Outcomes vary widely in patients with COVID-19. Whereas some patients have only mild symptoms of short duration, others develop severe disease that leads to acute respiratory distress syndrome requiring prolonged stays in intensive care units. Radiologically, the initial stage is characterized by viral pneumonia with mild expression. In some patients, however, the onset of the immune response results in acute lung damage with organizing pneumonia and diffuse alveolar damage.
Moderate-severe disease is associated with a high incidence of pulmonary embolisms, generally peripherally distributed and associated with endothelial damage, prolonged stays in bed, and coagulopathy. Other relatively common complications are spontaneous pneumothorax and pneumomediastinum due to the rupture of alveolar walls and barotrauma in mechanically ventilated patients. Superinfection, generally bacterial and less commonly fungal, is more common in patients with severe disease.
Los pacientes con COVID-19 presentan una evolución muy variable: desde enfermos con síntomas leves de corta duración a pacientes con enfermedad grave que desarrollan un síndrome de distrés respiratorio agudo, con ingresos prolongados en unidades de críticos. Desde el punto de vista radiológico, la etapa inicial se caracteriza por una neumonía viral poco expresiva. No obstante, en algunos pacientes, con el inicio de la respuesta inmunitaria se produce un daño pulmonar agudo con patrones radiológicos de neumonía organizada y daño alveolar difuso.
La enfermedad moderada-grave se asocia con una incidencia alta de tromboembolismo pulmonar, generalmente de distribución periférica y asociado al daño endotelial, encamamiento prolongado y coagulopatía de la enfermedad. Otras complicaciones relativamente frecuentes son: el neumotórax y el neumomediastino espontáneos por rotura de paredes alveolares, y el barotrauma en pacientes con ventilación mecánica. La sobreinfección es más frecuente en pacientes graves, generalmente de origen bacteriano y menos frecuente fúngico.
The clinical spectrum of patients infected by SARS-CoV-2 ranges from asymptomatic subjects with mild signs and symptoms to patients with severe hypoxaemia and characteristic pulmonary infiltrates that can progress to acute respiratory distress syndrome (ARDS). Most people with COVID-19 with a mild clinical presentation do not initially require hospitalisation, and many patients will be able to manage their illness at home.1
Patients with COVID-19 and severe illness will require hospitalisation, with supportive treatment for the most common complications of severe COVID-19: hypoxaemic respiratory failure, acute kidney injury and complications from prolonged hospitalisation, such as gastrointestinal bleeding and critical illness polyneuropathy and myopathy. Specifically, we will discuss the most common thoracic complications, such as pulmonary thromboembolism, pneumothorax, pneumomediastinum, secondary infections, barotrauma and myopericarditis.2
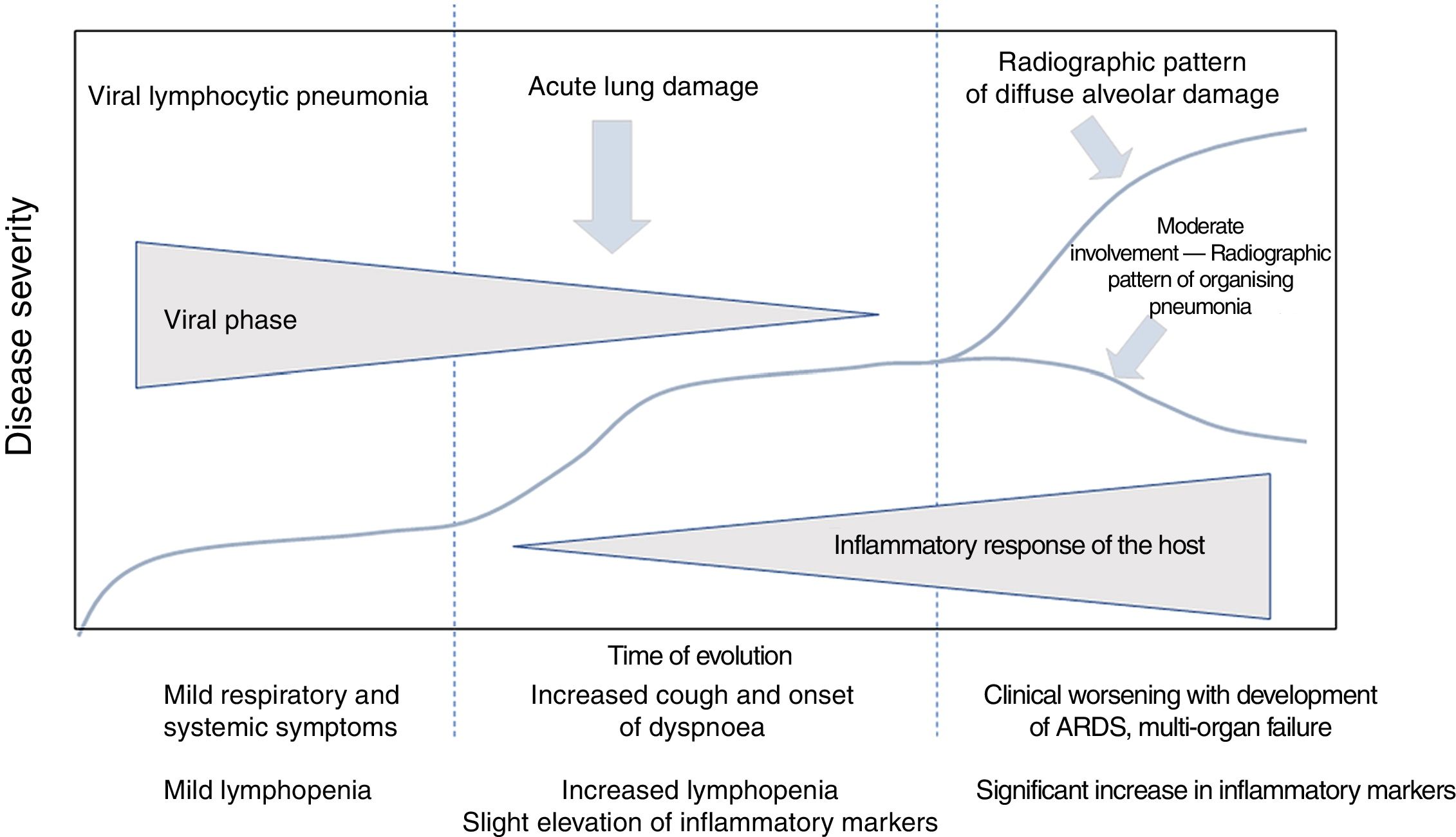
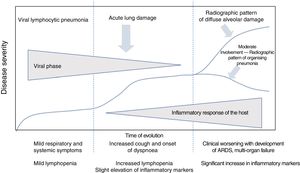
Clinical course of patients with SARS-CoV-2 infection. Laboratory parametersIn the clinical and laboratory course of patients with COVID-19, there are reportedly three disease phases, overlapping with each other and conditioned by two different pathological substrates (Fig. 1).3
Clinical course of patients with COVID-19. The initial phase is characterised by viral lymphocytic pneumonia with mild symptoms. Patients with moderate disease present a radiographic pattern of organising pneumonia on imaging tests. Patients who develop severe disease present respiratory distress syndrome and a pattern of diffuse alveolar damage on imaging tests. Adapted from Siddiqi et al.3
In the initial phase, early infection, clinical signs are secondary to the virus itself, with mild systemic and respiratory symptoms. During this period, the virus replicates and binds to angiotensin-converting enzyme II (ACE-II) receptors,4 which are more abundant in the pulmonary epithelium and vascular endothelium. There is usually mild lymphopenia, without other laboratory abnormalities. Most patients present only this phase of the disease,3 and this accounts for the fact that around 80% of cases of infection present mild symptoms.5
In some cases, the initial phase is followed by an intermediate phase, in which the host's immune response begins. Lung inflammation starts and viral pneumonia develops with cough and dyspnoea. The mean time from the first symptoms to the onset of dyspnoea is 5–8 days.6,7 Hospital admission usually occurs in this phase, and is required in 14% of patients.8 The mean time between the onset of symptoms and hospitalisation is 7 days.6
The most important risk factor for hospital admission is age over 65. Other risk factors are cardiovascular diseases, male sex, obesity, diabetes and chronic kidney disease.8,9
During this intermediate phase, an increase in lymphopenia is identified,3 with a lymphocyte count lower than 1,500/μl in 90% of patients.10 Other common laboratory findings are thrombocytopenia, prolonged prothrombin time, increased liver enzymes and slightly elevated systemic inflammatory markers,3 with increased lactate dehydrogenase enzyme, ferritin, C-reactive protein and erythrocyte sedimentation rate.6,10
The late or severe phase is characterised by systemic hyperinflammation syndrome,3,7 with marked pulmonary involvement and a poor prognosis. Up to 26% of admitted patients may require intensive care unit admission and invasive mechanical ventilation.6,8,9,11 Among these cases, 5% present critical illness, with ARDS, shock or multi-organ dysfunction.12
ARDS is the major complication in patients with severe disease11 and the main cause of intensive care unit admission in 61% of cases.6 In this phase, systemic inflammation markers, including proinflammatory cytokines, C-reactive protein, ferritin and D-dimer, are very high.3,7 Elevated troponin may also be seen.9 A gradual decrease in lymphocyte count, a gradual increase in D-dimer and deterioration of renal function are associated with higher mortality.6 Patients who require invasive mechanical ventilation have a high mortality rate, between 60% and 88%.9,11
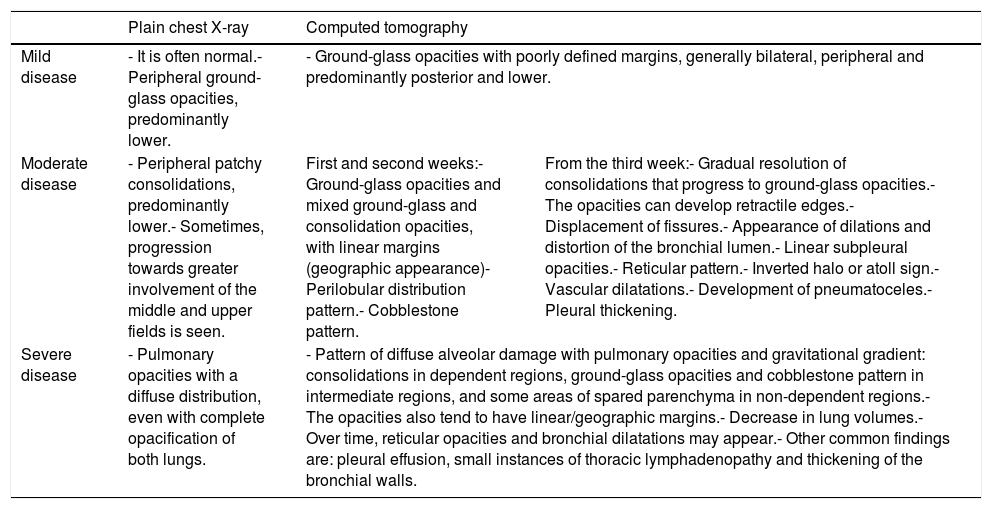
Radiological course of patients with SARS-CoV-2 infectionThe radiological course of patients with COVID-19 can be divided into three phases/degrees of involvement (Table 1).
