This study aimed to evaluate the role of chest computed tomography (CT) in complementing reverse transcription polymerase chain reaction (RT-PCR) in asymptomatic candidates for elective surgery in the context of the COVID-19 pandemic.
Material and methodsWe prospectively included 464 asymptomatic patients who underwent a triple screening workup for SARS-CoV-2 infection (health questionnaire, RT-PCR, and low-dose chest CT) during the 48 h prior to undergoing elective surgery. A positive RT-PCR and/or CT findings suggestive of COVID-19 (CO-RADS 4/5) were considered diagnostic criteria for SARS-CoV-2 infection.
ResultsMost patients (64.7%) underwent otorhinolaryngology surgery. No patients had positive RT-PCR results or symptoms suggestive of SARS-CoV-2 in the health questionnaire. Only 22 (4.7%) had signs compatible with lung infection; in 20 of these, the CT findings were atypical or indeterminate for COVID-19 (CO-RADS 2/3) and in 2 they were compatible with COVID-19 pneumonia in resolution. In the immediate postoperative period, no cases of SARS-CoV-2 infection were confirmed.
ConclusionIn our series of asymptomatic patients, low-dose CT did not add any value to the results of RT-PCR and a health questionnaire in preoperative screening for SARS-CoV-2.
El objetivo del presente estudio fue evaluar el papel de la tomografía computarizada (TC) de tórax complementaria a la prueba de la reacción en cadena de la polimerasa con transcripción inversa (RT-PCR) en pacientes asintomáticos candidatos a cirugía electiva en el contexto de la pandemia de COVID-19.
Material y métodosSe incluyeron, de forma prospectiva, 464 pacientes asintomáticos que se sometieron a una triple estrategia de cribado de infección por SARS-CoV-2 (cuestionario de salud, prueba de RT-PCR y TC torácica de baja dosis) durante las 48 horas previas a la realización de una cirugía electiva. Un resultado positivo en la prueba de RT-PCR y/o la identificación de hallazgos tomográficos sugestivos de neumonía COVID-19 (categorías CO-RADS 4 y 5) fueron considerados criterios diagnósticos de infección por SARS-CoV-2.
ResultadosLa mayor parte de los pacientes se sometieron a cirugías de otorrinolaringología (64,7%). Ningún paciente presentó un resultado positivo en la prueba de RT-PCR ni síntomas sugestivos de infección por SARS-CoV-2 en el cuestionario de salud. Únicamente 22 (4,7%) mostraron signos compatibles con infección pulmonar; 20 de ellos atípica o indeterminada para COVID-19 (CO-RADS 2, 3) y 2 compatibles con neumonía COVID-19 en resolución. Durante el postoperatorio inmediato no se confirmó ningún caso positivo para SARS-CoV-2.
ConclusiónEn nuestra serie, la realización de una TC torácica de baja dosis de radiación en pacientes asintomáticos para el cribado preoperatorio de infección por SARS-CoV-2 no proporcionó un valor diagnóstico adicional a la RT-PCR y el cuestionario de salud.
Since the first case was described in Wuhan (Hubei, China)1 in December 2019 and was declared a pandemic by the World Health Organization (WHO) in March 20202, the number of confirmed cases of coronavirus disease 2019 (COVID-19) has increased rapidly, to more than 228 million cases worldwide and more than 4.5 million deaths as of 10 August 20213.
In the first months of the pandemic, all elective surgery was delayed or cancelled, allowing to optimise resources and reduce the incidence of COVID-19. After the first wave, care activity was resumed and elective surgical interventions began to be scheduled. Given the epidemiological importance of subjects asymptomatic for SARS-CoV-2 infection in terms of the health risk to other hospitalised patients and healthcare workers4 and the possibility that they themselves may develop post-surgical respiratory complications5, was considered essential to implement measures for screening for the disease. At our centre, these measures were based on reverse transcription polymerase chain reaction (RT-PCR) testing, low-dose chest computed tomography (CT) and a health questionnaire. CT was chosen instead of chest X-ray for its greater sensitivity in detecting the disease in early stages6.
The aim of this study was to determine the role of performing a low-radiation-dose chest CT in asymptomatic patients who are candidates for elective surgery, in addition to the RT-PCR test.
