The evaluation of clinical outcomes in rheumatoid arthritis patients, including the patient's perspective, allows the inclusion of the patient's perspective in clinical decision-making using instruments designed for this purpose.
ObjectiveTo identify and describe the instruments validated in Spanish to evaluate clinical outcomes in rheumatoid arthritis in daily clinical practice and from the patient's perspective, including disease activity, functionality, impact of the disease, adherence to treatment and quality of life.
Materials and methodsA review of the literature in various databases (PubMed, Scopus, Bireme, Scielo) was conducted looking for questionnaires that evaluate clinical outcomes in rheumatoid arthritis patients, including those with a validated Spanish translation that allow self-assessment of the disease by the patient and clinical monitoring.
ResultsFifteen questionnaires were identified and selected that met the inclusion criteria. Four instruments were found that evaluate quality of life, three that evaluate functionality, four that evaluate disease impact, one that evaluate disease activity, and three that evaluate adherence to treatment.
ConclusionQuestionnaires and scales that evaluate rheumatoid arthritis allow a clinical approach to the evolution of the disease as perceived and reported by the patients, optimizing clinical follow up. Implementation of these tools in clinical decision making allows for an improvement in quality of care of patient with this condition.
La evaluación de desenlaces clínicos en pacientes con artritis reumatoide, considerando la perspectiva del paciente, permite la inclusión del concepto del paciente en la toma de decisiones clínicas mediante la utilización de instrumentos diseñados para tal fin.
ObjetivoIdentificar y describir los instrumentos validados en castellano para evaluar desenlaces clínicos en artritis reumatoide en la práctica clínica diaria y desde la perspectiva del paciente, incluyendo la actividad de la enfermedad, la funcionalidad, el impacto de la enfermedad, la adherencia al tratamiento y la calidad de vida.
Materiales y métodosSe realizó una búsqueda de artículos relacionados con la evaluación de desenlaces clínicos en artritis reumatoide. Se revisaron las siguientes bases de datos: PubMed, Scopus, Bireme y Scielo, en busca de instrumentos o cuestionarios autoadministrados, que tengan traducción validada al castellano y permitan la evaluación y monitorización clínica.
ResultadosSe identificaron y seleccionaron 15 cuestionarios que cumplen los criterios de inclusión. Se encontraron 4 instrumentos que evalúan la calidad de vida, 3 que valoran la funcionalidad, 4 que evalúan el impacto de la enfermedad y un instrumento que evalúa la actividad de la enfermedad. Así mismo, se reportan 3 instrumentos que evalúan la adherencia al tratamiento.
ConclusiónLos cuestionarios y las escalas de evaluación en artritis reumatoide permiten ofrecer una aproximación clínica de la evolución de la enfermedad percibida y reportada por los pacientes, con lo cual se optimiza el seguimiento clínico. La implementación de estas herramientas en la toma de decisiones permitirá mejorar la calidad de la atención de estos pacientes.
Rheumatoid arthritis (RA) is a chronic inflammatory disease that affects the population between 35 and 50 years of age, and without adequate treatment can lead to irreversible damage, disability and deterioration in the quality of life.1,2 This disease has a variable clinical presentation and multiple risk factors, both genetic and environmental. In developing countries, the prevalence of this disease ranges between 0.1 and 0.5%.1,2 In the particular case of Colombia, few records related to prevalence and incidence are available. In these studies, a greater predominance has been found in the female gender, with an average age of 50–54 years, and with comorbidities that represent an additional risk factor for this population.3
Patients with RA have a higher risk of infection, compared with the rest of the general population that does not suffer from this disease,4 which is due to factors such as immunosuppression, chronic inflammatory state, comorbidities, disease activity, and immunomodulatory drugs.5 However, the majority of infections related to these patients are of bacterial etiology, and mainly affect the respiratory tract, the skin, and the musculoskeletal system. Likewise, the evidence has not demonstrated an increased risk of COVID-19 infection in these patients.4,5 Nevertheless, patients with RA have had to face a great impact during the pandemic, due to less access to health systems, social isolation, anxiety and depression, among other factors that influence quality of life and prognosis.5
In recent years, the health systems have been faced with a new reality related to the COVID-19 pandemic.6 Due to the measures of mobility restriction and social isolation, the need to look for alternatives to reduce the exposure of patients, health professionals and general population was generated. In a recent survey, 82% of the rheumatologists switched from in-person to virtual consultations in 2020, with a subjective report of increase in exacerbations by 39%, with a high frequency of poor education or misinformation of the patients.7 In this context, it is necessary to generate other measures so that these patients have adequate access to health services, which guarantee optimal clinical follow-up of their condition.