Main radiological findings by severity of COVID-19 disease.
| Plain chest X-ray | Computed tomography | ||
|---|---|---|---|
| Mild disease | - It is often normal.- Peripheral ground-glass opacities, predominantly lower. | - Ground-glass opacities with poorly defined margins, generally bilateral, peripheral and predominantly posterior and lower. | |
| Moderate disease | - Peripheral patchy consolidations, predominantly lower.- Sometimes, progression towards greater involvement of the middle and upper fields is seen. | First and second weeks:- Ground-glass opacities and mixed ground-glass and consolidation opacities, with linear margins (geographic appearance)- Perilobular distribution pattern.- Cobblestone pattern. | From the third week:- Gradual resolution of consolidations that progress to ground-glass opacities.- The opacities can develop retractile edges.- Displacement of fissures.- Appearance of dilations and distortion of the bronchial lumen.- Linear subpleural opacities.- Reticular pattern.- Inverted halo or atoll sign.- Vascular dilatations.- Development of pneumatoceles.- Pleural thickening. |
| Severe disease | - Pulmonary opacities with a diffuse distribution, even with complete opacification of both lungs. | - Pattern of diffuse alveolar damage with pulmonary opacities and gravitational gradient: consolidations in dependent regions, ground-glass opacities and cobblestone pattern in intermediate regions, and some areas of spared parenchyma in non-dependent regions.- The opacities also tend to have linear/geographic margins.- Decrease in lung volumes.- Over time, reticular opacities and bronchial dilatations may appear.- Other common findings are: pleural effusion, small instances of thoracic lymphadenopathy and thickening of the bronchial walls. | |
Source: Dr. G. Buitrago Weiland.
In the first few days, findings correspond to viral lymphocytic pneumonia.13 Later, a pro-inflammatory state comes into play with production of cytokines, which cause acute lung damage along with viral damage.14 The lung response to the acute damage is limited and translates to identical histological and radiological patterns, regardless of the cause, which include organising pneumonia and diffuse alveolar damage.15 Findings in patients with moderate to severe involvement probably correspond to the response to lung damage,16 with radiographic patterns of organising pneumonia17,18 and diffuse alveolar damage (radiographic pattern of ARDS).18 This is consistent with the autopsy results of deceased COVID-19 patients, in which the predominant histological pattern is diffuse alveolar damage,19,20 associated with foci of organising pneumonia and acute fibrinous and organising pneumonia.19
First few days of the illness and mild involvementA chest X-ray often shows no abnormalities. This may be due to the limited extent of the involvement and because the findings can be too subtle to be detected, especially in portable examinations.18 When the initial X-ray is positive, the most common finding consists of “ground-glass” opacities18 of peripheral distribution, predominantly in lower fields.18,21
Computed tomography (CT) scans performed early in the disease may be normal. No findings are reported in 50% of patients evaluated by CT in the first two days.16 This percentage decreases to 14%–21% if patients with symptoms for less than 5 days are included.22–24
The predominant finding on CT in the first week corresponds to ground-glass opacities,22–27 generally bilateral.5,14,22,25,26,28–30 In the early phase, involvement may be unilateral more commonly than in late phases.22 The distribution is predominantly peripheral and subpleural,5,22,24,26,28–30 with greater involvement of the posterior region of the lower lobes.14,16,25,31
Ground-glass opacities initially show poorly defined margins and probably represent inflammatory exudates5 in the context of viral lymphocytic pneumonia, as has been demonstrated in autopsies performed in the first 5 days of the disease.13
Patients with mild involvement tend to show a lesser extent of ground-glass opacities on CT compared to patients with moderate to severe disease.24 The greatest degree of pulmonary involvement is reached in the first three days and gradually decreases, with near-complete resolution after 15 days.24
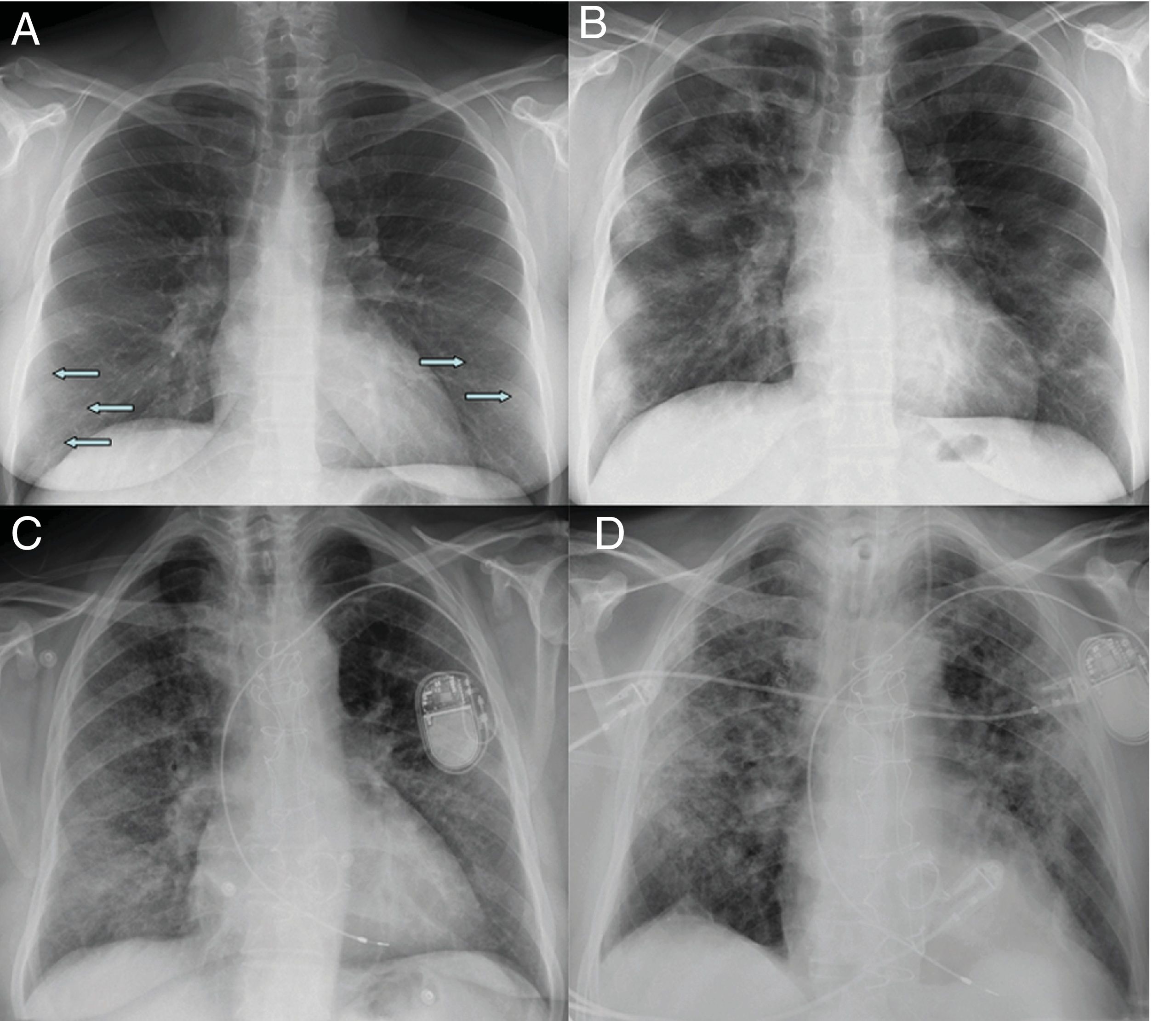
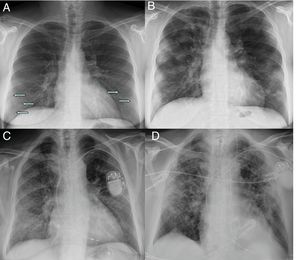
Moderate involvement. Radiographic pattern of organising pneumoniaAs the disease progresses, the opacities tend to increase, cluster and show greater density on chest X-ray, progressing to patchy consolidations, with a peak 10–12 days after the onset of symptoms.18,21 Some cases show progression towards greater involvement of the middle and upper lung fields18 (Fig. 2).
