“Tsutsugamushi triangle” is an imaginary geographical region (in Asia-Pacific area including, but not exclusively limited to, Korea, Japan, China, India, Taiwan, Thailand, Indonesia, Sri Lanka, and the Philippines) that borrowed its name from the mite-borne rickettsial zoonosis, scrub typhus, an acute undifferentiated febrile illness, caused by Orientia tsutsugamushi.1 Various neurological complications are ever-emerging among the plethora of clinical presentations.2 This spectrum of neurological manifestations includes aseptic meningitis, meningoencephalitis, cerebellitis, acute disseminated encephalomyelitis, cerebral vasculitis, stroke, myelitis, polyradiculoneuropathy, and cranial nerve palsies.2–4 However, the involvement of corpus callosum has been rarely reported.5
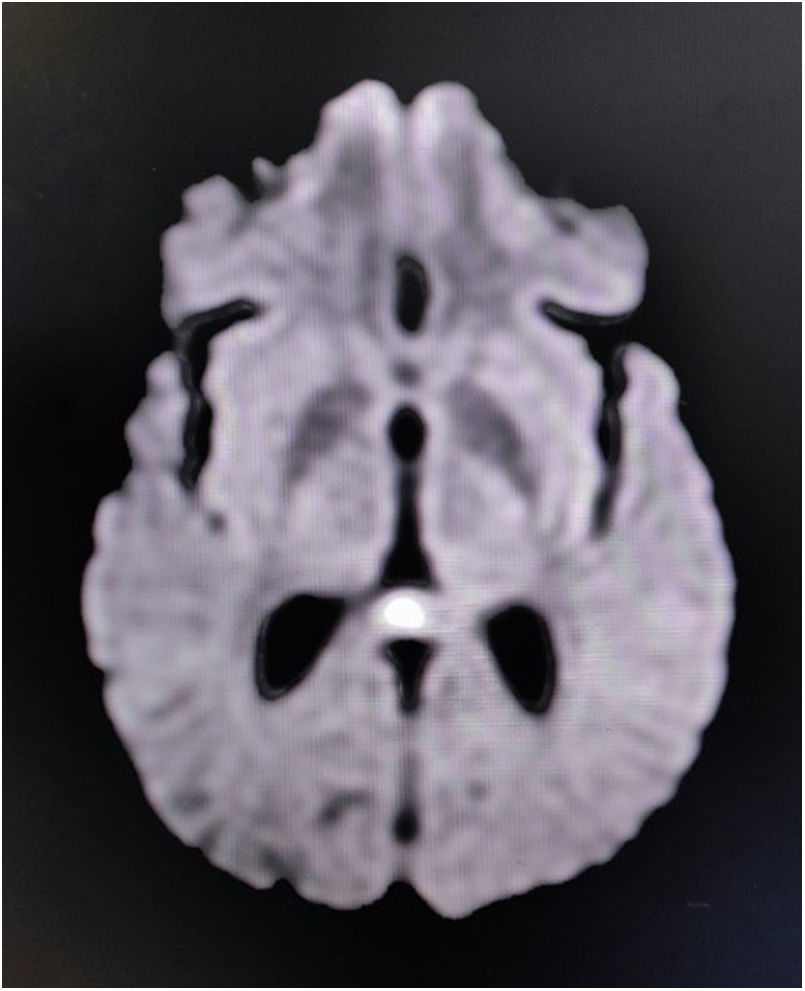
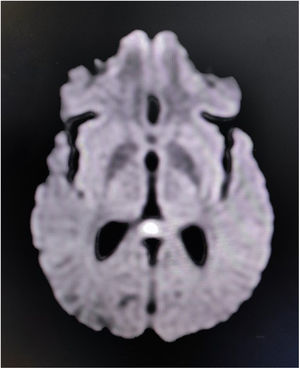
Reversible splenial lesion syndrome (RESLES) or cytotoxic lesion of the corpus callosum (CLOCCs) are usually reversible relatively symmetrical tend-to-be midline lesions, which are observed on brain magnetic resonance imaging (MRI) as unenhanced areas, which show low-diffusion on diffusion-weighted images.6,7 Amid multitudes of etiologies of RESLES/CLOCCs, acute infective encephalitis is one of the most important.6,7 RESLES/CLOCCs, particularly when involving the splenium, may sometimes give rise to callosal disconnection syndromes (mostly reversible).6,7
We report a novel case of pure alexia (also called alexia without agraphia or word blindness) and RESLES/CLOCCs, in a previously healthy young Indian man following Tsutsugamushi disease who responded well to antibiotic therapy.
