Major health challenges as the increasing number of cases of infections by antibiotic multiresistant microorganisms and cases of Alzheimer's disease have led to searching new control drugs. The present study aims to verify a new way of obtaining bioactive extracts from filamentous fungi with potential antimicrobial and acetylcholinesterase inhibitory activities, using epigenetic modulation to promote the expression of genes commonly silenced. For such finality, five filamentous fungal species (Talaromyces funiculosus, Talaromyces islandicus, Talaromyces minioluteus, Talaromyces pinophilus, Penicillium janthinellum) were grown or not with DNA methyltransferases inhibitors (procainamide or hydralazine) and/or a histone deacetylase inhibitor (suberohydroxamic acid). Extracts from T. islandicus cultured or not with hydralazine inhibited Listeria monocytogenes growth in 57.66±5.98% and 15.38±1.99%, respectively. Increment in inhibition of acetylcholinesterase activity was observed for the extract from P. janthinellum grown with procainamide (100%), when compared to the control extract (39.62±3.76%). Similarly, inhibition of acetylcholinesterase activity increased from 20.91±3.90% (control) to 92.20±3.72% when the tested extract was obtained from T. pinophilus under a combination of suberohydroxamic acid and procainamide. Concluding, increases in antimicrobial activity and acetylcholinesterase inhibition were observed when fungal extracts in the presence of DNA methyltransferases and/or histone deacetylase modulators were tested.
Multiresistant microbial strains have emerged over the last decades1,2 probably related to the extensive use of currently marketed antimicrobial agents. This scenario led to an increase on the search for novel sources of compounds with antimicrobial activity. Essential oils as lemongrass oil3 are an example of new source of compounds with antibiotic activity. In addition, screening of fungal extracts has demonstrated that Aspergillus and Penicillium species produce metabolites with antibacterial activity against potentially pathogenic bacteria species such as Staphylococcus aureus, Salmonella enterica serovar Typhimurium and Pseudomonas aeruginosa.4,5