Initial X-ray (A) and X-ray at 12 days (B) in a patient with COVID-19 and moderate disease. The initial X-ray shows subtle peripheral ground-glass opacities in the lower lung fields (arrows). The X-ray at 12 days shows predominantly peripheral multifocal consolidations. Initial X-ray (C) and X-ray at 20 days (D) of another patient with COVID-19 and moderate disease, in which ground-glass opacities initially seen at the base and in the peripheral region of the middle and upper fields progress to a gross alveolar interstitial pattern with a predominance in the peripheral region of the middle and upper fields.
It is rare to find patients without findings on CT, and as of the sixth day, only 1% of the studies are normal.22 The extent of the ground-glass opacities increases.27 They are bilateral in up to 90% of cases after the first week,16,26 and almost always involve multiple lobes.5,16,23,25,29,30
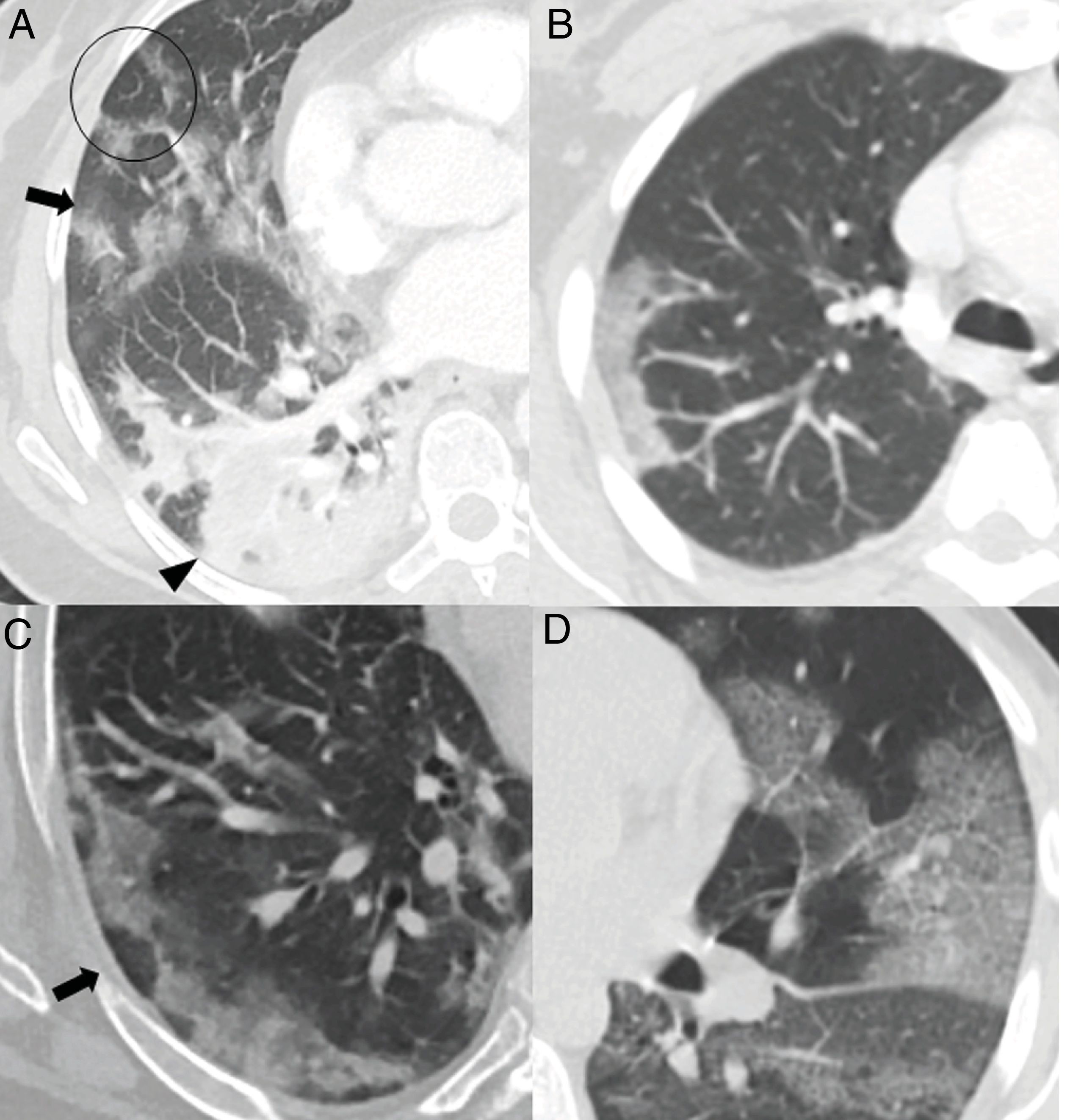
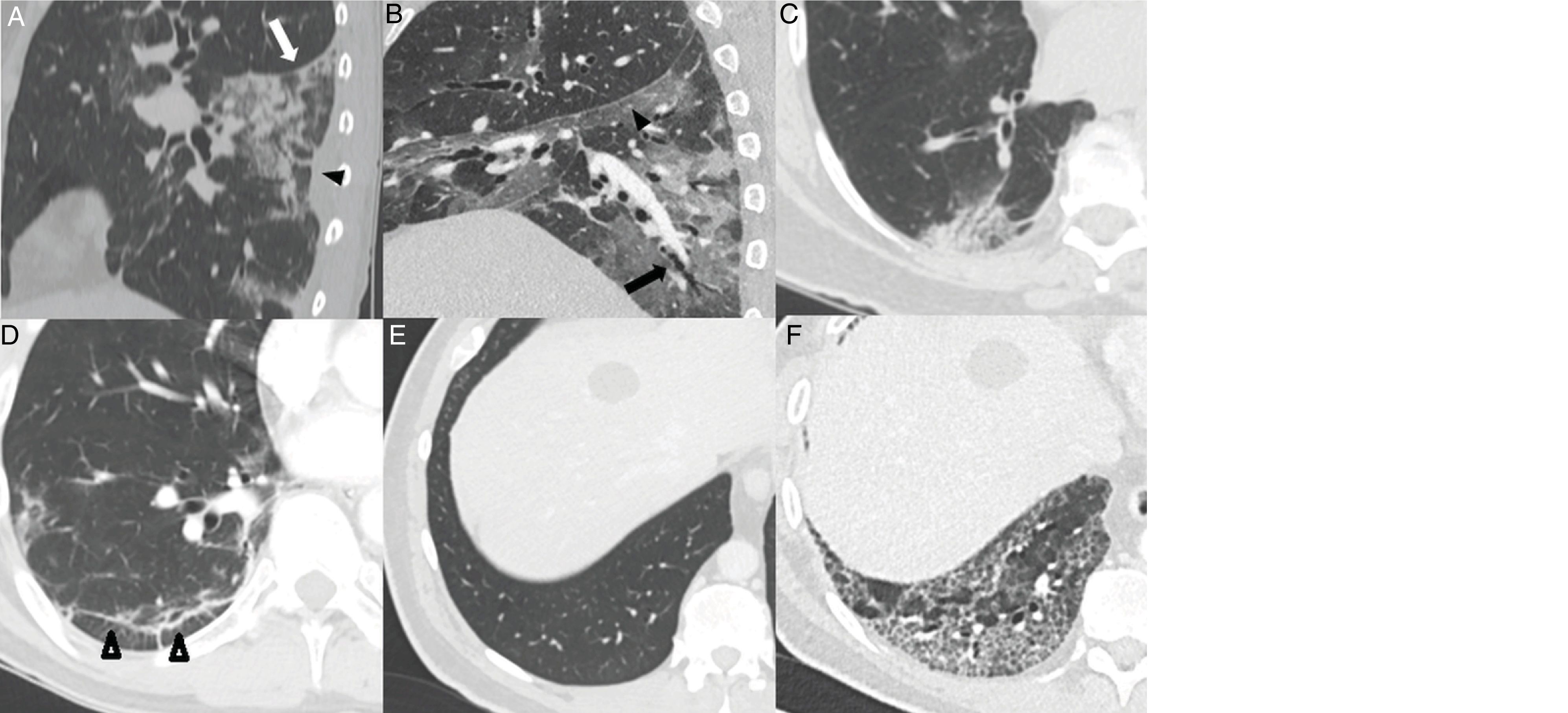
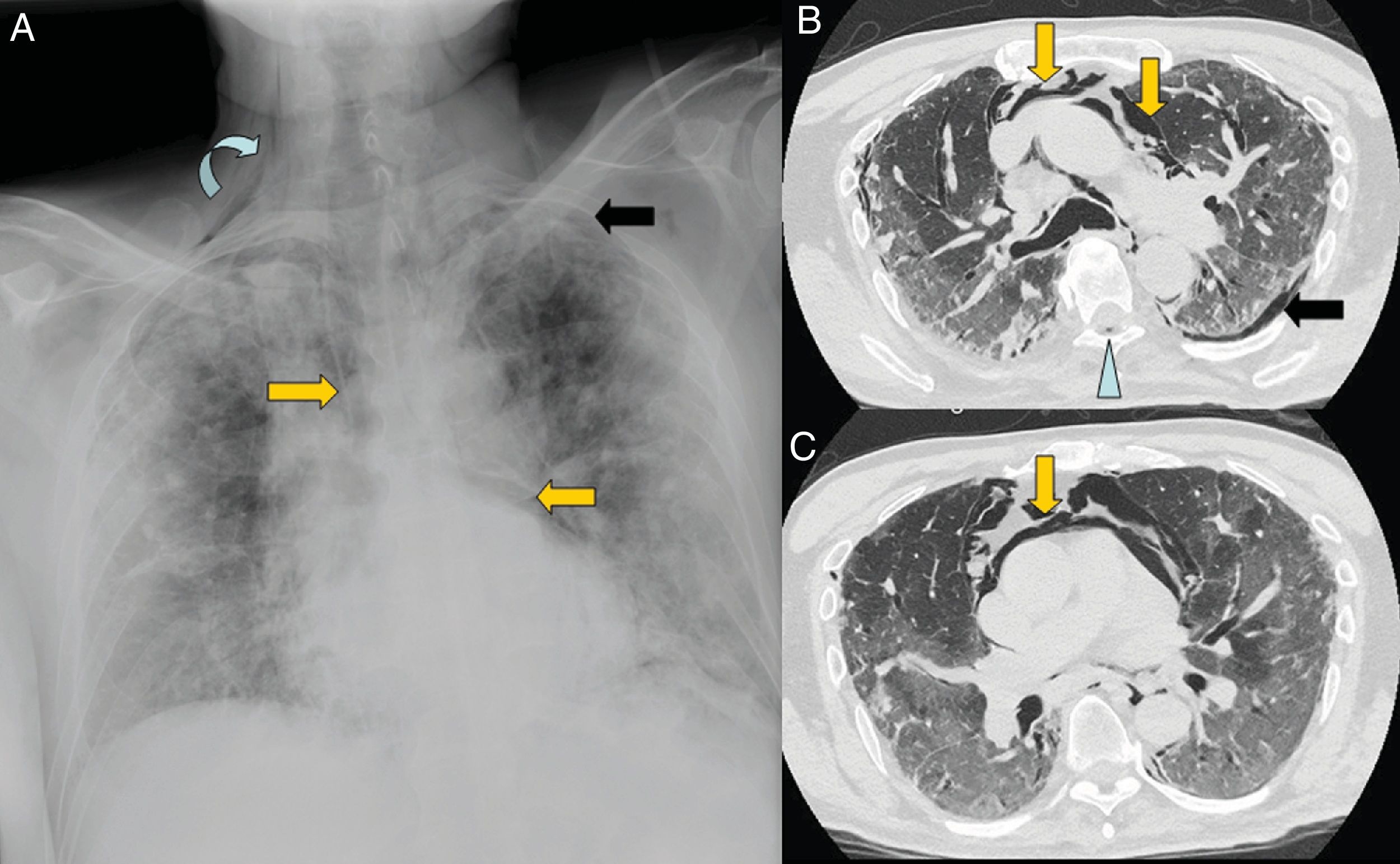
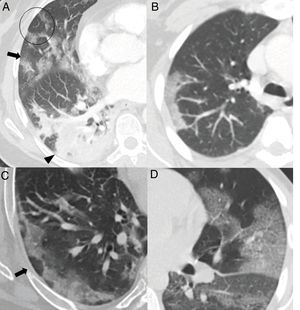
The greatest degree of lung involvement usually occurs around the tenth day22,25,27,30 or second week,24,26,28 when a gradual increase in consolidations is seen,23,25–28,30 especially in patients over 50 years of age.32 Consolidations are almost always associated with ground-glass opacities; isolated consolidations are very rare.31 At this stage, ground-glass consolidations and opacities usually show linear margins that take on a geographical appearance5 (Fig. 3A and B). This finding is typical of the radiographic pattern of organising pneumonia.5,15 Ground-glass opacities and consolidations also often present a subpleural perilobular distribution pattern, with curvilinear and polygonal opacities surrounding a secondary pulmonary lobule15,33 (Fig. 3A and C). This finding also suggests secondary organising pneumonia.15,22,34
Common findings in the first two weeks of the disease. A) Mixed ground-glass (arrow) and consolidation (arrow tip) opacities, predominantly posterior, with linear margins showing a geographic pattern. Some perilobular opacities (circle) are also observed. B) Peripheral consolidation opacity with linear margins. C) Peripheral perilobular opacities (arrow). D) Cobblestone pattern, with ground-glass opacities and overlapping reticular pattern secondary to thickening of the interlobular and intralobular septa.
