1875 "Ancho" => 2500 "Tamanyo" => 733253 ] ] "descripcion" => array:1 [ "en" => "<p id="spar0005" class="elsevierStyleSimplePara elsevierViewall">(A) Signs and symptoms: indurated infiltrated erythematous nodules in the arm; (B) Pathological anatomy, HE: dense granulomatous infiltrates accompanied by neutrophils; (C and D) Methenamine silver staining: linear chains formed by round structures corresponding to the fungus.</p>" ] ] ] "autores" => array:1 [ 0 => array:2 [ "autoresLista" => "Irene Salgüero Fernández, Dolores Suarez Massa, Gastón Roustan Gullón" "autores" => array:3 [ 0 => array:2 [ "nombre" => "Irene" "apellidos" => "Salgüero Fernández" ] 1 => array:2 [ "nombre" => "Dolores" "apellidos" => "Suarez Massa" ] 2 => array:2 [ "nombre" => "Gastón" "apellidos" => "Roustan Gullón" ] ] ] ] ] "idiomaDefecto" => "en" "Traduccion" => array:1 [ "es" => array:9 [ "pii" => "S0025775317308801" "doi" => "10.1016/j.medcli.2017.11.005" "estado" => "S300" "subdocumento" => "" "abierto" => array:3 [ "ES" => false "ES2" => false "LATM" => false ] "gratuito" => false "lecturas" => array:1 [ "total" => 0 ] "idiomaDefecto" => "es" "EPUB" => "https://multimedia.elsevier.es/PublicationsMultimediaV1/item/epub/S0025775317308801?idApp=UINPBA00004N" ] ] "EPUB" => "https://multimedia.elsevier.es/PublicationsMultimediaV1/item/epub/S2387020618303036?idApp=UINPBA00004N" "url" => "/23870206/0000015100000009/v1_201811220618/S2387020618303036/v1_201811220618/en/main.assets" ] "en" => array:14 [ "idiomaDefecto" => true "cabecera" => "<span class="elsevierStyleTextfn">Scientific letter</span>" "titulo" => "Rescue treatment of patients with relapsed acute leukemia after first allogeneic hematopoietic stem cell transplantation" "tieneTextoCompleto" => true "saludo" => "Dear Editor," "paginas" => array:1 [ 0 => array:2 [ "paginaInicial" => "376" "paginaFinal" => "377" ] ] "autores" => array:1 [ 0 => array:4 [ "autoresLista" => "Anna Torrent, Christelle Ferrá, Josep-Maria Ribera" "autores" => array:3 [ 0 => array:4 [ "nombre" => "Anna" "apellidos" => "Torrent" "email" => array:1 [ 0 => "atorrent@iconcologia.net" ] "referencia" => array:4 [ 0 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">a</span>" "identificador" => "aff0005" ] 1 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">b</span>" "identificador" => "aff0010" ] 2 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">c</span>" "identificador" => "aff0015" ] 3 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">*</span>" "identificador" => "cor0005" ] ] ] 1 => array:3 [ "nombre" => "Christelle" "apellidos" => "Ferrá" "referencia" => array:3 [ 0 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">a</span>" "identificador" => "aff0005" ] 1 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">b</span>" "identificador" => "aff0010" ] 2 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">c</span>" "identificador" => "aff0015" ] ] ] 2 => array:3 [ "nombre" => "Josep-Maria" "apellidos" => "Ribera" "referencia" => array:3 [ 0 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">a</span>" "identificador" => "aff0005" ] 1 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">b</span>" "identificador" => "aff0010" ] 2 => array:2 [ "etiqueta" => "<span class="elsevierStyleSup">c</span>" "identificador" => "aff0015" ] ] ] ] "afiliaciones" => array:3 [ 0 => array:3 [ "entidad" => "Servicio de Hematología Clínica, Institut Català d’Oncologia-Hospital Germans Trias i Pujol, Badalona, Barcelona, Spain" "etiqueta" => "a" "identificador" => "aff0005" ] 1 => array:3 [ "entidad" => "Institut de Recerca contra la Leucèmia Josep Carreras, Badalona, Barcelona, Spain" "etiqueta" => "b" "identificador" => "aff0010" ] 2 => array:3 [ "entidad" => "Universitat Autònoma de Barcelona, Barcelona, Spain" "etiqueta" => "c" "identificador" => "aff0015" ] ] "correspondencia" => array:1 [ 0 => array:3 [ "identificador" => "cor0005" "etiqueta" => "⁎" "correspondencia" => "Corresponding author." ] ] ] ] "titulosAlternativos" => array:1 [ "es" => array:1 [ "titulo" => "Tratamiento de rescate de pacientes con leucemia aguda en recaída después de un primer trasplante alogénico de progenitores hematopoyéticos" ] ] "textoCompleto" => "<span class="elsevierStyleSections"><p id="par0005" class="elsevierStylePara elsevierViewall">An allogeneic hematopoietic stem cell transplant (HSCT) is the most effective post-consolidation treatment and the best curative option for patients with acute leukemia (AL). The main cause of HSCT failure, and in many cases of the death of the patient, is the relapse of the disease. However, after a post-HSCT relapse, the probability of survival is very low. The rescue treatment in this situation is not well established and varies depending on the type of relapse and the characteristics of each patient and each AL, with generally very disappointing results.