Alzheimer's disease is the most common cause of dementia and affected approximately 35 million people around the world in 2010.6 One of the mechanisms of Alzheimer's disease involves the reduction in acetylcholine production which implies in gradual loss of memory and learning ability along with other risk factors.7 Based on the cholinergic hypothesis it was discovered that some molecules inhibit acetylcholine degradation by acetylcholinesterase enzyme, increasing the time of action of acetylcholine in the synaptic cleft.8
Fungi are important sources of bioactive secondary metabolites.9,10 It is believed that the fungi represent the second largest kingdom in terms of diversity.11 Ascomycota phylum, in which the genera Penicillium and Talaromyces pertain, has a greater genetic diversity for secondary metabolites production when compared to other fungi groups.12
Besides antimicrobial activity, fungi are important sources of bioactive molecules with acetylcholinesterase inhibition activity.13 This high metabolite diversity observed in fungi could be due to the development of mechanisms for communication, competition and chemical defense over evolution process.14
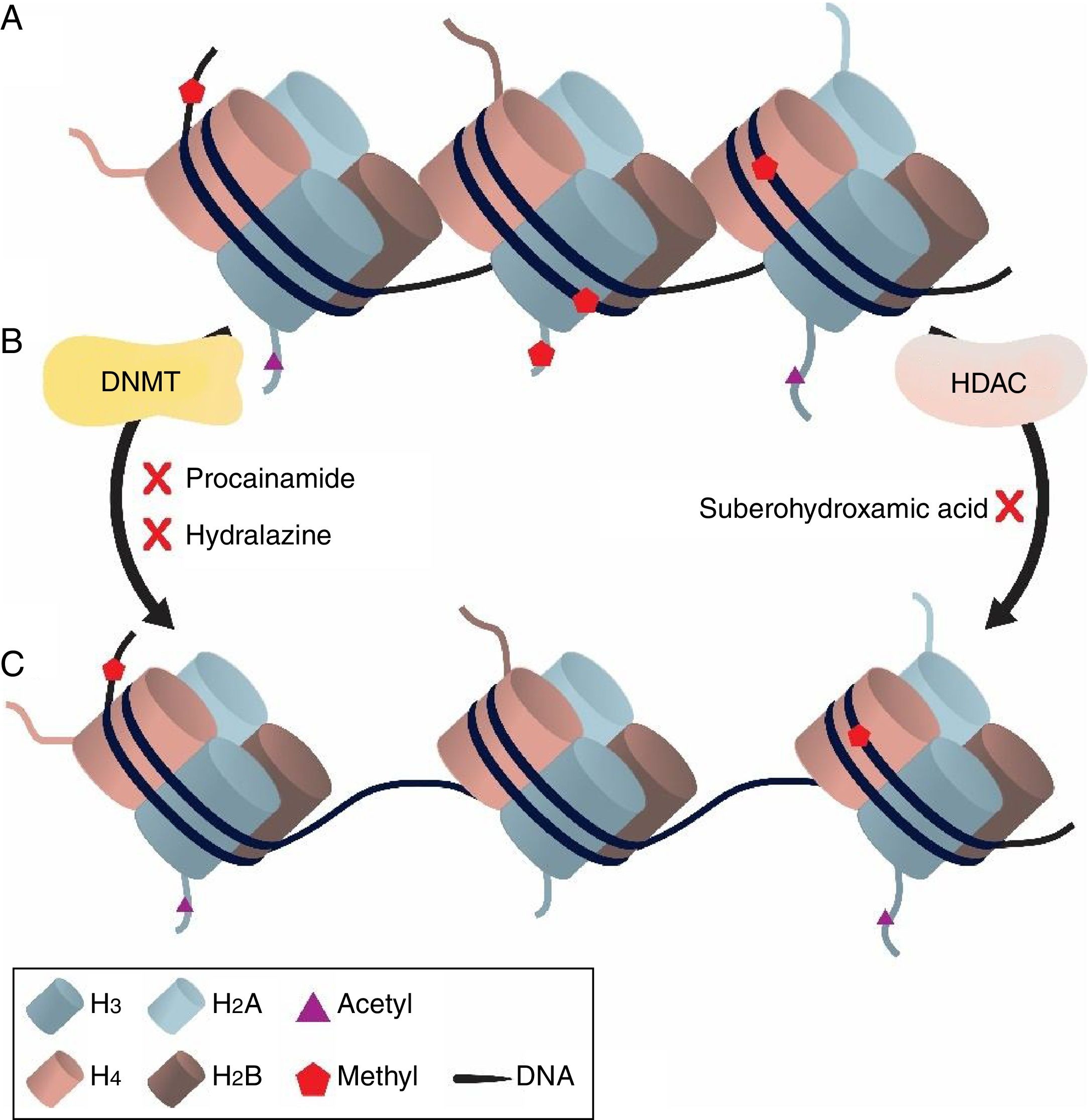
Gene expression in filamentous fungi can be modulated by different conditions such as nutrients availability, light incidence, cultivation temperature, co-cultivation with other microorganisms, and epigenetic modifications.15 The term epigenetics refers to chemical modifications in chromatin structure that can be able to change the conformation of this molecule in order to affect gene expression.16 The basic structure of chromatin organization is given by the formation of nucleosomes, a protein octamer conglomerate (pairs of histone proteins H3, H4, H2A and H2B) in which the double-stranded deoxyribonucleic acid molecule is rolled.17 Heterochromatic regions are less active in terms of gene transcription when compared to euchromatic regions.18 Genes encoding secondary metabolites are usually organized in clusters19 and silenced genes expression can be activated using epigenetic approaches.
Small chemicals, such as hydralazine, suberohydroxamic acid and procainamide are used as inhibitors of enzymes related to epigenetic modification mechanisms, including DNA methyltransferases (DNMT) and histone deacetylases (HDACs). Suberohydroxamic acid acts as a HDACs inhibitor while other epigenetic modulators, e.g. procainamide, inhibit DNMTs.20 Despite results showing that secondary metabolism is affected by epigenetic modulators, few evidences prove a change in qualitative and quantitative patterns of metabolites with biological potential after epigenetic induction of the producing microorganisms.