The cobblestone pattern is reported in highly variable proportions, between 15% and 77% of patients,5,16,28,29 with a peak during the second week28,30 and a decrease during the third week.25 It is uncommon in later stages28 (Fig. 3D).
In the third week, gradual resolution of consolidations is seen, with progression again to ground-glass opacities.22,23,25,27,28 In this stage, opacities can develop retractile concave edges in 18% of cases. It is interpreted as an organisational and reparative process of inflammatory changes.5
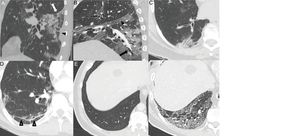
Displacement of fissures28 (24%) and bronchial dilatations can be seen in highly variable proportions (5%–45%) 5,23,26,28–30 (Fig. 4A,B and C). The natural history of the infection has not been studied, and it is premature to deem these changes irreversible.28
Typical findings in the third and fourth weeks of the disease. A) Consolidation focus with architectural distortion causing retraction of the fissure (white arrow) and the adjacent pleura (black arrow tip). B) Ground-glass and consolidation opacities associated with bronchiectasis (arrow) and pleural thickening of the oblique fissure (arrow tip). C) Subpleural consolidation focus with associated bronchial dilatations. D) Subpleural line parallel to the pleura, associated with architectural distortion (arrow tips). Patient with a previously healthy lung parenchyma (E) having developed an extensive reticular pattern secondary to thickening of the intralobular septa with some associated bronchial dilation (F).
Ground-glass opacities with linear subpleural opacities and the parallel pleura sign are usually observed late, around the third or fourth week14,23,25,27,30 in 60%–75% of cases5,28 (Fig. 4D). They may represent subsegmental atelectasis or secondary organising pneumonia.22,34 A reticular pattern can also develop late,14,26,27 and is seen in 6% of patients, after the fourth week of the disease22 (Fig. 4E).