As a consequence of this epidemiological situation, the frequency of visits of patients with immune-mediated diseases to care centers was considerably reduced. In this sense, the need arises to implement other forms of follow-up for these chronic diseases, without requiring direct contact with the specialist or the hospital environment.8 In this way, the evaluation of patient-reported outcomes (PRO) is performed physically, filling out validated questionnaires or scales, with the limitation that it is carried out in person.9
All of the above has encouraged the electronic use of PROs, since this gives the patients the possibility of evaluating their symptoms periodically, and allows the clinician to do a better follow-up.9,10 In this context, different strategies have been implemented, such as applications for mobile phones and online platforms. These applications allow to follow-up the disease through the evaluation from the patient's perspective.11 However, the application of these strategies may be limited in Latin America due to language barriers (instruments not translated or validated into Spanish) and in some regions due to limited technological access (Internet).
Therefore, the objective of the present review is to identify and describe the instruments (e.g., questionnaires or PROs) validated in Spanish to evaluate clinical outcomes in RA from the patient's perspective (disease activity, functionality, impact of the disease, adherence to treatment and quality of life).
Materials and methodsThe study population is made up of patients with RA, in whom the aim is to evaluate through questionnaires in virtual scenarios, the impact of the disease as perceived by the patient himself, in terms of the impact on quality of life, functional capacity, adherence to treatment, disease activity and impact. The PICO strategy (patient or problem, intervention, comparison, outcome) was used to define the search parameters, as described below:
P: Adult patients with a diagnosis of RA.
I: Questionnaires, instruments, PROs translated and validated in Spanish.
C: Does not apply.
O: Disease activity, functionality, impact of the disease, adherence to treatment and quality of life.