In this context, the goal of the present study was to evaluate the effect of epigenetic modulation on the antimicrobial activity and acetylcholinesterase inhibition by extracts from fungal growth media of species pertaining to the Penicillium and Talaromyces genera.
Materials and methodsMicroorganisms and inoculum preparationFive filamentous fungi species (Talaromyces funiculosus, Talaromyces islandicus, Talaromyces minioluteus, Talaromyces pinophilus, Penicillium janthinellum) were isolated from soil and stored at the Laboratory of Biotechnology and Bioassays (Federal University of Minas Gerais, Belo Horizonte, MG, Brazil). They were reactivated in tubes containing potato dextrose agar (PDA, Himedia Labs) for 7 days to promote sporulation. Five milliliters of aqueous Tween 80 0.5% (w/v) were added to the tubes containing the reactivated fungal strains and mixed to suspend spores. Spores suspension was standardized using a Neubauer chamber to obtain about 1×107 spores mL−1.
Epigenetic modulationSuberohydroxamic acid, hydralazine and procainamide (Sigma–Aldrich, Darmstadt, Germany) were used as epigenetic modulators. All experiments were conducted in duplicate in Erlenmeyer flasks (500mL) containing 125mL of potato dextrose broth prepared in distilled water (PDB Himedia Labs) previously sterilized in autoclave. In a first group of experiments, epigenetic modulators were added individually (0.1mM) before inoculation with 0.5mL of pre-standardized spore suspension. The second set of experiments was conducted, also in duplicate, in which two epigenetic modulators were added together in the same concentration used in the first experiment (0.1mM each). The following combinations were used: procainamide+suberohydroxamic acid; procainamide+hydralazine; and hydralazine+suberohydroxamic acid. As controls, experiments without addition of epigenetic modulators were run in parallel for each species and to exclude modulators interference in posterior chromatographic analysis, controls were carried without inoculation. Culture was carried out with shaking (100rpm) at room temperature for 15 days. After this, liquid-liquid extraction was performed using ethyl acetate (25mL) for three times. The organic solvent was evaporated down and the resulting extracts were kept in desiccator.
Chromatographic analysis of fungi extractsCrude extracts were submitted to high performance liquid chromatography (HPLC UV/Vis – Shimadzu Prominence LC-20AT) in order to evaluate a possible effect on metabolite diversification promoted by epigenetic modulation by comparing the chromatographic profiles of the experimental and control assays. Methodology used was as described by Bracarense & Takahashi21 by injecting 20μL of each extract (1mgmL−1) with a mobile phase consisting of water acidified with formic acid 0.1% (v/v) and methanol in a C18 column with linear gradient H2O/methanol from 70:30 to 0:100 over 30min. Elution was kept by 10min in the final concentration with a total flow of 0.7mLmin−1. Column was conditioned using the initial concentration of mobile phase for 15min between sample injections. All samples were analyzed at 244nm.
Antimicrobial activity evaluationExtracts antimicrobial activities were evaluated by determining the sensibility percentage based on CLSI broth microdilution standard methodology22 against the following indicator strains: Escherichia coli ATCC 25922, Listeria monocytogenes ATCC 15313 and Candida albicans ATCC 18804. Extracts were diluted in dimethyl sulfoxide (DMSO). Extract concentration varied from 200, 100, 50, 25, 12.5, 6.26, 3.13 to 1.16μgmL−1 after microdilution. Final DMSO concentrations in the samples were kept under 5% (v/v). Ampicillin and miconazole were used as antibiotic and antifungal standards, respectively.