Material and methodsPatient recruitment and COVID-19 infection screening protocolDuring the period from 20 April 2020 to 15 March 2021, we prospectively included 464 asymptomatic subjects admitted to our centre for elective surgery. Following the policy of our institution to ensure the safety of patients and hospital staff, an initial screening of patients was carried out based on a telephone questionnaire about their health status, to prevent admissions of infected people and unnecessary travel to our hospital. Once admitted, the patients had a triple screening strategy applied for SARS-CoV-2 infection, which consisted of an RT-PCR test, a further health questionnaire, and a low-radiation-dose chest CT performed in the 24−48 h prior to the elective surgery. The RT-PCR test was obtained by nasopharyngeal swab. The health questionnaire included questions about symptoms suggestive of infection, such as fever, shortness of breath, dry cough, myalgia, anosmia, dysgeusia and diarrhoea, as well as possible exposure to or contact with people with COVID-19. Analysing the results, those who had any positive result in the tests carried out would be excluded from surgical treatment.
The CT scans were performed with multidetector CT equipment (SOMATOM Force and SOMATOM X.cite; Siemens Healthineers, Forchheim, Germany) using low radiation dose protocols (100 or 150 kV and tin filter, with application of an anthropomorphic tube current modulation system, CareDose 4 D). The images were reconstructed with a slice thickness of 1 mm and soft tissue filters (Br32) and high resolution (Bl60) to assess the mediastinum and lung parenchyma respectively, and were stored in Picture Archiving and Communication Systems (PACS) to be studied.
By protocol, during the immediate postoperative period (72 h after the intervention), it was decided not to repeat the RT-PCR test on all patients, but only on those who developed symptoms suggestive of COVID-19.
The hospital's ethics committee approved the study protocol, and informed consent was obtained from all patients.
Imaging analysisTwo radiologists independently analysed the chest CT scans (a radiology resident and a chest radiologist with two and 16 years of experience respectively), who did not know the results of the RT-PCR and the clinical questionnaire. The following findings were considered suggestive of COVID-19 pneumonia7: peripherally distributed ground-glass opacities, cobblestone pattern, or bilaterally distributed consolidation; round/pseudonodular opacities; subpleural bands and radiological pattern similar to that of organising pneumonia. Following the COVID-19 Reporting and Data System (CO-RADS) standardised classification, six categories were determined that indicated the likelihood of COVID-19 pneumonia based on the radiological findings8: 1, very low; 2, low; 3, indeterminate; 4, high; 5, very high; and 6, demonstrated by PCR. In the event of discrepancy between the observers, a consensus was reached on the conclusion.
A positive result in the RT-PCR test or the presence of tomographic findings suggestive of COVID-19 pneumonia (CO-RADS categories 4 and 5) were considered diagnostic criteria for SARS-CoV-2 infection and an indication for delaying the surgical procedure.
Statistical analysisThe characteristics of the patients and surgical procedures performed are described in percentages for qualitative variables, and are expressed as mean ± standard deviation or as median and interquartile range (IQR) for quantitative variables. The results of the health questionnaire, RT-PCR and CT were considered categorical variables (negative/positive) and are expressed as proportions and percentages.
The validity of the diagnostic tests was evaluated, considering the RT-PCR as the reference standard for the diagnosis of the infection. A true positive was considered when radiological findings suggestive of COVID-19 occurred in a patient with a positive microbiological test for the infection. The results were considered false negative if there were no radiological findings suggestive of COVID-19 pneumonia on CT, and the patient had a positive RT-PCR. A false positive was considered if the radiological findings suggest COVID-19 pneumonia, but the RT-PCR was negative. Lastly, patients without radiological findings suggestive of infection and with negative RT-PCR were considered true negatives.
The statistical analysis was performed with the software IBM SPSS Statistics for Mac, version 25.0 (IBM Corp., Armonk, N.Y., USA). A two-tailed p value of less than 0.05 was considered statistically significant.
ResultsTable 1 shows the demographic characteristics of the study participants and the main findings. The mean age was 54 ± 12 years and 57.1% were male. Most of the patients were from the Ear, Nose and Throat (64.7%), General Surgery (9.6%) and Orthopaedic Trauma (8.6%) departments.