</p><p id="par0010" class="elsevierStylePara elsevierViewall">We retrospectively analyzed the overall survival (OS), the type of rescue treatment and the response of relapsed AL (lymphoblastic and myeloblastic) after an allogeneic HSCT in a single center. The main endpoint was to analyze the OS of this population.</p><p id="par0015" class="elsevierStylePara elsevierViewall">From March 2001 to December 2016, 182 allogeneic HSCTs were performed in patients with AL in complete remission (CR), of whom 45 relapsed (25%). <a class="elsevierStyleCrossRef" href="#tbl0005">Table 1</a> shows the characteristics of the patients and the different rescue treatments used. The median time between HSCT and relapse was 5.93 months (range: 0.97–101.17). At the time of analysis, 5 patients were alive, of whom 4 were disease-free and one was lost to follow-up. The probability of OS at 6 months after relapse was 28% (95% CI: 15–41%) with a median survival of 2.73 months (95% CI 2.09–3.37). This probability was lower when relapse occurred during the first year of HSCT, especially before 6 months, with a median of 1.93 months (95% CI: 0.78–3.08) in relapses before 6 months, and of 4.20 months (95% CI: 3.01–5.39) after 6 months (<span class="elsevierStyleItalic">p</span><span class="elsevierStyleHsp" style=""></span>=<span class="elsevierStyleHsp" style=""></span>0.031), and of 2.67 months (95% CI: 1.74–3.59) and 6.93 months (95% CI: 0–18.99) before and after 12 months, respectively (<span class="elsevierStyleItalic">p</span><span class="elsevierStyleHsp" style=""></span>=<span class="elsevierStyleHsp" style=""></span>0.012). In our series, we also observed a longer survival trend for acute lymphoblastic leukemia (ALL) compared to acute myeloblastic leukemia (AML), with a median survival of 2.53 months (95% CI: 1.51–3.65) for the AMLs and of 10.60 months (95% CI: 3.48–17.72 months) for the ALLs (<span class="elsevierStyleItalic">p</span><span class="elsevierStyleHsp" style=""></span>=<span class="elsevierStyleHsp" style=""></span>0.077).</p><elsevierMultimedia ident="tbl0005"></elsevierMultimedia><p id="par0020" class="elsevierStylePara elsevierViewall">In this study, a quarter of patients with AL relapsed after an allogeneic HSCT. The rescue treatments have been very diverse and of little efficacy. Only a small proportion of patients (6/45, 13%) received a second allogeneic HSCT. The probability of survival of patients with AL in post-allogeneic HSCT relapse in our series was very low, especially if the relapse was an early one.</p><p id="par0025" class="elsevierStylePara elsevierViewall">Although HSCT is a curative option for patients with AL, when relapse occurs after it, the prognosis worsens substantially, with a 5-year overall survival probability of 14%.<a class="elsevierStyleCrossRefs" href="#bib0030"><span class="elsevierStyleSup">1,2</span></a> and an accumulated incidence of relapse higher than 45–60% (Yan et al., 2017).<a class="elsevierStyleCrossRef" href="#bib0040"><span class="elsevierStyleSup">3</span></a> Several studies have shown that if relapse occurs during the first 6 months after the first HSCT, survival worsens.<a class="elsevierStyleCrossRefs" href="#bib0030"><span class="elsevierStyleSup">1,4</span></a> A recent study of post-HSCT AML relapse found that a shorter duration of remission after HSCT and the presence of acute graft-versus-host disease (GvHD) of grade 3–4 were independent prognostic variables after relapse.<a class="elsevierStyleCrossRef" href="#bib0030"><span class="elsevierStyleSup">1</span></a></p><p id="par0030" class="elsevierStylePara elsevierViewall">The therapeutic options in these patients are scarce and ineffective, and are chosen depending on the patient's condition, type of relapse, availability of cell therapy (donor lymphocytes or hematopoietic stem cells), the presence and severity of the GvHD at the time of relapse, the degree of immunosuppression, response to immunosuppression discontinuation and the preferences of the institution and the patient.<a class="elsevierStyleCrossRefs" href="#bib0030"><span class="elsevierStyleSup">1,5</span></a> For all these reasons, the treatment of these patients is poorly standardized, as it occurred in the present study. Therefore, it is essential to emphasize relapse prevention. One possible option would be to make successive infusions of donor lymphocytes according to the post-HSCT minimum residual disease (MRD) value and the presence and degree of GvHD.