Acetylcholinesterase activity evaluationTests were conducted based on the Ellman spectrophotometric method modified by Rhee et al.23 in 96-well microplates. Eserine was used as standard inhibitor (10mgmL−1), and control test was performed without the presence of acetylcholinesterase inhibitors. Experiments were performed in triplicate. Twenty-five microliters of acetylcholine iodide were dispensed in each well together with 125μL of 5,5′-dithiobis 2-nitrobenzoic acid (DTNB); 50μL of Tris–HCl buffer pH 8, 0.1% (w/v) bovine serum albumin and 25μL of each extract (10mgmL−1) solubilized in DMSO. After a first reading at 405nm, 25μL of acetylcholinesterase (Electrophorus electricus) solution (0.222UmL−1) were added and then, nine readings were performed over a period of 10min. The interference of spontaneous hydrolysis of the substrate was corrected using the average value of the inhibition absorbance (%) before addition of the enzyme. Inhibition of enzyme activity was calculated from the following equation: Inhibition %=[(C−A)×100]/C, where C: control containing enzyme and substrate; A: test containing extract, enzyme and substrate.
Statistical analysisMeans were compared using a multivariable comparing Tukey test using Microsoft Excel 2016 software with Action Stat extension.24 Differences were considered significant for p<0.05.
ResultsRegarding to the five fungal species evaluated in this study, it must be noted that the taxonomic classification of some of them has been recently modified by Yilmaz et al.,25 transferring T. funiculosus, T. pinophilus, T. minioluteus and T. islandicus from Penicillium genus to Talaromyces genus. There are evidences that even close fungi can express only few taxon-specific secondary metabolites.26 Epigenetic treatment of the filamentous fungi with three epigenetic modulators, individually or combined, led to the acquisition of extracts that were then analyzed by HPLC. In addition, antimicrobial activity and acetylcholinesterase inhibition of these extracts were determined.
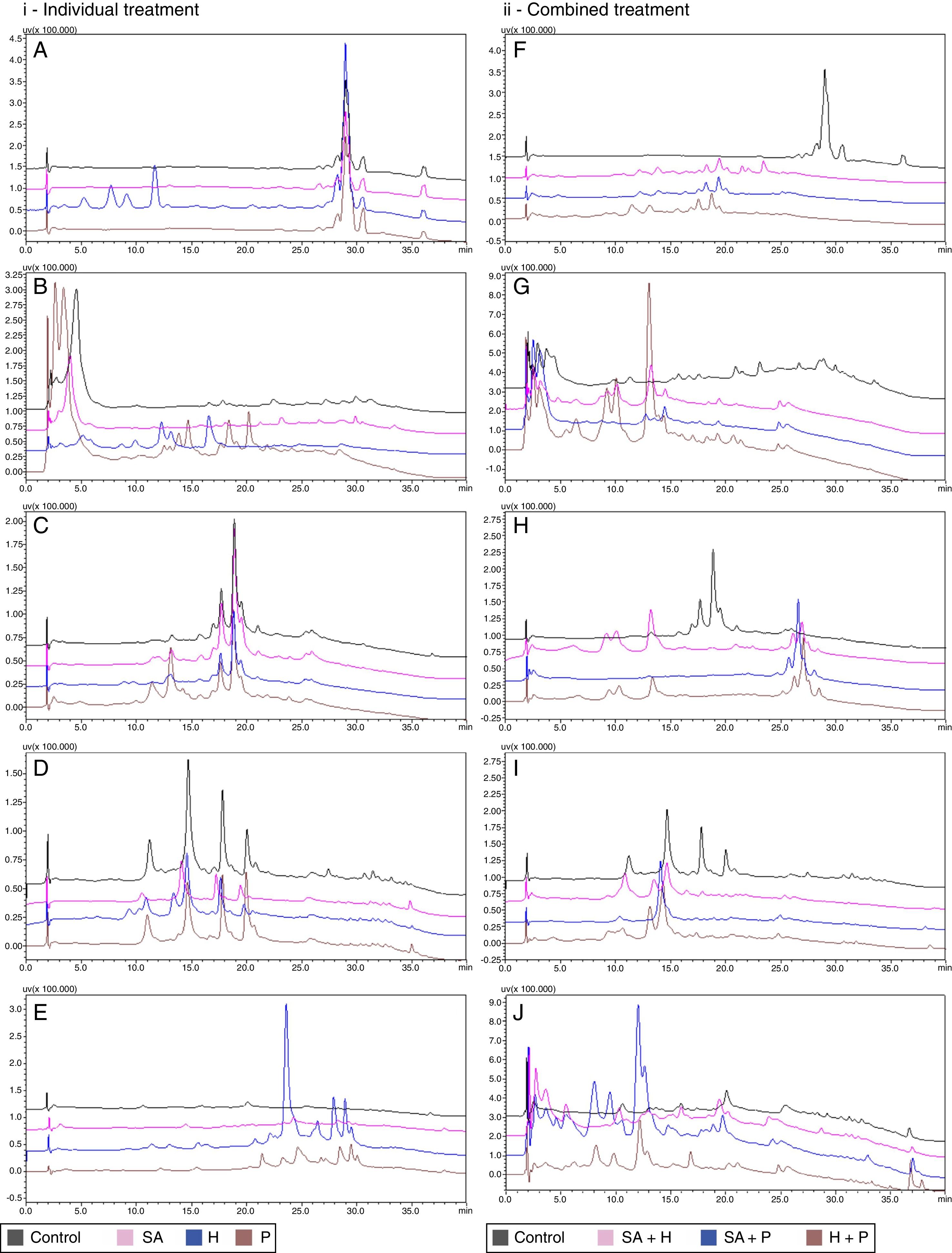