Demographic characteristics of the study with the main and secondary findings.a
| ENT | General Surgery | Orthopaedic Trauma | Thoracic Surgery | Other | Total | |
|---|---|---|---|---|---|---|
| No. of patients | 300 (64.7%) | 45 (9.6%) | 40 (8.6%) | 30 (6.5%) | 49 (10.5%) | 464 |
| Gender | ||||||
| Male | 159 (34%) | 30 (6.5%) | 28 (6%) | 21 (4.5%) | 27 (5.8%) | 265 (57.1%) |
| Female | 141 (30%) | 13 (2.8%) | 12 (2.6%) | 11 (2.4%) | 22 (4.7%) | 199 (42.9%) |
| Age | ||||||
| Median (IQR) | 52 (43–61) | 61 (54–69) | 59 (48–67) | 67 (62–72) | 56 (49–63) | 54 (47–62) |
| PCR + | 0 | 0 | 0 | 0 | 0 | 0 |
| Lung opacities on CT | ||||||
| CO-RADS 1-3 | 13 (2.8%) | 1 (0.2%) | 3 (0.6%) | 1 (0.2%) | 2 (0.4%) | 20 (4.3%) |
| CO-RADS 4-6 | 0 | 1 (0.2%) | 0 | 0 | 1 (0.2%) | 2 (0.4%) |
| Incidentals findings-CT | ||||||
| Total | 13 (2.8%) | 4 (0.9%) | 3 (0.6%) | 9 (1.9%) | 12 (2.6%) | 41 (8.8%) |
| Lung nodules | 10 (2.1%) | 1 (0.2%) | 2 (0.4%) | 9 (1.9%) | 5 (1.1%) | 27 (5.8%) |
| Neoplasms | 0 | 1 (0.2%) | 0 | 0 | 0 | 1 (0.2%) |
| Coronary calciumb | 3 (0.6%) | 2 (0.4%) | 1 (0.2%) | 0 | 7 (1.6%) | 13 (2.8%) |
None of the patients had a positive result in the RT-PCR test performed 48 h before the procedure (Fig. 1). In the health questionnaire, they reported no symptoms suggestive of a COVID-19 infection, nor close contact with individuals with confirmed SARS-CoV-2 infection in the weeks prior to the intervention.
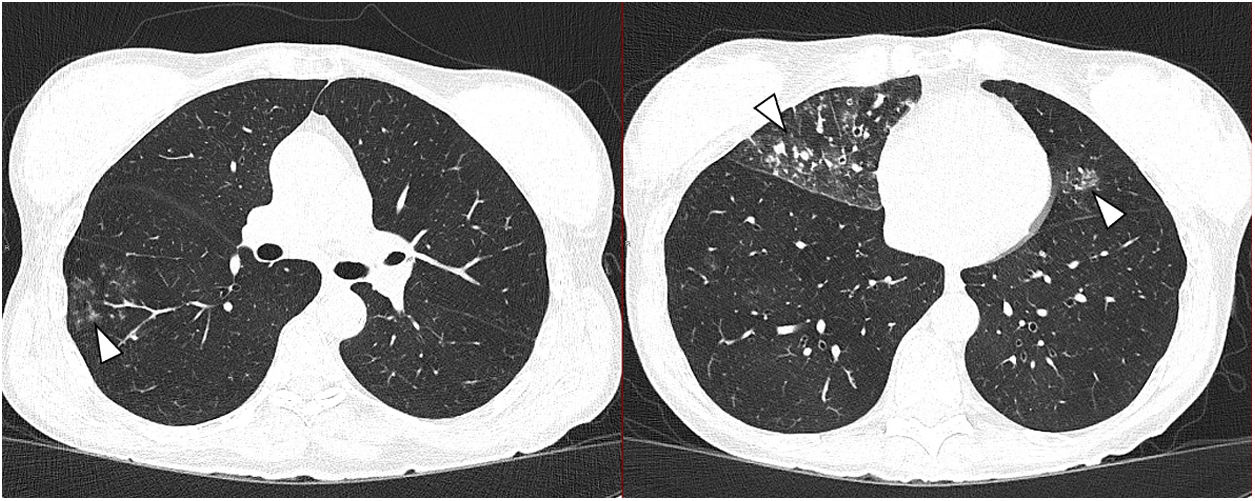
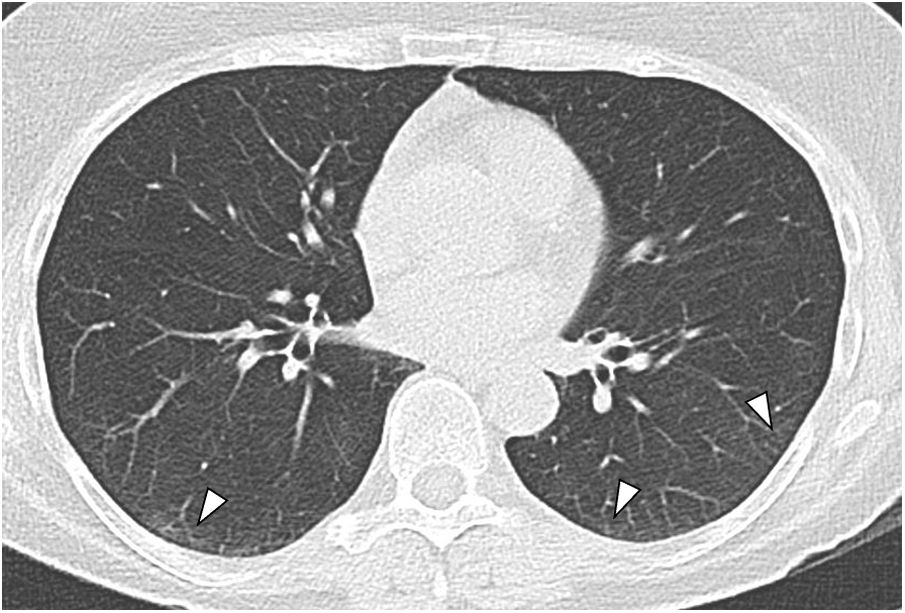
Of the 464 patients studied, 20 (4.3%) had findings consistent with lung infection suggestive of an atypical bronchiolar inflammatory or pneumonic process (CO-RADS 2, Fig. 2) or indeterminate for COVID-19 (CO-RADS 3). Two patients had peripheral ground-glass lung opacities which were interpreted as resolving COVID-19, in view of the history of recent infection in these patients (Fig. 3). Both had had a positive RT-PCR two weeks before the intervention. Serological testing subsequently confirmed seroconversion in these patients.
Axial low-dose chest computed tomography (CT) image. Parenchymal window. Peripheral micronodules, bronchiolar distribution, suggestive of inflammatory disease of the small airway, with the presence of bronchiectasis. Mucus plugs and bronchiolar involvement can be seen in the middle lobe, right lower lobe and lingula (arrowheads). The radiological findings in this 64-year-old patient's chest CT are suggestive of an infectious process in the lungs other than COVID-19.
Incidental findings included pulmonary nodules (27 patients; 5.8%). One person was found to have primary lung cancer (0.2%). Moderate or severe coronary calcification was found in 3% of the patients (Table 1).
None of the participants developed signs or symptoms suggestive of COVID-19, and no new RT-PCR was obtained with a positive result during the immediate postoperative period.