<a class="elsevierStyleCrossRef" href="#bib0040"><span class="elsevierStyleSup">3</span></a> Another option would be to apply specific treatments to the subgroup of patients with positive MRD before HSCT. In this sense, immunotherapy is offering promising results. Likewise, post-HSCT maintenance treatment may be another useful preventive strategy in certain AL subtypes.</p></span>" "pdfFichero" => "main.pdf" "tienePdf" => true "NotaPie" => array:1 [ 0 => array:2 [ "etiqueta" => "☆" "nota" => "<p class="elsevierStyleNotepara" id="npar0005">Please cite this article as: Torrent A, Ferrá C, Ribera J-M. Tratamiento de rescate de pacientes con leucemia aguda en recaída después de un primer trasplante alogénico de progenitores hematopoyéticos. Med Clin (Barc). 2018;151:376–377.</p>" ] ] "multimedia" => array:1 [ 0 => array:8 [ "identificador" => "tbl0005" "etiqueta" => "Table 1" "tipo" => "MULTIMEDIATABLA" "mostrarFloat" => true "mostrarDisplay" => false "detalles" => array:1 [ 0 => array:3 [ "identificador" => "at1" "detalle" => "Table " "rol" => "short" ] ] "tabla" => array:2 [ "leyenda" => "<p id="spar0010" class="elsevierStyleSimplePara elsevierViewall">mAb: monoclonal antibody; SD: related donor; UD: unrelated donor; GvHD: graft disease against the recipient; DLI: donor lymphocyte infusion; TKI: tyrosine kinase inhibitor; ALL: acute lymphoblastic leukemia; AML: acute myeloblastic leukemia; NR: no response; CR1: first complete remission; CR2: second complete remission; PR: partial remission; UCB: umbilical cord blood; HSCT: allogeneic hematopoietic stem cell transplant.</p>" "tablatextoimagen" => array:1 [ 0 => array:2 [ "tabla" => array:1 [ 0 => """ <table border="0" frame="\n \t\t\t\t\tvoid\n \t\t\t\t" class=""><thead title="thead"><tr title="table-row"><th class="td" title="table-head " align="left" valign="top" scope="col" style="border-bottom: 2px solid black">Characteristic \t\t\t\t\t\t\n \t\t\t\t</th><th class="td" title="table-head " align="" valign="top" scope="col" style="border-bottom: 2px solid black"> \t\t\t\t\t\t\n \t\t\t\t</th><th class="td" title="table-head " align="left" valign="top" scope="col" style="border-bottom: 2px solid black">No. (%) \t\t\t\t\t\t\n \t\t\t\t</th></tr></thead><tbody title="tbody"><tr title="table-row"><td class="td" title="table-entry " rowspan="2" align="left" valign="top">Age (years)</td><td class="td" title="table-entry " align="left" valign="top">Median (range) \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">46 (18, 69) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">Mean (SD) \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">43.36 (15, 12) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">Gender \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">Male \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">30 (67) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">Diagnosis \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">AML \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">33 (73) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td" title="table-entry " align="" valign="top"> \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">ALL \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">12 (27) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td" title="table-entry " rowspan="2" align="left" valign="top">Treatment lines prior to the first HSCT</td><td class="td" title="table-entry " align="left" valign="top">1 \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">27 (60) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">≥2 \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">18 (40) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td" title="table-entry " rowspan="2" align="left" valign="top">State of the disease at HSCT</td><td class="td" title="table-entry " align="left" valign="top">CR1 \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">26 (58) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">CR<span class="elsevierStyleHsp" style=""></span>><span class="elsevierStyleHsp" style=""></span>1 \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">19 (42) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td" title="table-entry " rowspan="3" align="left" valign="top">Type of allogeneic HSCT</td><td class="td" title="table-entry " align="left" valign="top">RD \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">28 (62) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">UD \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">10 (22) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">UCB \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">7 (16) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td" title="table-entry " rowspan="2" align="left" valign="top">Type of conditioning</td><td class="td" title="table-entry " align="left" valign="top">Myeloablative \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">27 (60) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">Non-myeloablative \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">18 (40) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td" title="table-entry " colspan="2" align="left" valign="top">Acute GvHD</td><td class="td" title="table-entry " align="left" valign="top">14 (31) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td" title="table-entry " colspan="2" align="left" valign="top">Chronic GvHD</td><td class="td" title="table-entry " align="left" valign="top">7/44 (16) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td" title="table-entry " rowspan="11" align="left" valign="top">Therapeutic approach after relapse</td><td class="td" title="table-entry " align="left" valign="top">Withdrawal of immunosuppression \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">25/43 (58) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">Palliative treatment \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">11/43 (26) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">Chemotherapy \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">13/43 (30) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">Chemotherapy<span class="elsevierStyleHsp" style=""></span>+<span class="elsevierStyleHsp" style=""></span>DLI \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">1/43 (2) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">DLI \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">6/43 (14) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">Chemotherapy<span class="elsevierStyleHsp" style=""></span>+<span class="elsevierStyleHsp" style=""></span>new HSCT \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">5/43 (12) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">mAb \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">2/43 (5) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">Chemotherapy<span class="elsevierStyleHsp" style=""></span>+<span class="elsevierStyleHsp" style=""></span>mAb \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">1/43 (2) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">TKI \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">2/43 (5) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">mAb<span class="elsevierStyleHsp" style=""></span>+<span class="elsevierStyleHsp" style=""></span>chemotherapy<span class="elsevierStyleHsp" style=""></span>+<span class="elsevierStyleHsp" style=""></span>DLI \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">1/43 (2) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">Chemotherapy<span class="elsevierStyleHsp" style=""></span>+<span class="elsevierStyleHsp" style=""></span>TKI<span class="elsevierStyleHsp" style=""></span>+<span class="elsevierStyleHsp" style=""></span>nuevo HSCT \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">1/43 (2) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td" title="table-entry " rowspan="3" align="left" valign="top">Second response</td><td class="td" title="table-entry " align="left" valign="top">NR/progression \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">16/27 (59) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">RC2 \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">10/27 (37) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">PR \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">1/27 (4) \t\t\t\t\t\t\n \t\t\t\t</td></tr><tr title="table-row"><td class="td-with-role" title="table-entry ; entry_with_role_rowhead " align="left" valign="top">Second relapse \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="" valign="top"> \t\t\t\t\t\t\n \t\t\t\t</td><td class="td" title="table-entry " align="left" valign="top">6/10 (60) \t\t\t\t\t\t\n \t\t\t\t</td></tr></tbody></table> """ ] "imagenFichero" => array:1 [ 0 => "xTab1900783.png" ] ] ] ] "descripcion" => array:1 [ "en" => "<p id="spar0005" class="elsevierStyleSimplePara elsevierViewall">Characteristics of the 45 post-HSCT relapsed patients.