Epigenetic modulationThe HPLC profile of the extracts obtained when suberohydroxamic acid, hydralazine and procainamide were added to the culture broth showed that these compounds act in different ways according to the fungal species utilized. In this study, hydralazine had a higher modulation effect compared to the other epigenetic agents used, showing stimulation of the metabolic differentiation of T. islandicus, T. funiculosus and T. pinophilus (Fig. 1A, D and E) when compared to the control profiles. Similarly, Williams et al.27 related the production of new metabolites by Cladosporium cladosporioides and Verticillium psalliotae by hydralazine induction. P. janthinellum exhibited a modified profile after hydralazine or procainamide modulation (Fig. 1B), whereas for T. minioluteus, HPLC profile was modified only by exposure to procainamide (Fig. 1C).
(i) Metabolic profile after individual epigenetic modulation SA, suberohydroxamic acid; H, hydralazine; P, procainamide. (A) T. islandicus; (B) P. janthinellum; (C) T. minioluteus; (D) T. funiculosus; (E) T. pinophilus. (ii) Metabolic profile after combined epigenetic modulation SA, suberohydroxamic acid. H, hydralazine; P, procainamide. (F) T. islandicus; (G) P. janthinellum; (H) T. minioluteus; (I) T. funiculosus; (J) T. pinophilus.
T. islandicus, T. funiculosus and T. pinophilus showed different response when submitted to epigenetic modulation with combined agents (Fig. 1F, I and J). Procainamide/hydralazine was the only combination able to promote high metabolic diversification in P. janthinellum (Fig. 1G). Inversely, treatment with suberohydroxamic acid/procainamide reduced metabolite expression under the conditions analyzed, highlighting that the balance between reactions of methylation and acetylation end up in complex results.
T. minioluteus exhibited similar metabolic profiles after exposure to both combinations of suberohydroxamic acid/hydralazine and hydralazine/procainamide (Fig. 1H). It must be pointed out that increase of metabolites production observed when the fungal species was grown in the presence of some epigenetic modulators, such as hydralazine, was resulting of the production of novel metabolites but also of preexisting metabolites. In this way, extracts that showed HPLC profiles more successfully affected by the epigenetic induction were assayed for their antimicrobial activity and acetylcholinesterase inhibition.