A search and subsequent review of articles related to the evaluation of clinical outcomes using instruments/questionnaires in patients with RA was conducted. The search was carried out in the Pubmed, Bireme, Scopus and Scielo databases, and included the following MeSH terms: [«rheumatoid arthritis», «questionnaire», «instrument», «quality of life», «outcome assessment», «Disease activity», «disease impact», «physical function» «treatment adherence» «compliance»], both in English and Spanish. The bibliographic references of the articles found were also analyzed and searched, in order to include studies that could be valid for the review and that were not found in the databases.
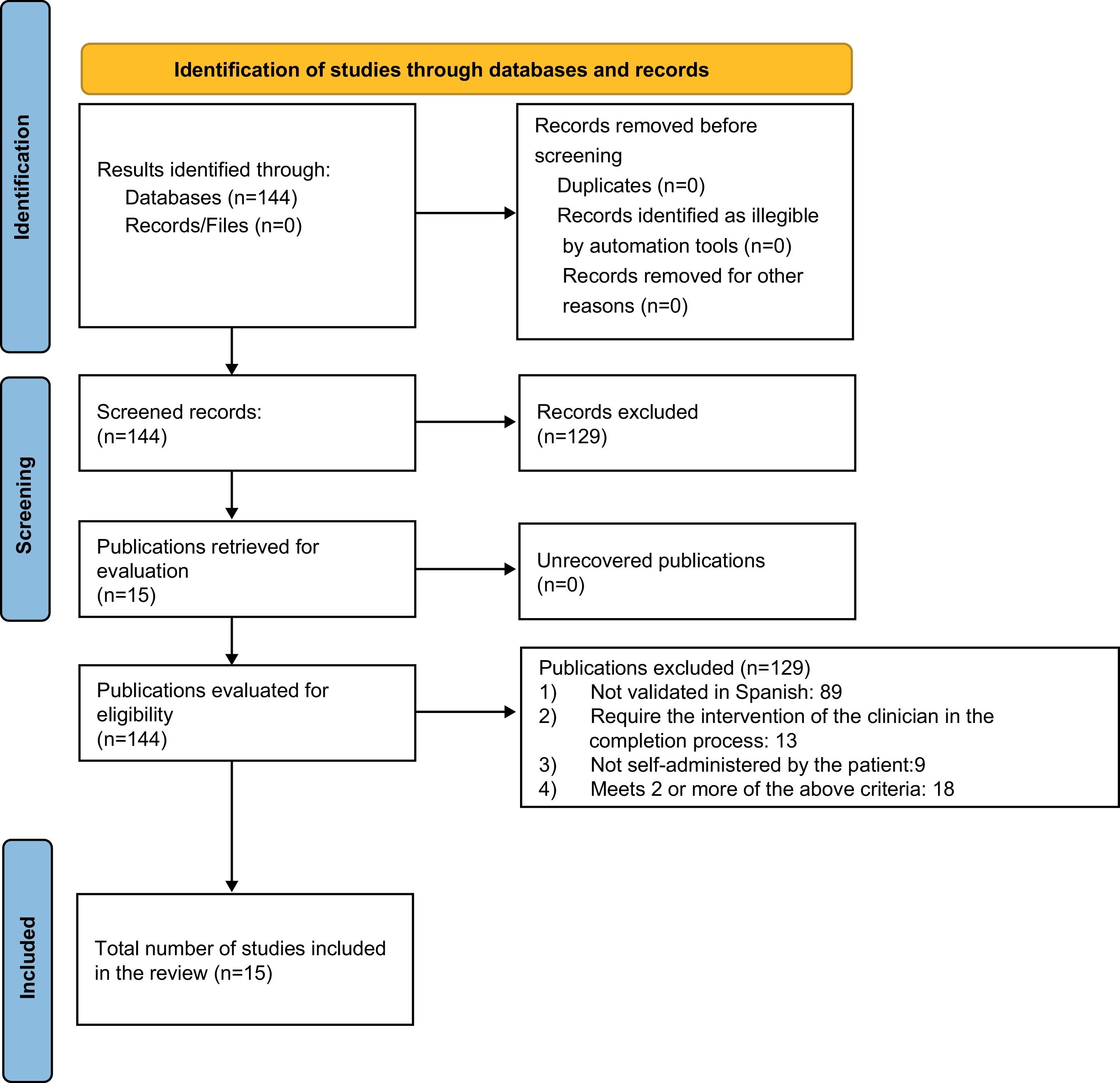
Within the inclusion criteria, instruments validated in Spanish, used to evaluate the outcomes of the disease and that can be answered in their entirety by the patient without the need for physician intervention or additional laboratory data (self-administered) were selected (see the Prisma diagram, Fig. 1). The data collected were analyzed using tables that describe the name of the instrument, the outcome evaluated (disease activity, functionality, quality of life, adherence to treatment, and impact of the disease), the number of questions in the instrument, the time it takes the patient to answer it, and the interpretation of the score.
PRISMA diagram.
*Consider, if feasible, reporting the number of records identified from each database or record searched (rather than the total number across all databases/records).
**If automation tools were used, indicate how many records were excluded by a human being and how many were excluded by the automation tools.
Source: Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:71, https://doi.org/10.1136/bmj.n71.
A total of 144 questionnaires were found, of which 15 met the inclusion criteria for the review. They were divided into 5 categories according to the characteristic or outcome assessed in the disease grouped as follows: quality of life (Table 1), functionality (Table 2), impact of the disease (Table 3), disease activity (Table 4), and adherence to treatment (Table 5).