DiscussionIn our study, performing a chest CT as part of screening for SARS-CoV-2 infection in asymptomatic patients who were candidates for elective surgery did not provide any diagnostic value additional to the RT-PCR.
Asymptomatic or presymptomatic patients with SARS-CoV-2 infection may contribute to the transmission and nosocomial spread of the infection, particularly if they undergo surgical interventional procedures. In addition, it has been reported that up to 44% of patients with COVID-19 who undergo surgery require intensive care and that 51.2% suffer from postoperative pulmonary complications. In these patients, the 30-day postoperative mortality rate is as high as 38%9. From this it follows that in order to implement measures that guarantee the safety of surgical procedures, it is essential to screen for SARS-CoV-2 infection. Such screening usually includes performing the RT-PCR test. However, the test can be negative during the window period of SARS-CoV-2 infection (which ranges from 1-5 days) and can show false negative results, even at the time of onset of symptoms (median false negative around 38%)10. In that context, at the beginning of the pandemic, it was quickly found that chest CT could play an important role in diagnosing COVID-19 pneumonia, especially in the first phase of infection, when RT-PCR can be negative11.
During the first months of the pandemic, different algorithms were published that supported the use of chest CT as a diagnostic and management tool for patients with COVID-19 prior to surgical procedures. For example, in March 2020, the British Society for Thoracic Imaging (BSTI) and the British Society for Gastrointestinal and Abdominal Radiology (BSGAR) recommended chest CT for patients undergoing urgent abdominal surgery12. Other working groups, such as the Italian, proposed a preoperative screening for elective surgery, aimed at identifying the greatest number of asymptomatic or presymptomatic patients, as well as patients who might be positive during their stay in hospital. They recommended a detailed history of contacts five days before admission, performing RT-PCR, laboratory tests (including C-reactive protein [CRP], D-dimer, blood count and ferritin) and chest X-ray two days before admission, and repeat RT-PCR test 12–14 days after discharge13. Along the same lines, the Asociación Española de Cirujanos (AEC) [Spanish Association of Surgeons] ruled that COVID-19 status should be determined in all surgical patients, regardless of whether they had symptoms, by means of RT-PCR for SARS-CoV-2 and further investigations, such as chest X-ray, CT or abdominal ultrasound (POCUS, Point-of-Care Ultrasound). Chest X-ray was recommended in all patients, recommending chest CT in cases of extreme urgency (when delaying the intervention was not possible) with clinical suspicion of SARS-CoV-2 infection14.