Other late findings are:
- •
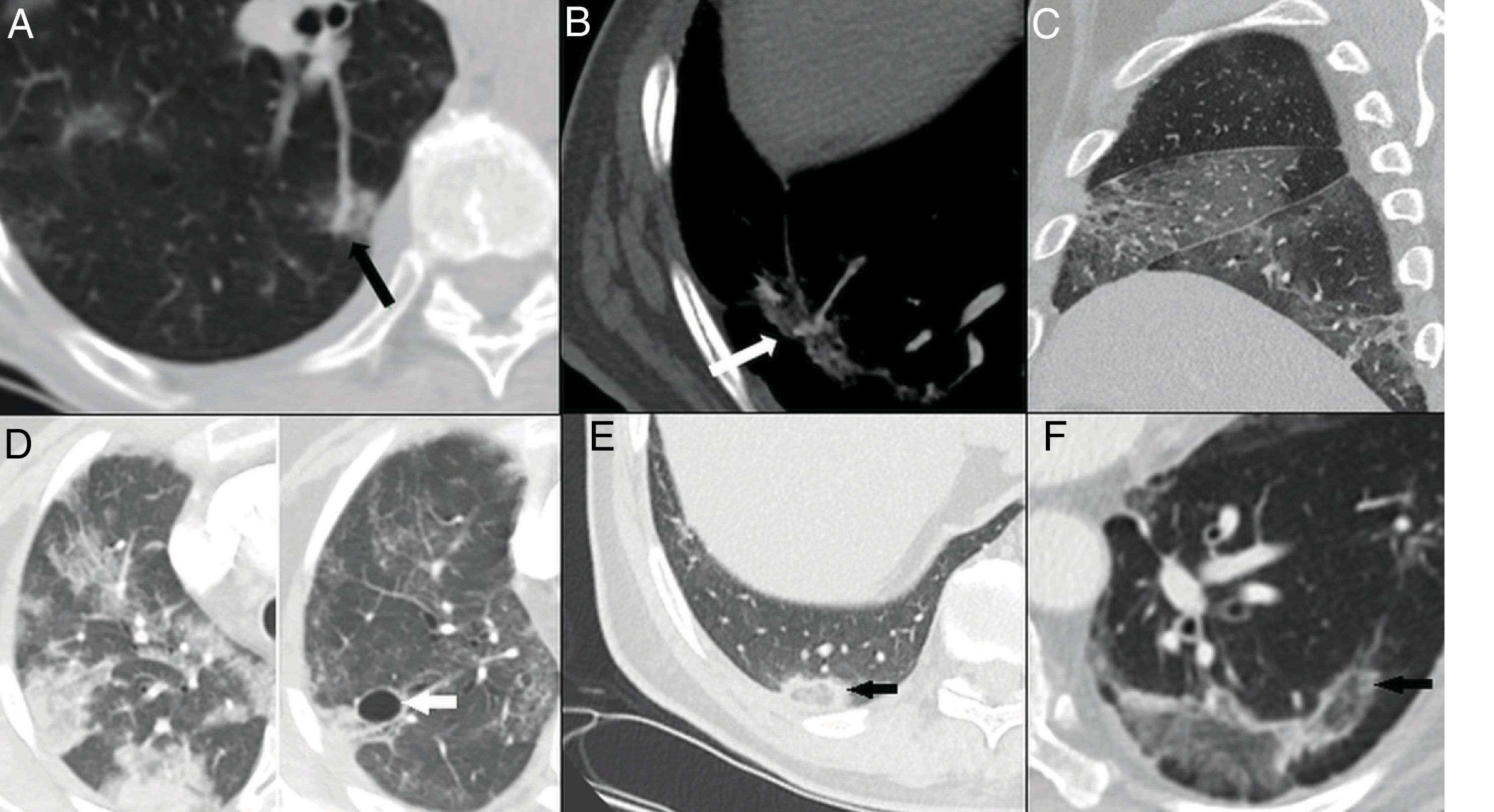
The inverted halo or atoll sign, seen in 4%–5% of cases.5,16 It is a finding that suggests the presence of organising pneumonia,15,34 although it should also raise suspicion of the possibility of pulmonary infarctions18 (Fig. 5E and F).
Figure 5.Late findings. A and B) Vascular dilation, with a focal increase in the thickness of subsegmental arteries within the pulmonary opacities. C) Pleural thickening evident at the level of the right major and minor fissures. D) Resolution of the consolidation foci that progressed to ground-glass opacities in the late phase and development of pneumatoceles (arrow). E and F) Inverted halo or atoll sign, consisting of a ground-glass opacity surrounded by a complete or incomplete ring of consolidation (arrows).
(0.35MB). - •
Vascular dilation or congestion, reported in 77% of cases.5,14,28 It could indicate infiltration of the vessels by inflammatory cells28 or hyperaemia induced by pro-inflammatory factors35 (Fig. 5A and B).
- •
Cystic changes, observed in up to 10% of cases.5,26 They could be secondary to damage to the alveolar walls with the development of pneumatoceles26 (Fig. 5D).
As the disease progresses, the consolidations resolve and revert to ground-glass opacities, comprising the most common late pattern (60%). Up to 98% of patients continue to present abnormalities 28 days after the onset of symptoms.23
In the course of the disease, dynamic changes are reported in approximately one-third of cases, with improvement in some opacities and development or worsening of others.5
Regarding pleural involvement, the most common type is pleural thickening. This constitutes a late finding, visualised after the third week,26,28,30 in up to 52% of cases29 (Fig. 5C). Pleural effusion is an uncommon finding, especially at the onset of the disease.23 It is reported in a variable proportion of cases, between 1% and 7%, in series assessing patients with mild to moderate disease.16,22,26,29 It usually increases with the duration of the course of the disease, being more common as of the second week and peaking in the third week.28
Severe disease. Respiratory distress syndrome and radiographic pattern of diffuse alveolar damagePatients with severe disease generally have ARDS,11 which is characterised, from a histological point of view, by diffuse alveolar damage.36 Post-mortem studies of patients with COVID-19 have revealed histological patterns of the exudative and proliferative phases of diffuse alveolar damage19,20 and acute fibrinous and organising pneumonia,13 with a clinical and radiological presentation that may be indistinguishable from ARDS and the radiological pattern of diffuse alveolar damage, respectively.15
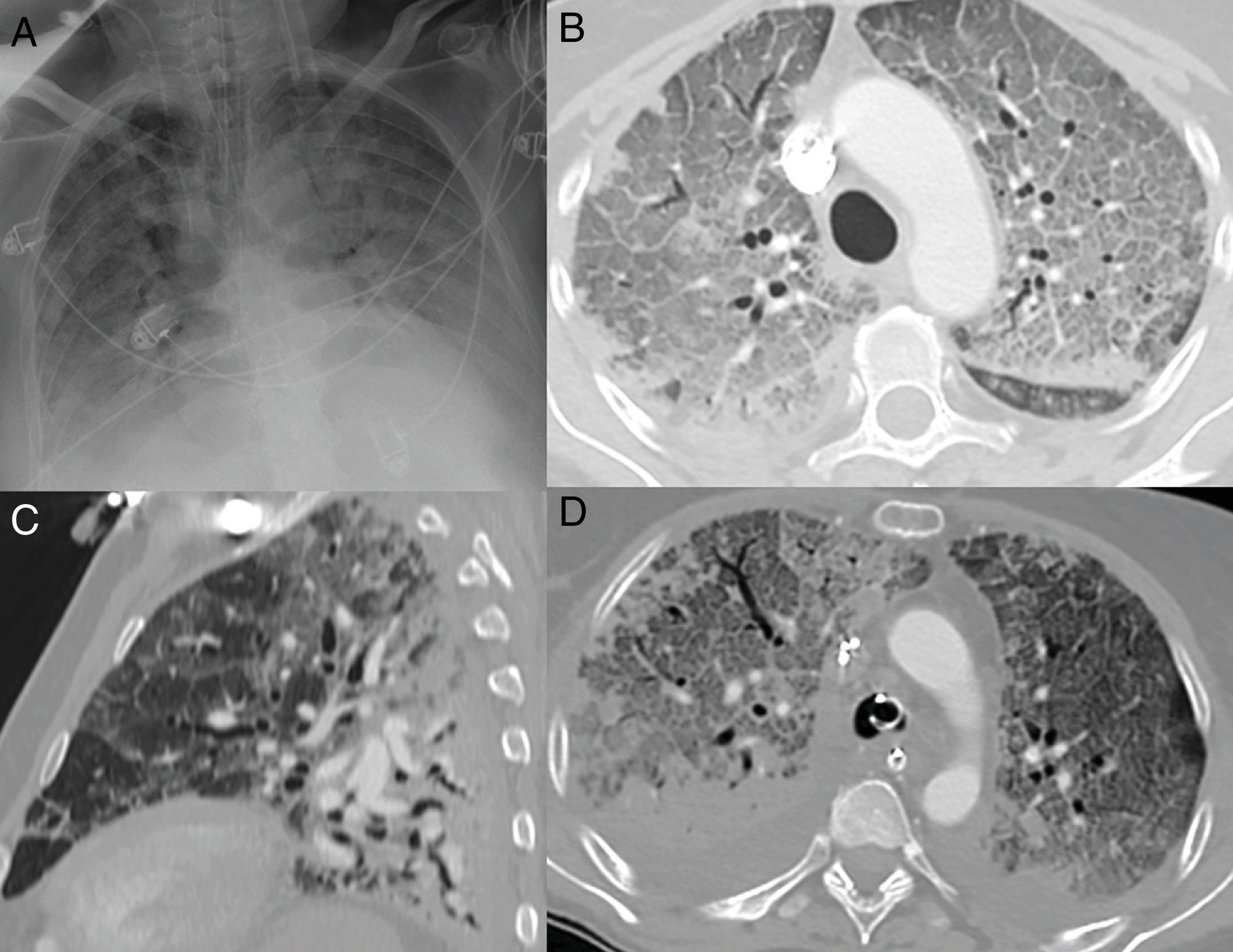
On chest X-ray, more diffuse pulmonary opacities18 are seen; there may even be complete opacification of both lungs,5 imitating the radiographic pattern of diffuse alveolar damage18 (Fig. 6A).
Radiographic pattern of diffuse alveolar damage in patients with COVID-19. A) Complete opacification of both lungs with air bronchogram. B) Extensive cobblestone pattern with small consolidation foci with a posterior predominance in a patient with symptoms for 10 days, requiring intensive care unit admission. C) Sagittal reconstruction where the typical gravitational gradient is identified with consolidations in sloping areas, ground-glass opacities and a cobblestone pattern in intermediate areas, with relatively spared areas in non-sloping areas. D) Axial image with gravitational gradient, consolidations predominantly in dependent areas and an extensive anterior cobblestone pattern. Bilateral pleural effusion, which is much more common in patients with severe disease, is seen.