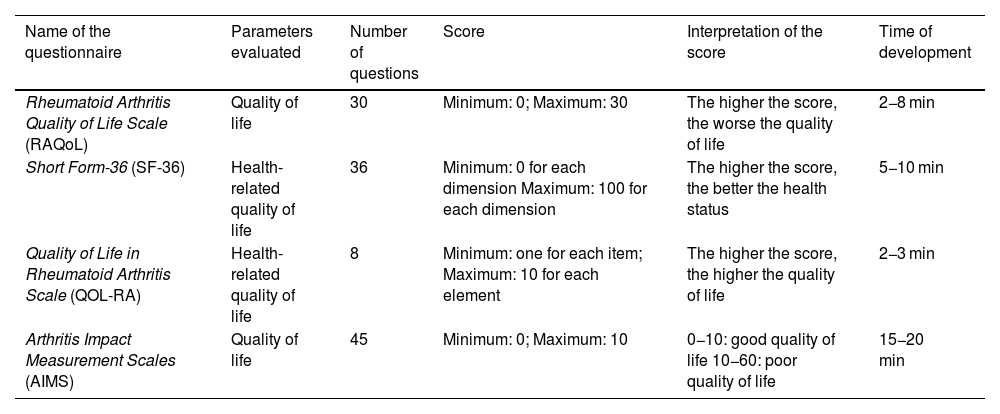
Instruments that assess quality of life in RA.
| Name of the questionnaire | Parameters evaluated | Number of questions | Score | Interpretation of the score | Time of development |
|---|---|---|---|---|---|
| Rheumatoid Arthritis Quality of Life Scale (RAQoL) | Quality of life | 30 | Minimum: 0; Maximum: 30 | The higher the score, the worse the quality of life | 2−8 min |
| Short Form-36 (SF-36) | Health-related quality of life | 36 | Minimum: 0 for each dimension Maximum: 100 for each dimension | The higher the score, the better the health status | 5−10 min |
| Quality of Life in Rheumatoid Arthritis Scale (QOL-RA) | Health-related quality of life | 8 | Minimum: one for each item; Maximum: 10 for each element | The higher the score, the higher the quality of life | 2−3 min |
| Arthritis Impact Measurement Scales (AIMS) | Quality of life | 45 | Minimum: 0; Maximum: 10 | 0−10: good quality of life 10−60: poor quality of life | 15−20 min |
RA: rheumatoid arthritis.
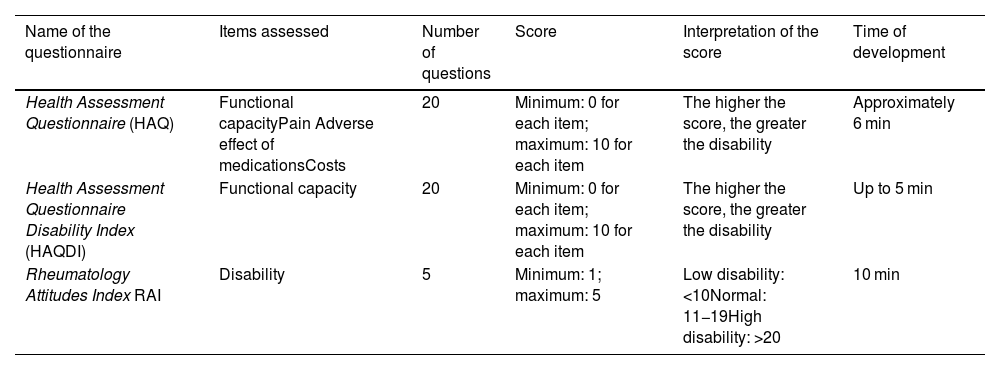
Instruments that assess patient functionality.