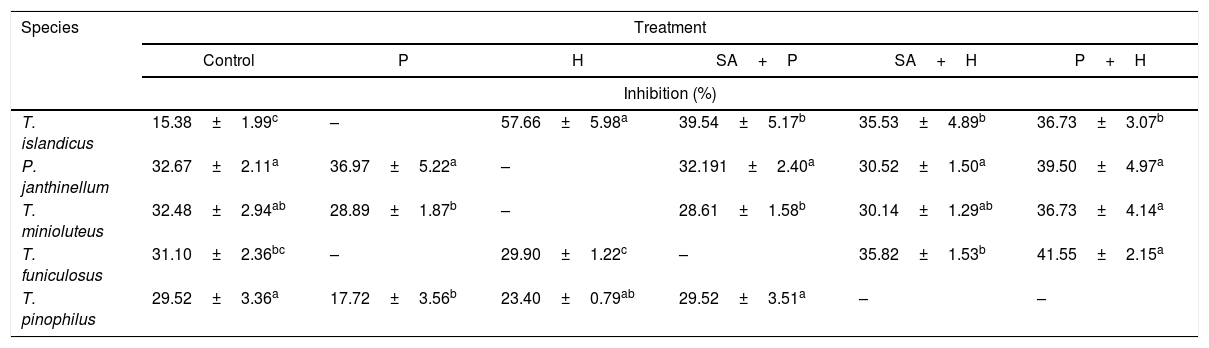
Antimicrobial activityScreening of extracts from fungi, such as of Penicillium genus, has consistently demonstrated that these microorganisms are potential sources of molecules with wide antibacterial activity.9 However, none of the extracts tested in this work were active against E. coli. The extracts were also not active against C. albicans. By contrast, extracts obtained after exposure to hydralazine or procainamide showed higher inhibition potential against L. monocytogenes growth (Table 1) when compared to the control extract (15.38±1.99%). L. monocytogenes growth inhibition was more pronounced under extract concentration equivalent to 200μgmL−1. Considering that only the extract of T. islandicus treated with hydralazine promoted an inhibition superior to 50%, it was not possible to determine the minimal inhibition concentration of the extracts. Nevertheless, antimicrobial activity potential of the extracts at the concentration of 200μgmL−1 is presented in Table 1. The most significant increases in inhibition, when comparing extracts obtained in the absence and in the presence of epigenetic modulators were observed with extracts from T. islandicus treated with hydralazine (from 15.38±1.99 to 57.66±5.98%) and T. funiculosus treated with procainamide and hydralazine (from 31.10±2.36 to 41.55±2.15%).
Effect of the extracts obtained from the treatments with SA, P and H, alone or in combination, over L. monocytogenes growth (%).
| Species | Treatment | |||||
|---|---|---|---|---|---|---|
| Control | P | H | SA+P | SA+H | P+H | |
| Inhibition (%) | ||||||
| T. islandicus | 15.38±1.99c | – | 57.66±5.98a | 39.54±5.17b | 35.53±4.89b | 36.73±3.07b |
| P. janthinellum | 32.67±2.11a | 36.97±5.22a | – | 32.191±2.40a | 30.52±1.50a | 39.50±4.97a |
| T. minioluteus | 32.48±2.94ab | 28.89±1.87b | – | 28.61±1.58b | 30.14±1.29ab | 36.73±4.14a |
| T. funiculosus | 31.10±2.36bc | – | 29.90±1.22c | – | 35.82±1.53b | 41.55±2.15a |
| T. pinophilus | 29.52±3.36a | 17.72±3.56b | 23.40±0.79ab | 29.52±3.51a | – | – |
Control, absence of epigenetic modulators; SA, suberohydroxamic acid; P, procainamide; H, hydralazine. Test control: ampicilin, 95.03%±0.59 of inhibition. –, not analyzed. Extract concentration, 200μgmL−1. Different superscripts in a row differ significantly (p<0.05).