On CT in patients with severe disease, pulmonary opacities continue to increase after the fifteenth day from the onset of symptoms,24 with a peak between days 22 and 28.23 By contrast, CT in patients with moderate disease shows more extensive opacities in the second week.24 Diffuse opacities are seen in 95% of cases,31 with areas of consolidation being significantly more extensive31,37 compared to patients with mild to moderate disease. The cobblestone pattern is also significantly more common37 (Fig. 6B). In addition, patients with severe disease have lower overall lung volumes, lower unaffected lung volumes,24 and more extensive opacities than patients with less severe disease.24,31
On CT, the diffuse alveolar damage pattern is characterised by a gravitational density gradient, with dense consolidation areas in dependent regions, diffuse ground-glass opacities with thickening of septa (cobblestone pattern) in intermediate zones and some areas of normal-appearing or hyperinflated parenchyma in non-dependent regions, generally featuring linear margins with a geographic appearance between the spared and affected areas15,36 (Fig. 6C and D). Posterior consolidations appear to be due to compressive atelectasis caused by the weight of the overlying lung parenchyma. Lung volumes are reduced.15,33,36 As the disease progresses, reticular opacities and often bronchial and bronchiolar dilatations appear.15,33,36 However, neither bronchiectasis nor the reticular pattern necessarily indicate fibrosis, and both findings can be reversible.36 Development of air cysts is common.33,36
Other findings present in patients with severe disease are:
- •
Pleural effusion, seen in 40% of patients31,37 and considered a poor prognostic factor.37
- •
Pericardial effusion, reported in 16% of patients.37
- •
Thoracic lymphadenopathy, which is not usually seen in patients with mild disease16,37 and is uncommon in patients with moderate disease,26,29 but is observed in one-third of cases with severe disease.31,37
- •
Thickening of the bronchial walls, reported in two-thirds of patients with severe disease.37
It is postulated that lymphadenopathy and pericardial and pleural effusion could be secondary to the severe inflammatory process,37 although other authors consider a congestive origin secondary to fluid overload due to underlying renal or cardiovascular compromise to be more likely.31
Thoracic complicationsPatients with SARS-CoV-2 infection present with different short-term chest complications.
Pulmonary embolismPatients with COVID-19 infection are at high thrombotic risk due to the systemic inflammatory state. However, there is little information on how to address thrombotic risk, coagulopathy and anticoagulant therapy in these patients. An increased risk of venous thromboembolic disease has been identified in hospitalised patients with COVID-19, especially in those admitted to intensive care units.38 These patients have reduced venous flow due to prolonged bed rest, prothrombotic changes and endothelial damage secondary to the binding of the virus to ACE-II receptors. D-dimer elevation has been associated with a worse prognosis and is a predictor of mortality.39,40 These patients are candidates for low–molecular-weight heparin thromboprophylaxis.
The International Society on Thrombosis and Haemostasis (ISTH) proposes the determination and monitoring of four parameters — D-dimer, prothrombin time, platelet count and fibrinogen — to identify patients with a poor prognosis who require more intensive monitoring.38 If a diagnosis of venous thromboembolic disease is made, then the low–molecular-weight heparin should be changed to therapeutic doses. Bleeding in these patients is common and replacement therapy should be administered, maintaining a platelet count higher than 50 × 109/L, fibrinogen >2 g/l, and a prothrombin time ratio <1.5.41
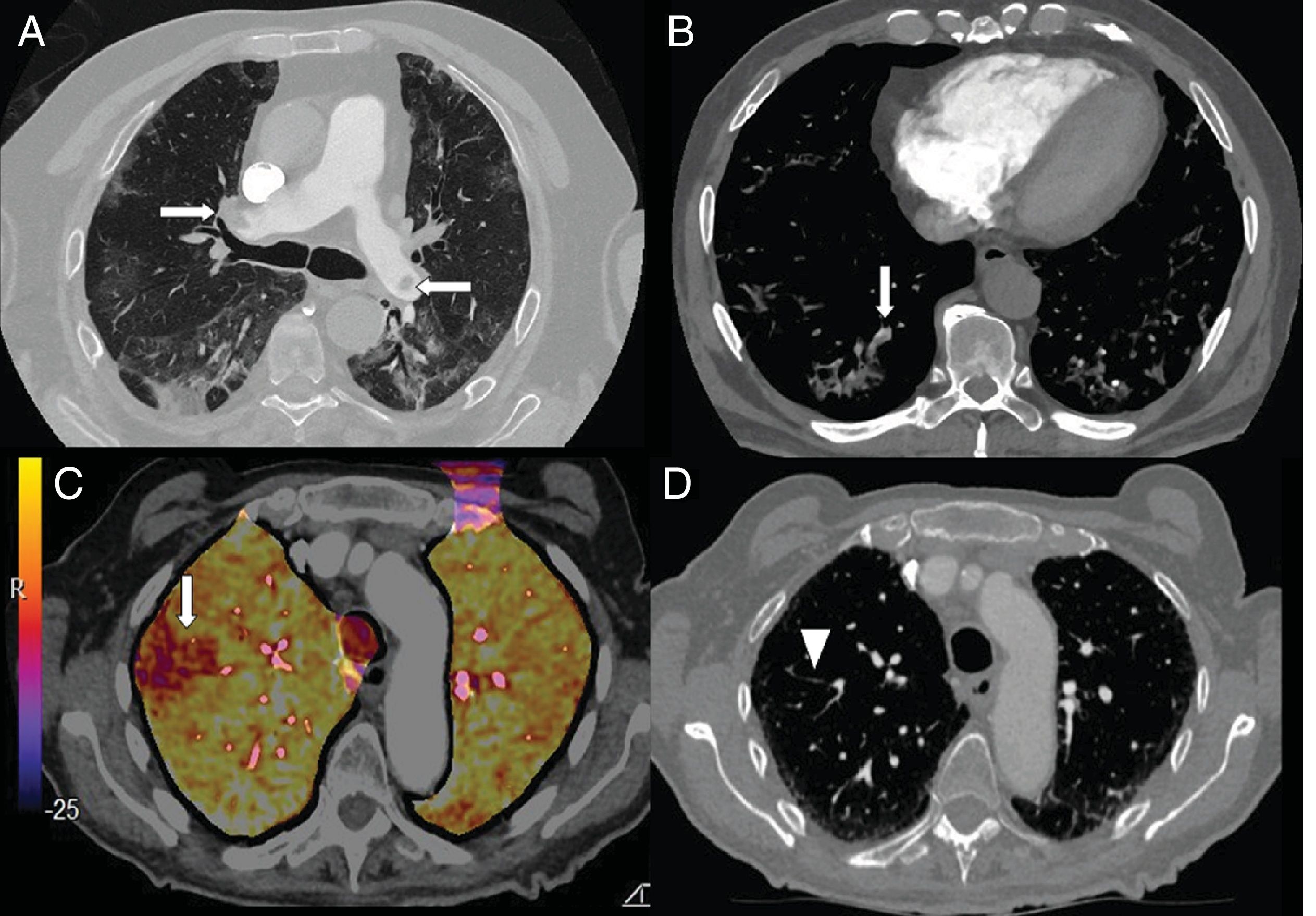
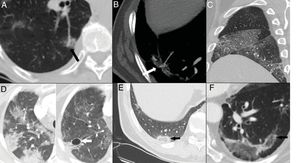
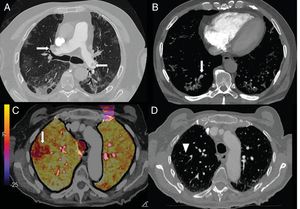
Elevated levels of D-dimer have been used in hospital emergency departments to screen for patients with COVID-19 pneumonia who require CT angiography of the pulmonary arteries to rule out pulmonary embolism (Fig. 7). In a series of 72 pulmonary CT angiographies of patients with COVID-19 pneumonia, 18% had acute pulmonary embolism, with a segmental distribution in 55% of cases, a lobar distribution in 30%, and a distribution in the main arteries in 15%. The severity and radiological findings of COVID-19 pneumonia did not present statistically significant differences between patients with and without pulmonary embolism.42
A) A 73-year-old man with bilateral central pulmonary embolism (arrows). B) A 77-year-old man with COVID-19 and segmental pulmonary embolism related to consolidation in the right lower lobe, suggesting vascular thrombosis (arrow). C and D) A 89-year-old woman with COVID-19 and D-dimer elevation. A CT angiogram with iodine subtraction mapping showed a peripheral perfusion defect (arrow) correlated with a subtle segmental and subsegmental filling defect in the right upper lobe (arrow tip).