| Name of the questionnaire | Items assessed | Number of questions | Score | Interpretation of the score | Time of development |
|---|---|---|---|---|---|
| Health Assessment Questionnaire (HAQ) | Functional capacityPain Adverse effect of medicationsCosts | 20 | Minimum: 0 for each item; maximum: 10 for each item | The higher the score, the greater the disability | Approximately 6 min |
| Health Assessment Questionnaire Disability Index (HAQDI) | Functional capacity | 20 | Minimum: 0 for each item; maximum: 10 for each item | The higher the score, the greater the disability | Up to 5 min |
| Rheumatology Attitudes Index RAI | Disability | 5 | Minimum: 1; maximum: 5 | Low disability: <10Normal: 11−19High disability: >20 | 10 min |
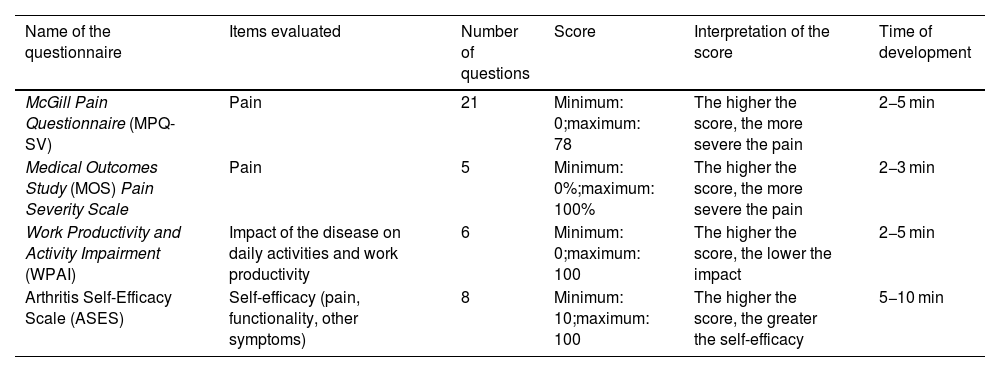
Questionnaires that assess the impact of the disease on the patient.
| Name of the questionnaire | Items evaluated | Number of questions | Score | Interpretation of the score | Time of development |
|---|---|---|---|---|---|
| McGill Pain Questionnaire (MPQ-SV) | Pain | 21 | Minimum: 0;maximum: 78 | The higher the score, the more severe the pain | 2−5 min |
| Medical Outcomes Study (MOS) Pain Severity Scale | Pain | 5 | Minimum: 0%;maximum: 100% | The higher the score, the more severe the pain | 2−3 min |
| Work Productivity and Activity Impairment (WPAI) | Impact of the disease on daily activities and work productivity | 6 | Minimum: 0;maximum: 100 | The higher the score, the lower the impact | 2−5 min |
| Arthritis Self-Efficacy Scale (ASES) | Self-efficacy (pain, functionality, other symptoms) | 8 | Minimum: 10;maximum: 100 | The higher the score, the greater the self-efficacy | 5−10 min |
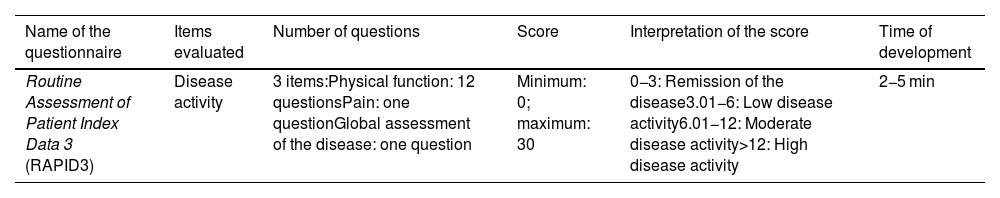
Instruments that assess disease activity.
| Name of the questionnaire | Items evaluated | Number of questions | Score | Interpretation of the score | Time of development |
|---|---|---|---|---|---|
| Routine Assessment of Patient Index Data 3 (RAPID3) | Disease activity | 3 items:Physical function: 12 questionsPain: one questionGlobal assessment of the disease: one question | Minimum: 0; maximum: 30 | 0−3: Remission of the disease3.01−6: Low disease activity6.01−12: Moderate disease activity>12: High disease activity | 2−5 min |
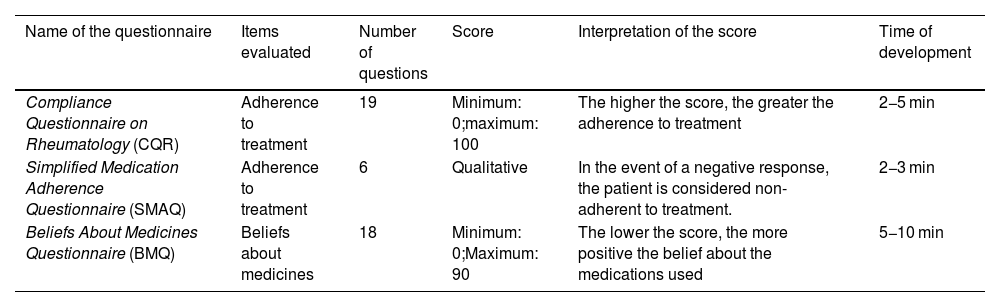
Questionnaires that assess adherence to treatment.