</p>" ] ] ] "bibliografia" => array:2 [ "titulo" => "References" "seccion" => array:1 [ 0 => array:2 [ "identificador" => "bibs0015" "bibliografiaReferencia" => array:5 [ 0 => array:3 [ "identificador" => "bib0030" "etiqueta" => "1" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Acute myeloid leukaemia relapsing after allogeneic haematopoietic stem cell transplantation: prognostic factors and impact of initial therapy of relapse" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "A.B.M. Lim" 1 => "C. Curley" 2 => "C.Y. Fong" 3 => "I. Bilmon" 4 => "A. Beligaswatte" 5 => "D. Purtill" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1111/imj.13522" "Revista" => array:2 [ "tituloSerie" => "Intern Med J" "fecha" => "2017" ] ] ] ] ] ] 1 => array:3 [ "identificador" => "bib0035" "etiqueta" => "2" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Donor lymphocyte infusion following allogeneic hematopoietic stem cell transplantation" "autores" => array:1 [ 0 => array:2 [ "etal" => false "autores" => array:2 [ 0 => "C. Roddie" 1 => "K. Peggs" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1517/14712598.2011.554811" "Revista" => array:6 [ "tituloSerie" => "Expert Opin Biol Ther" "fecha" => "2011" "volumen" => "11" "paginaInicial" => "473" "paginaFinal" => "487" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/21269237" "web" => "Medline" ] ] ] ] ] ] ] ] 2 => array:3 [ "identificador" => "bib0040" "etiqueta" => "3" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Prophylactic donor lymphocyte infusion (DLI) followed by minimal residual disease and graft-versus host disease guided multiple DLIs could improve outcomes after allogeneic hematopoietic stem cell transplantation in patients with refractory/relapsed acute leukemia" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "C.H. Yan" 1 => "Q.F. Liu" 2 => "D.P. Wu" 3 => "X. Zhang" 4 => "L.P. Xu" 5 => "X.H. Zhang" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1016/j.bbmt.2017.04.028" "Revista" => array:6 [ "tituloSerie" => "Biol Blood Marrow Transplant" "fecha" => "2017" "volumen" => "23" "paginaInicial" => "1311" "paginaFinal" => "1319" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/28483716" "web" => "Medline" ] ] ] ] ] ] ] ] 3 => array:3 [ "identificador" => "bib0045" "etiqueta" => "4" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Second reduced intensity conditioning allogeneic transplant as a rescue strategy for acute leukaemia patients who relapse after an initial RIC allogeneic transplantation: analysis of risk factors and treatment outcomes" "autores" => array:1 [ 0 => array:2 [ "etal" => true "autores" => array:6 [ 0 => "R. Vrhovac" 1 => "M. Labopin" 2 => "F. Ciceri" 3 => "J. Finke" 4 => "E. Holler" 5 => "J. Tischer" ] ] ] ] ] "host" => array:1 [ 0 => array:1 [ "Revista" => array:5 [ "tituloSerie" => "Bone Marrow Transpl" "fecha" => "2016" "volumen" => "51" "paginaInicial" => "186" "paginaFinal" => "193" ] ] ] ] ] ] 4 => array:3 [ "identificador" => "bib0050" "etiqueta" => "5" "referencia" => array:1 [ 0 => array:2 [ "contribucion" => array:1 [ 0 => array:2 [ "titulo" => "Clinical options after failure of allogeneic hematopoietic stem cell transplantation in patients with hematologic malignancies" "autores" => array:1 [ 0 => array:2 [ "etal" => false "autores" => array:2 [ 0 => "A. Petrovic" 1 => "G. Hale" ] ] ] ] ] "host" => array:1 [ 0 => array:2 [ "doi" => "10.1586/eci.11.24" "Revista" => array:6 [ "tituloSerie" => "Expert Rev Clin Immunol" "fecha" => "2011" "volumen" => "7" "paginaInicial" => "515" "paginaFinal" => "527" "link" => array:1 [ 0 => array:2 [ "url" => "https://www.ncbi.nlm.nih.gov/pubmed/21787195" "web" => "Medline" ] ] ] ] ] ] ] ] ] ] ] ] ] "idiomaDefecto" => "en" "url" => "/23870206/0000015100000009/v1_201811220618/S2387020618303073/v1_201811220618/en/main.assets" "Apartado" => array:4 [ "identificador" => "43311" "tipo" => "SECCION" "en" => array:2 [ "titulo" => "Scientific letters" "idiomaDefecto" => true ] "idiomaDefecto" => "en" ] "PDF" => "https://static.elsevier.es/multimedia/23870206/0000015100000009/v1_201811220618/S2387020618303073/v1_201811220618/en/main.pdf?idApp=UINPBA00004N&text.app=https://www.elsevier.es/" "EPUB" => "https://multimedia.elsevier.es/PublicationsMultimediaV1/item/epub/S2387020618303073?idApp=UINPBA00004N" ]
Journal Information
Share
Download PDF
More article options
Scientific letter
Rescue treatment of patients with relapsed acute leukemia after first allogeneic hematopoietic stem cell transplantation
Tratamiento de rescate de pacientes con leucemia aguda en recaída después de un primer trasplante alogénico de progenitores hematopoyéticos
Visits
1
This item has received
Article information
These are the options to access the full texts of the publication Medicina Clínica (English Edition)
Subscriber
Subscribe
Purchase
Contact
Phone for subscriptions and reporting of errors
From Monday to Friday from 9 a.m. to 6 p.m. (GMT + 1) except for the months of July and August which will be from 9 a.m. to 3 p.m.
Calls from Spain
932 415 960
Calls from outside Spain
+34 932 415 960
E-mail