A previously healthy right-handed 25-year-old man from West Bengal (India) was admitted to our outpatient clinic with fever, holocranial headache for the last ten days, and inability to read any written document for the last three days; however, he had no problem in seeing objects. His past medical, travel and surgical histories were non-contributory. The patient's oropharyngeal swab test for SARS-CoV-2 was negative by qualitative real-time reverse-transcriptase-polymerase-chain-reaction assay. He was febrile (38.5°C) and tachycardic (102min–1). Other vital signs were within physiological ranges. On neurological examination, we found that he could not read a written script, albeit he could normally write when asked. Other language domains were normal, including comprehension, word expression, and repetition. The remaining neurological and neuro-ophthalmological examination was normal. Signs of intracranial hypertension and meningeal irritation were absent.
Complete blood cell count revealed neutrophilic leukocytosis with relative lymphopenia and a raised erythrocyte sedimentation rate (56mm/h). Liver, kidney, and thyroid function tests were normal, as well as arterial blood gases and serum electrolytes. Serologic tests for malaria parasites, dengue, Japanese encephalitis, and SARS-CoV-2 were negative. Urinalysis and urinary culture ruled out urinary tract infection. Sputum culture failed to show any evidence of infection. Serologic tests for hepatitis B, C, and HIV (1,2) were negative. A chest X-ray and ultrasonographic scan of the abdomen were done and were normal. The brain's MRI revealed non-enhancing low-diffusion signals arising from the splenium of corpus callosum, suggestive of RESLES/CLOCCs (Fig. 1). Cerebrospinal fluid (CSF) analysis was suggestive of infectious etiology (mild increased opening pressure, cell count 30/μl, all lymphocytes; raised protein levels of 80mg/dL; and low glucose levels 30mg/dL, corresponding capillary blood glucose level was 118mg/dL). Gram staining and Ziehl-Neelsen staining were negative. CSF was further tested by PCR method for relevant neuro-infectious agents (e.g., Japanese encephalitis, HSV-1,2, VZV, EBV, CMV, HHV-6,7, enteroviruses, adenoviruses, neurotuberculosis, neuroborreliosis, and neurosyphilis), which came out negative. However, serologic testing for scrub typhus infection (by IgM-ELISA method) was positive. According to the existing institutional management policy, he was put on oral doxycycline (200mg/day) and azithromycin (500mg/day) for a week. He became afebrile within 48–72h of systemic antibiotic therapy. Cognitive dysfunction persisted for another two weeks before complete resolution.
Pure alexia without visual deficits and agraphia is an extremely rare and complex neurological deficit in which the person cannot read, and the ability to write down remains grossly intact.8–11 Alexia has been hypothesized to be caused by damage to the pathways carrying visual input from both striatal cortices to the dominant angular gyrus via the splenium of the corpus callosum.8–10 The dominant angular gyrus is situated in the inferior parietal lobule of the cerebral hemisphere, usually the left, and is engaged in phoneme processing during language comprehension and phoneme production during repetition and speech.8–10 Several studies have supported this theory by describing variants such as (1) the splenial-occipital syndrome, which also involves the dominant occipital region12; (2) the spleniogeniculate variation following a left lateral geniculate body infarct with associated damage to the dominant occipitotemporal paraventricular white matter13; and (3) subangular alexia caused by more dorsal and rostral lesions in the dominant parieto-occipital white matter.14 Except for the latter, these variants are associated with right homonymous hemianopia or hemiachromatopsia due to damage to the direct visual pathways. All these studies suggest that white matter damage is the pathological basis of the disconnection syndrome, even when the lesion was located in the occipitotemporal junction or the inferior temporal gyrus with no visible involvement of white matter tracts. However, in 1997, one of the authors of the present manuscript (J.B.-L.) proposed that other critical areas might be affected in cases of pure alexia without obvious white matter lesion, calling the disconnection syndrome paradigm into question.11 From this different perspective, later studies have identified a functional region in the inferior temporal cortex, namely the fusiform gyrus and adjacent occipitotemporal cortex, called the visual word formation area, which is involved in rapid word recognition and fluent reading.15
Our patient had pure alexia without visual deficits and agraphia secondary to scrub typhus. Despite the existence of a radiologically demonstrable splenial lesion, we suspect that the cause was an ultrastructural/functional involvement of the visual word formation area, which conventional MRI did not pick up. Unfortunately, due to infrastructural shortcomings, rapid clinical reversibility, and financial restraints, we could not perform a functional MRI on this patient.
Authors’ contributionsAll authors contributed to the study conception. RG carried outpatient information, diagnosis and management and data collection. The first draft of the manuscript was written by RG and edited by SD, DR, and JBL. All authors read and approved the final manuscript.
ConsentInformed consent was obtained from the patient for inclusion into this study.
FundingThis research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interestThe authors have no relevant financial or non-financial interests to declare.