| Name of the questionnaire | Items evaluated | Number of questions | Score | Interpretation of the score | Time of development |
|---|---|---|---|---|---|
| Compliance Questionnaire on Rheumatology (CQR) | Adherence to treatment | 19 | Minimum: 0;maximum: 100 | The higher the score, the greater the adherence to treatment | 2−5 min |
| Simplified Medication Adherence Questionnaire (SMAQ) | Adherence to treatment | 6 | Qualitative | In the event of a negative response, the patient is considered non-adherent to treatment. | 2−3 min |
| Beliefs About Medicines Questionnaire (BMQ) | Beliefs about medicines | 18 | Minimum: 0;Maximum: 90 | The lower the score, the more positive the belief about the medications used | 5−10 min |
Questionnaire that evaluates quality of life, specifically in patients with RA, through 30 questions with a “yes” or “no” response regarding activities of daily living.12 It has been validated in Spanish by Pacheco-Tena et al. in the Mexican population.13
Short Form-36 (SF-36)Generic questionnaire that evaluates health-related quality of life, through 36 questions that cover 8 of the main health dimensions evaluated by questionnaires. A higher score is related to a better health status.14,15 It has been validated in Spanish in 1995 in the Spanish population.16
Quality of Life in Rheumatoid Arthritis Scale (QOL-RA)Specific questionnaire for RA that assesses health-related quality of life. It consists of 8 questions that allow us to assess quality of life in different aspects, where each question gives a score from 1 to 10, and a higher total score is related to a better quality of life. It has been validated in Spanish by Danao et al. in 2001 in the American population.17
Arthritis Impact Measurements Scales (AIMS)Generic questionnaire developed for patients with different types of arthritis that assesses their physical, emotional, and social well-being. It consists of 45 questions divided into 9 categories, where a score will be given to each one and a higher score will be related to a worse quality of life.18 It has been validated in Spanish by Abello-Banfi et al. in 1994 in the Mexican population.19
Instruments that assess functional statusHealth Assessment Questionnaire (HAQ)The HAQ is considered the gold standard for the assessment of functional capacity in patients with RA, since it makes an overall evaluation thereof. It is a questionnaire originally developed for patients with rheumatic diseases and in its complete version it evaluates the following components: disability, pain, adverse effect of medications and economic cost.20 It has been validated in Spanish by Esteve-Vives et al. in 1993 in a Spanish population.21
Health Assessment Questionnaire Disability Index (HAQDI)Questionnaire originally developed for patients with RA and osteoarthritis, now widely used in other rheumatic diseases for the assessment of the patient's functional status. It consists of 20 questions distributed into 8 different categories in relation to the activities of daily living. Each question assigns a score, in which a higher final score is related to a higher degree of disability.12 It differs from the HAQ since it only evaluates the disability component.22 It has a validation into Spanish performed by Cardiel in 1993 in a Mexican population.23
Rheumatology Attitudes Index (RAI)Questionnaire developed to assess disability or helplessness in patients with rheumatic diseases. The original questionnaire consists of 15 questions and has been shortened and validated to a questionnaire of 5 questions specific to the disability component. According to the score given in each question, the total score is directly related to the degree of disability.24 Its 5-item version has been validated in Spanish by Escalante et al. in 1999 in the Mexican population.25
Questionnaires that assess the impact of the disease on the patientMcGill Pain Questionnaire (MPQ-SV)Questionnaire developed to evaluate the sensitive, affective and evaluative aspects of pain in patients with chronic pain. It contains 21 questions and each descriptor is assigned a score that in the total sum relates a higher score to a greater perception of pain.26 Validated in Spanish by Lázaro et al. in Spanish population in 1994.27
Medical Outcomes Study (MOS) Pain Severity ScaleQuestionnaire developed in a population of patients with chronic pathologies to assess the intensity, duration and frequency of pain. It contains 5 questions that generate a total score of 0−100, where a higher score is related to more severe pain.28 Questionnaire validated in Spanish by González et al. in 1995 in the Hispanic population in the United States.29
Work Productivity and Activity Impairment (WPAI)Questionnaire developed for patients with any health problem, it has 6 items that evaluate the impact of the disease on daily activities and work production.30 Validated in Spanish by Lambert et al. in 2014.31