More recently, in a multicentre study, Callaway et al. found a low sensitivity (68.4%) of chest CT for identifying COVID-19 preoperatively, and therefore concluded it was difficult to justify performing such an examination in this group of patients15. We found no positive cases for COVID-19 in our study. We believe this is largely due to the measures adopted at our hospital during the pandemic, which established initial screening of patients based on a telephone questionnaire about their health status to prevent admission of people infected. In the CT scans performed, only two patients showed signs of SARS-CoV-2 pneumonia, but the findings, the medical history and the RT-PCR result confirmed that the process was in resolution. The cases of active infection observed on CT (4.7%) showed signs of lung infection due to other germs or undetermined for COVID-19. In our group of patients, performing a chest CT as part of screening for SARS-CoV-2 infection in asymptomatic patients who were candidates for elective surgery did not provide any value additional to the RT-PCR. This finding is in line with the results from other working groups16, which underline that isolation, clinical assessment and RT-PCR are effective measures to minimise the surgical risk related to COVID-19 and that adding a CT does not help any. In fact, the most recent recommendations made by different organisations and scientific societies, such as the Centers for Disease Control (CDC)17, the Society of Thoracic Radiology (STR)18 and the update of the Royal College of Surgeons of England's preoperative guidelines19, support performing RT-PCR in conjunction with a health questionnaire as the only preoperative screening tools in urgent and elective surgery.
The main limitation of our work is the absence of a positive result from RT-PCR or chest CT prior to the intervention. The low incidence of SARS-CoV-2 at the start of the study (36 cases in Navarra on 20 April 2020 and 1137 in Spain) probably meant the pre-test likelihood of being positive for COVID-19 was low. In addition, as these were asymptomatic patients with a negative first telephone screening and a negative health questionnaire on admission, the likelihood of finding a positive result at the points of maximum incidence (39,046 on 18 January 2021 in Spain and 1750 on 26 October 2020 in Navarra20,21) could be lower than in the general population. Therefore, the lack of a positive RT-PCR or chest CT prior to the intervention meant we were unable to establish the utility and real diagnostic validity of the test. However, during the immediate postoperative follow-up, no positive cases for SARS-CoV-2 were confirmed, so it seems unlikely that we had any false negative results. Samples from patients in a different epidemiological environment might produce different results, giving chest CT a greater degree of utility.
In conclusion, in our population of patients who were candidates for elective surgery, performing a low-dose radiation chest CT did not provide additional value for the detection of COVID-19 in asymptomatic patients. Our view is that preoperative screening through a health questionnaire and an RT-PCR test guarantees a safe surgical procedure, without the need for imaging tests.
Authorship- 1
Responsible for study integrity: GB and AEC.
- 2
Study conception: GB, MBP, AEC, JLL and JPV.
- 3
Study design: GB, MBP, AEC, JLL and JPV.
- 4
Data collection: AEC, GB, JLL and JPV.
- 5
Data analysis and interpretation: GB, GB and CU.
- 6
Statistical processing: AEC, MBP and CUI.
- 7
Literature search: MBP, AEC, CUI, JLL and JPV.
- 8
Drafting of the article: AEC, GB and CUI.
- 9
Critical review of the manuscript with intellectually significant contributions: GB and AEP.
- 10
Approval of the final version: MBP, AEC, CUI, JLL, JPV and GB.
The authors listed above attest that they have contributed substantially to: 1) the conception, the design, data collection, or the analysis and interpretation of data; 2) writing of the article or critically reviewing it for important content; and 3) final approval of the version to be submitted for review. All authors have had full access to the study data and accept responsibility for submitting this paper.
Conflicts of interestThe authors declare that they have no conflicts of interest.
The study was approved by the research ethics committee of Clínica Universidad de Navarra.
To the nursing staff, technicians and auxiliaries in the CT area of Clínica Universidad de Navarra for their efforts and dedication during the pandemic.
Please cite this article as: Barrio Piqueras M, Ezponda Casajús A, Urtasun Iriarte C, Larrache Latasa J, Pueyo Villoslada J, Bastarrika G. TC torácica de baja dosis de radiación como técnica de cribado preoperatorio deinfección por SARS-CoV-2. Radiología. 2022;64:317–323.