In patients admitted for COVID-19 pneumonia, the prevalence of acute pulmonary embolism is high (23%), with a diagnosis after 12 days on average from the onset of symptoms. These patients required intensive care unit admission and mechanical ventilation more often than those without pulmonary embolism.43
In a retrospective study that included 101 hospitalised patients with COVID-19 and a pulmonary CT angiography between March and May 2020 in Rimini (Italy), pulmonary embolism was diagnosed in 40.6% of patients, more often in men than women (35% versus 6%). The distribution was bilateral in 48.7%, right-sided only in 41.5% and left-sided only in 9.8%. Involvement of the pulmonary artery trunk was detected in 2.4%, involvement of the main pulmonary arteries was detected in 22% and involvement of the lobar arteries was detected in 51.2% of cases. There was involvement of the segmental arteries in 90.2% of cases and of the subsegmental arteries in 61%. Pulmonary embolism was more common in the lower lobes (73.2%) than in the middle lobe and lingula (14.6%) or in the arteries of the upper lobes (12.2%). Parenchymal lesions associated with segmental pulmonary embolism were found: pulmonary consolidations in 67.6% of cases, a ground-glass pattern in 29.7% and no pulmonary opacities in 2.7%. Dilation of the pulmonary artery (17%) and of the right ventricle (4.9%) were uncommon. These patients presented higher values and with statistically significant differences in D-dimer (p < 0.001), lactate dehydrogenase (p < 0.001) and C-reactive protein (p = 0.0420). In summary, COVID-19 patients with more severe pulmonary involvement tend to be at higher risk of segmental and subsegmental embolism.44
Pulmonary emboli in patients with COVID-19–associated pulmonary embolism are more localised in small- to medium-sized pulmonary arteries and are less extensive compared to pulmonary embolism in non–COVID-19 patients. The absence of clinical signs of deep vein thrombosis in COVID-19 patients may also support the concept of in situ immunothrombosis rather than venous thromboembolism, although the origin of thrombotic lesions in COVID-19 patients remains unknown.45
Currently, few publications have evaluated whether there is a cut-off point for D-dimer levels in patients with COVID-19 pneumonia that suggests a diagnosis of pulmonary embolism.46
Pneumothorax and spontaneous pneumomediastinumIn patients with COVID-19 infection and gradual worsening of respiratory function, the presence of spontaneous pneumomediastinum and pneumothorax has been reported, either in isolation47–49 or in association,50 with no history of mechanical ventilation.
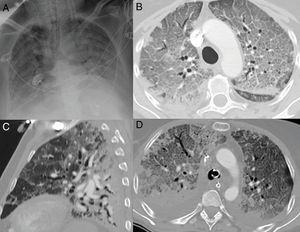
Pneumomediastinum is caused by an increase in intra-alveolar pressure, alveolar rupture, and air migration that dissects the peribronchial and perivascular sheaths of the pulmonary hilum (Maclin effect).49 Once it reaches the mediastinum, the gas spreads to the subcutaneous tissue, pleura, peritoneum and spinal canal (Fig. 8). The pronounced cough that occurs in response to the virus is one of the causes of spontaneous pneumomediastinum. It is a complication that has a good prognosis and is treated with analgesia and oxygen therapy.48
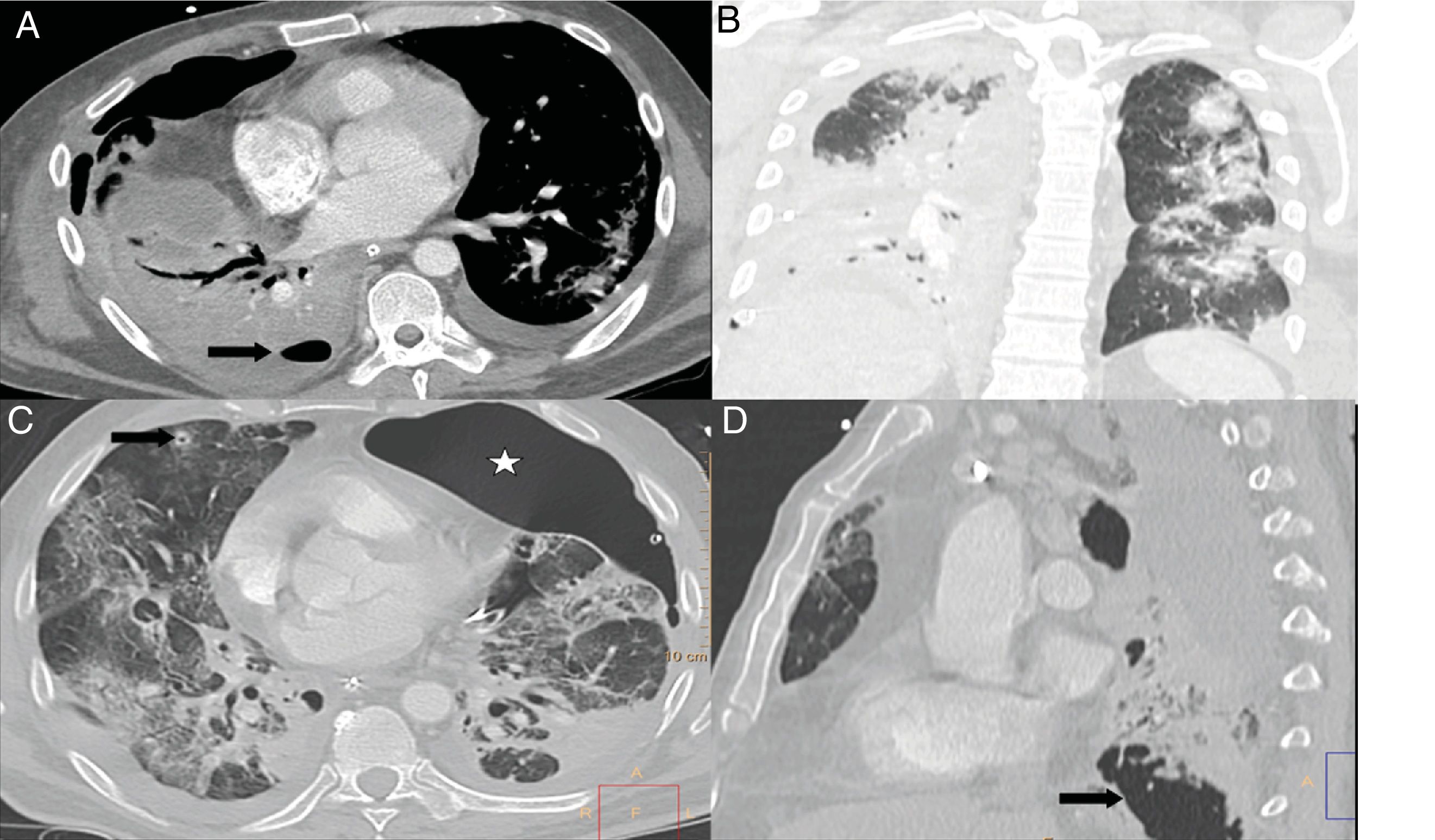
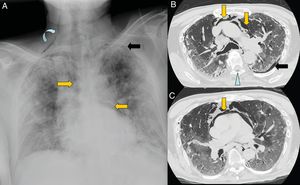
A 67-year-old man with extensive bilateral COVID-19 involvement and severe respiratory failure. Notable on the portable chest X-ray (A) are right supraclavicular subcutaneous emphysema (curved arrow), pneumomediastinum (orange arrows) and left apical pneumothorax (black arrow). Images B and C from chest CT confirm all findings and also show pneumorrhachis (blue arrow tip).
Pneumopericardium and tension pneumothorax as a complication of pneumomediastinum are considered serious complications and require emergency drainage. López-Vega et al. associated the presence of pneumothorax and pneumomediastinum with greater severity and a fatal outcome in patients, although a larger number of cases will be required to assess correlation versus causation.50
In patients with severe SARS-CoV-2 lung infection and diffuse alveolar damage, the presence of pneumothorax and spontaneous mediastinal emphysema has been associated with the appearance of pneumatoceles and bullae related to pneumonia, which did not appear in the initial stages of the infection.51
Bacterial superinfectionIt is estimated that around 15% of patients hospitalised with COVID-19 develop secondary infections, mainly due to bacteria or, more rarely, fungi. This superinfection may affect up to 50% of patients who die. Secondary infection is diagnosed when patients show clinical symptoms or signs of pneumonia or bacteraemia and a positive culture of a new pathogen is obtained from lower respiratory tract specimens (sputum, endotracheal aspirate or bronchoalveolar lavage fluid) or positive blood cultures after admission.52
A multicentre study by Xing et al.53 enrolled 68 patients with a confirmed diagnosis of SARS-CoV-2 infection, recruited from two hospitals with patients with different characteristics. The patients from Qingdao had a mean age of 50 years (range 37–59 years), and the patients from Wuhan had a mean age of 31 years (range 28–38 years). The study showed that 80% of patients from Qingdao had specific IgM antibodies against at least one common respiratory pathogen, whereas this percentage was only 2.63% in the patients from Wuhan. The most common respiratory pathogens detected were influenza A virus (60%), influenza B virus (53%), Mycoplasma pneumoniae (23%) and Legionella pneumophila (20%).