Arthritis Self-Efficacy ScaleQuestionnaire developed to assess the self-efficacy of patients with arthritis. The original questionnaire contains 20 items, each of which has a score between 1 and 10, and a total sum with a higher score being related to a greater self-efficacy.32 It was validated in Spanish by González et al. in 1995 in the Hispanic population in the United States, with a modification to a total of 8 items.29
Instruments that assess disease activityRoutine Assessment of Patient Index Data 3 (RAPID3)Questionnaire developed for patients with RA to evaluate physical function, pain, and global assessment of the disease. Each item has its own score and, according to the total sum, ranges are given to evaluate the disease activity (Table 3).33,34 Questionnaire validated by Maldonado et al. in an Argentinian population in 2011.33
Instruments that assess adherence to treatmentCompliance Questionnaire of Rheumatology (CQR)Questionnaire originally developed to assess adherence to pharmacological treatment in patients with RA, polymyalgia rheumatica and gout. It contains 19 items in which the patient expresses his/her level of agreement with each one and generates a final score from 0 to 100. A higher score reflects greater adherence to treatment.26 Questionnaire validated in Spanish by Fernández-Ávila et al. in the Colombian population in 2019.35
Simplified Medication Adherence Questionnaire (SMAQ)Questionnaire originally developed in Spanish for the evaluation of adherence to pharmacological treatment in a group of Spanish population with the human immunodeficiency virus (HIV); however, it has been widely used for the assessment of adherence in different clinical situations.
It consists of 6 questions that evaluate different facets of non-compliance with treatment; 4 of a dichotomous nature with answers (Yes/No) and 2 of a semi-quantitative nature. A result of a non-adherent patient is obtained when there is any response that shows non-compliance with treatment and gives a positive result to the questionnaire.36
Beliefs about Medicines Questionnaire (BMQ)Questionnaire developed for the evaluation of the cognitive representation of medications in patients with different morbidities that has been used for the evaluation of beliefs regarding medicines. It consists of two scales, one of the opinion of the individual regarding medications in general and another of the opinion regarding the specific medications the patient takes for his/her pathology. A value from 1 to 5 is given to each parameter, depending on the response, and the total sum is correlated with greater belief in the concepts assessed in the questionnaire.37 It has been validated in Spanish in different groups with different diseases.38,39
DiscussionThe evaluation of clinical outcomes in musculoskeletal diseases, including RA, taking into account the patient's perspective, is an area of interest in recent years. In the approach to patient-centered care, the incorporation of the patient's perspective in the care process has led to a paradigm shift, because it allows to integrate the concept of the patient into clinical decision-making in a more objective way, by using instruments designed for this purpose.
The follow-up of patients with RA requires to monitor remotely the disease in terms of quality of life, functionality, disease activity and adherence to treatment. In this narrative review, we found a total of 15 questionnaires that were used as scales in various chronic diseases and with specific application in the RA scenario, which evaluate different components of the disease and have been validated in Spanish for their clinical application.
These instruments are particularly useful in patients with RA. This is a condition that can have a negative impact on quality of life (4 questionnaires).40 In this domain, the questionnaires vary in the number of questions, but they coincide in evaluating the physical, social and mental environment according to the patient's self-perception, giving a score that is related to better or worse quality of life.
Another element that must be taken into account is the degree of functionality of the patient, mainly due to the fact that patients with RA present a progressive deterioration in up to 60–90 % of cases, associated with functional and structural changes that lead to physical limitation and subsequent disability.41 Given the foregoing, it is crucial for a comprehensive treatment to be able to evaluate the degree of functionality of the patient, as well as the degree of disability produced by the disease. For this purpose, 3 questionnaires were found where the main item evaluated is functional capacity/disability, while the HAQ is the only one that assesses other additional dimensions, such as pain.