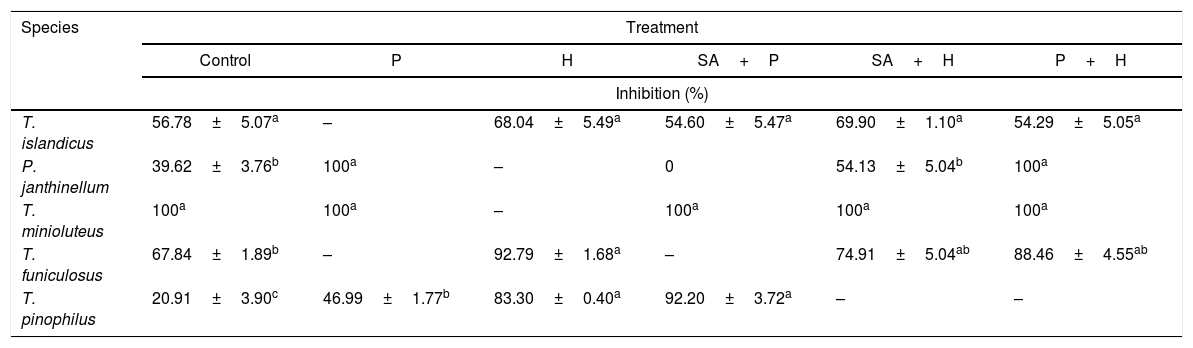
Some epigenetic modulators used in the present study were also able to increase the expression of secondary metabolites with acetylcholinesterase inhibitory activity (Table 2). Without epigenetic modulation, T. minioluteus extract was extremely active (100% of inhibition) against acetylcholinesterase activity, followed by T. funiculosus (67.84±1.89%) and T. islandicus (56.78±5.07%). On the other hand, P. janthinellum and T pinophilum extracts exhibited low acetylcholinesterase inhibitory activity, of 39.92±3.76% and 20.91±3.90%, respectively.
Acetylcholinesterase inhibition activity of the extracts prepared using epigenetic modulators (%).
| Species | Treatment | |||||
|---|---|---|---|---|---|---|
| Control | P | H | SA+P | SA+H | P+H | |
| Inhibition (%) | ||||||
| T. islandicus | 56.78±5.07a | – | 68.04±5.49a | 54.60±5.47a | 69.90±1.10a | 54.29±5.05a |
| P. janthinellum | 39.62±3.76b | 100a | – | 0 | 54.13±5.04b | 100a |
| T. minioluteus | 100a | 100a | – | 100a | 100a | 100a |
| T. funiculosus | 67.84±1.89b | – | 92.79±1.68a | – | 74.91±5.04ab | 88.46±4.55ab |
| T. pinophilus | 20.91±3.90c | 46.99±1.77b | 83.30±0.40a | 92.20±3.72a | – | – |
Control, absence of epigenetic modulators; SA, suberohydroxamic acid; P, procainamide; H, hydralazine. Test control, eserine 94.47±2.45 of inhibition. –, not analyzed. Different superscripts in a row differ significantly (p<0.05).
After epigenetic modulation, P. janthinellum extracts treated with procainamide increased significantly its acetylcholinesterase inhibition capacity, and T. minioluteus extract did not lose its total inhibitory ability. The high acetylcholinesterase inhibitory activity of T. minioluteus could be related to the pool of metabolites with retention times of 19 and 27min (Fig. 1C and H) in the HPLC analysis. After procainamide induction, T. pinophilus had a significant increase in acetylcholinesterase inhibitory activity, as well as after exposure to suberohydroxamic acid (92.20±3.72%) and the combination suberohydroxamic acid/hydralazine (83.30±0.40%).
DiscussionFilamentous fungi are eukaryotic organisms that have their genetic content organized as chromatin. Chromatin organization can be modified by epigenetic agents modulating the diversity of genes transcribed. Methylations reduce gene transcription activity while acetylations increase gene transcription28 rates (Fig. 2).