A determining factor in the course and prognosis of RA is the adherence of the patient to immunomodulatory treatments. The lack of adherence is associated with increased relapses of the disease and greater functional limitation.42 Although there may be changes in the patient's self-reported adherence and in that observed in real life, estimating adherence is important for the adjustment and follow-up of treatment.42 To know adherence from the perception of the patient, 3 scales that allow us to assess this component were found; one of these, the CQR, was originally developed in patients with RA, while the SMAQ and the BMQ were developed for patients with other diseases, but have been validated for the assessment of adherence in general. The BMQ questionnaire has a special approach, which evaluates the belief that the patient has regarding the medications he/she uses for his/her disease, so it can be used as a complement to comprehensively address the evaluation of adherence to treatment.
Among the characteristics that determine the impact of the disease on the patient, one of the most prominent is pain, this being the main symptom and the one that has the greatest impact on the patient in several areas, including the psychological one.43 If the variables that are evaluated to perform clinical follow-up in RA are taken into account, both the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) agree that pain is a main variable, both in number of painful joints and global pain, which is what the patient specifically evaluates.43 This variable can be assessed using the visual analogue scale, but interaction with the doctor is ideally required, being a more subjective measure, or by means of two scales that we report, which can give an approximation towards more or less severe pain.
Just as the impact of the disease can be measured in variables of pain and adherence to treatment, which allow an approximation to the prognosis and follow-up, this characteristic can also be measured by analyzing how much impact the disease has had on the productivity of the patient, both in terms of work productivity and daily activities. The foregoing, with the purpose of determining the evolution of the disease, understanding that the objective of the treatment is to improve the quality of life of the patient through a comprehensive intervention that allows a low disease activity, improvement of functionality and finally, remission.44
This review, taking into account the above, constitutes a synthesis of the instruments self-administered by the patient and used to evaluate PRO in RA that are validated in Spanish for use in Spanish-speaking patients in Latin America. The diffusion of these instruments among health professionals will facilitate their implementation in daily clinical practice on a broader basis. In routine clinical practice, the implementation of these questionnaires has several limitations, among which we could mention: restriction of time in the care process for their completion, lack of understanding and comprehension by patients of the questionnaires and lack of knowledge of these tools by health professionals in contact with patients with RA. For this reason, this review aims to be part of the solution in the process of dissemination and socialization of the Patient Reported Outcome Measures (PROMs) in RA.
Among the limitations found in the review, the limited availability of questionnaires validated in Spanish and the limited number of specific instruments for RA are evident. The majority of scales available in the literature are for the assessment of chronic diseases in general, which limits the specificity of the instruments towards this specific disease. Furthermore, these are usually available in the original language of the questionnaire, without a validated translation, which leads to the use of non-validated translations that can alter the result of the original document. Another limitation is that many of these questionnaires are not currently used routinely in the clinical setting.
Given that virtual tools are currently becoming more and more important and that patients must also be knowledgeable about their own disease, with this review we intend to open a door to future research on additional tools that allow remote monitoring of patients. This is because such instruments complement medical practice by facilitating patient monitoring in clinical decision-making and optimizing in-person visits.
ConclusionA set of validated questionnaires in Spanish that allow the evaluation of patients with RA in the multiple components of the disease in daily clinical practice is reported and summarized in this review. These patients require the follow-up of their disease activity, which is decisive for the prognosis and adjustment of treatment. With this information, a better implementation of these questionnaires is expected, allowing a clinical evaluation of the state of the disease from the patient's perspective for decision-making. Further studies and information are required in this area for the generation and validation of questionnaires that allow the evaluation of additional aspects such as clinical outcomes of the disease and that are specific to RA. Likewise, their process of validation in languages other than English, including Spanish, is important so that their scope is much greater and can be implemented routinely in daily clinical practice.
FundingNo funding was received for the development of this work.
Conflict of interestThe authors declare that they have no conflict of interest.