The HPLC profiles of the extracts obtained after epigenetic induction showed several effects on the metabolite production when compared to the extracts obtained without addition of inducers. When grown without epigenetic induction, T. islandicus, T. minioluteus and T. funiculosus exhibited a high basal (control) production of metabolites when compared to P. janthinellum and T. pinophilus (Fig. 1). It is well known that basal metabolism of filamentous fungi, grown under laboratorial conditions, shows a reduction of the expression of cryptic gene clusters and that epigenetics plays an important role in cryptic gene expression.29
DNMT inhibitors, hydralazine and procainamide, were generally more effective metabolite modulators compared to suberohydroxamic acid, meaning that these modulators promoted the diversification of the HPLC profile or increase of the production of basal metabolites. This may be related to modulation of CpG islands, which are methylation hotspots constituted by cytosine-guanine residues.30 Hydralazine and procainamide effectiveness must be related to the fact that promoter regions are rich in CpG islands,31 which are usually undermethylated and when DNMT inhibitors are applied the result is increasing in allowing gene expression. This is related to a structural characteristic of promoter regions, indeed. In addition to that, epigenetic modulation has a complex balance considering the rate of methylations and acetylations. According to Kondo et al.32 the different epigenetic modifications may play a synergic or an antagonist role in gene expression, which can explain the diversity of results described in the present study.
According to the acquired data, the lower susceptibility of E. coli to the extracts tested could be related to the higher resistance of Gram-negative bacteria to antibiotics due to lipopolysaccharides in their cell wall constitution.33 Zutz and collegues26 described an increase in antimicrobial activity of Aspergillus clavatus extracts after modulation with azacitidine, a DNTM inhibitor as procainamide and hydralazine. This same modulator was related to the activation of antimicrobial activity of Streptomyces coelicolor extracts against five bacterial strains.34 Recently, Byrne and colleagues35 demonstrated the occurrence of L. monocytogenes in vegetables and the resistance of some strains to tetracycline and penicillin G, important antibiotics commercialized. These finding raises the importance of the screening of new sources of antimicrobial products with potential activity toward L. monocytogenes as described in this work. Larsen and Knochel4 also demonstrated the susceptibility of pathogenic bacteria to Penicillium camemberti extracts, with a wide spectrum of activity. The inhibitory selectivity of the extracts obtained in the present study can be of interest depending on the objective of a therapeutic.
Acetylcholinesterase inhibitory activity screening also demonstrated different responses to epigenetic modulation and biological activity. For T. funiculosus, hydralazine seems to play an important role in modulation, as both hydralazine alone and combined with syberohydroxamic acid or procainamide led to high inhibitory activities (74.91±5.04% and 88.46±4.55% respectively). The greatest effect obtained by epigenetic modulation was observed for P. janthinellum extracts after exposure to procainamide alone or combined with hydralazine, reaching 100% of inhibitory activity when compared to 39.61±3.76% in control condition. Interestingly, this effect was associated with a high diversification in the production of compounds with retention times of 9 and 13min in HPLC profile. On the other hand, when cultivated with suberohydroxamic acid/hydralazine, the inhibitory activity reached only 54.13±5.04% and dropped to 0% with suberohydroxamic acid/procainamide combined. Inhibitory reduction of this extract could be related to the lower metabolite production observed after treatment with suberohydroxamic acid/procainamide. Few information is available in the literature on the use of epigenetic modulation to obtain extract of filamentous fungi inhibiting acetylcholinesterase activity, but Talaromyces sp. extracts have been reported to possess acetylcholinesterase inhibition activity.36
The present work showed the differential expression of cryptic genes of filamentous fungi by epigenetic modulation based on the comparison of metabolic profiles, suggesting that epigenetic modulation promoted the expression of cryptic genes or promoted distinct effects toward genes generally expressed under laboratorial conditions. Activation of silenced genes was more efficient with DNMTs inhibitors than with HDACs inhibitor used in this study, and the fungal species tested responded differently to each epigenetic treatment. After epigenetic modulation, some fungal extracts showed increased antibacterial activity, but only when L. monocytogenes was used as indicator strain, whereas inhibition of acetylcholinesterase activity was improved by extracts of T. islandicus, P. janthinellum and T. pinophilus. These epigenetic modifications of chromatin could be used to improve the production of bioactive compounds.
Conflicts of interestThe authors declare no conflicts of interest.
Authors thank Ms. Mirra Angelina Neres da Silva and Dr. Vany Perpétua Ferraz for technical support at the high-performance liquid chromatography laboratory at the Department of Chemistry of UFMG; and the support by grants from Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).