Patients with ARDS triggered by a viral infection, especially influenza, and who require invasive mechanical ventilation can develop concomitant infection with Aspergillus, even in the absence of prior immunodeficiency.54
There is growing evidence that this situation can occur in patients with COVID-19, especially in patients treated with corticosteroids, with the development of invasive pulmonary aspergillosis centred in the bronchi and alveolar tissue.55–57 The radiographic pattern is one of bronchopneumonia with airspace consolidations. However, unlike that which has been reported in COVID-19 pulmonary involvement, areas of cavitation are common (Fig. 9). Nodular images with a halo sign or inverted halo sign may also be seen.55,57
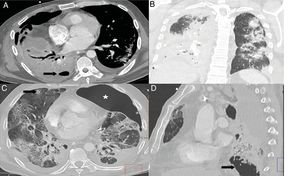
A and B) Patient with COVID-19 and moderate disease who presented worsening 10 days after admission, with leukocytosis, increased procalcitonin, complete consolidation of the right lower lobe, partial opacification of the right upper and middle lobe, and hydropneumothorax (arrow) in a context of bacterial superinfection. C) Patient with COVID-19 and severe disease, diagnosed with invasive pulmonary aspergillosis. CT showed a pulmonary nodule in the right upper lobe with an area of central cavitation (arrow) and the presence of a left pneumothorax (star). D) The same patient presented extensive cavitated lesions (arrow) at the right lung base.
The results of the analysis of the clinical characteristics in a selected cohort of 1,099 COVID-19 patients throughout China39 have shown that up to 15% (173/1,099) developed severe disease according to the American Thoracic Society clinical criteria for severe community-acquired pneumonia.58 Of these patients with severe disease, 19% (33/173) required intensive care unit admission, and 46% required the use of mechanical ventilation, both invasive and non-invasive (81/173).
A high number of patients with COVID-19 infection have developed multiple episodes of barotrauma spaced out over time, such as pneumothorax (Fig. 10), pneumomediastinum, pneumopericardium and subcutaneous emphysema, on both chest X-ray and chest CT.
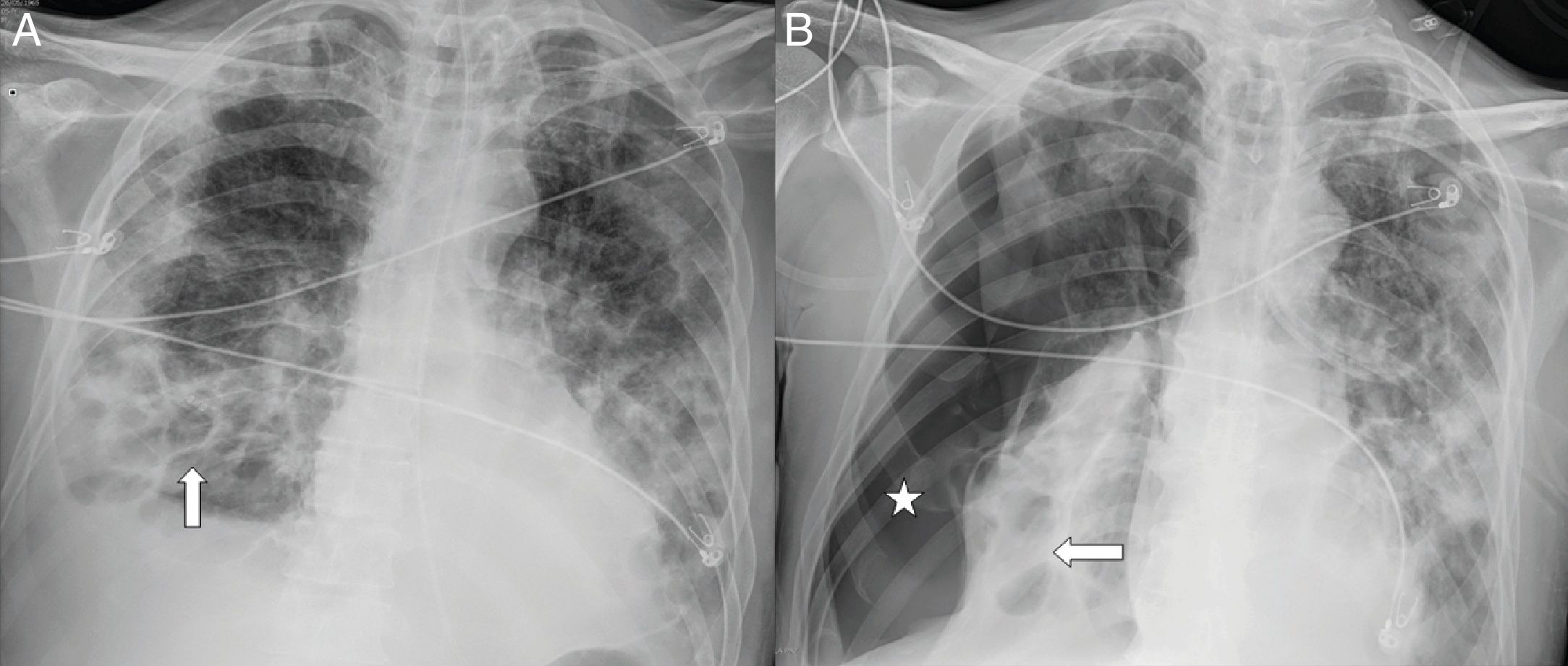
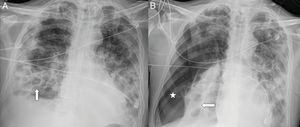
A 54-year-old man with COVID-19 pneumonia who required intubation and intensive care unit admission, one week after the onset of symptoms, due to marked clinical and radiological worsening. A follow-up X-ray one month later (A) showed pneumatoceles at the right lung base (arrows) secondary to COVID-19. B) Four days later, the patient abruptly desaturated with a significant right pneumothorax (asterisk).
A retrospective study conducted at New York University Langone Health during the peak of the pandemic,59 found a high incidence of barotrauma in patients with COVID-19 and invasive mechanical ventilation with a total incidence of 24%. The time between invasive mechanical ventilation and the first documented episode of barotrauma was 5.3 days, with a range of 0–25 days. During the same period, the rate of barotrauma in the group of patients with invasive mechanical ventilation for other causes and without COVID-19 infection was 0.5%. In this study, patients with barotrauma and COVID-19 were younger than those without this complication. There were no significant differences in mortality in patients with COVID-19 infection with or without barotrauma. Although advanced age is a risk factor for higher mortality, barotrauma was less common in these patients. In patients with COVID-19 infection, barotrauma was associated with a longer hospital stay compared to patients who did not present this complication (25 versus 18 days, p < 0.001).
Cardiac complicationsDifferent mechanisms have been reported for acute myocardial injury due to SARS-CoV-2:60
- 1
Direct myocardial injury caused by the virus binding to ACE-II receptors, which is expressed in heart and lung cells.
- 2
Abnormal myocardial oxygen demand and supply due to systemic infection, respiratory failure and hypoxia.
- 3
Acute systemic inflammatory response and cytokine storm.
- 4
Increased coronary blood flow can cause rupture of atheroma plaques, while the prothrombotic state can induce formation of intracoronary thrombi.
- 5
Adverse effects of antiviral drugs, corticosteroids and chloroquine that can cause arrhythmias and myocardial fibrosis.
Elevated troponin is a poor prognostic marker along with D-dimer and fibrinogen. The direct vascular action of SARS-CoV-2 has been responsible for late-onset myocardial infarctions and an increase in out-of-hospital sudden deaths, especially in Lombardy (Italy) and New York (United States). On other occasions, marked elevation of troponin is seen, without coronary involvement, in relation to myocardial distress of extracoronary origin, such as myopericarditis,61 cardiac tamponade, sepsis, pulmonary embolism and heart failure.
In a published study of 138 hospitalised COVID-19 patients, acute cardiac injury, shock and arrhythmia were present in 7.2%, 8.7%, and 16.7% of patients, respectively, with a higher prevalence among patients who required intensive care.6
An increased incidence of stress cardiomyopathy or Tako-Tsubo syndrome has been reported due to the psychological, social and economic stress associated with the COVID-19 pandemic. Cardiomyopathy was significantly more common (incidence 7.8%) in patients with acute coronary syndrome between 1 March and 30 April 2020, compared to four control groups on pre-pandemic timelines. The incidence of stress cardiomyopathy in the control groups was 1.5%–1.8%. There were no differences with respect to mortality and rehospitalisation at 30 days. However, patients with stress cardiomyopathy hospitalised during the pandemic had a significantly longer hospital stay. The psychological anguish and social and economic distress accompanying the pandemic represent the factors most likely associated with the increase in cases of stress cardiomyopathy, rather than direct viral involvement and the sequelae of the infection, as supported by the negative COVID-19 test results in all the patients diagnosed.62
Echocardiography is indicated for the diagnosis of acute cardiac injury related to COVID-19,18 with cardiomyopathy and cardiogenic shock identified as causes of decompensation, pericardial effusion and cardiac tamponade.63 Echocardiographic abnormalities are associated with more severe disease and a worse prognosis. Echocardiography can also reveal highly specific signs of acute pulmonary embolism that include right cardiac thrombus, McConnell’s sign (akinesia of the free wall of the right ventricle and hypercontractility of its apical wall) and paradoxical interventricular septal movement, as the main differential diagnosis.
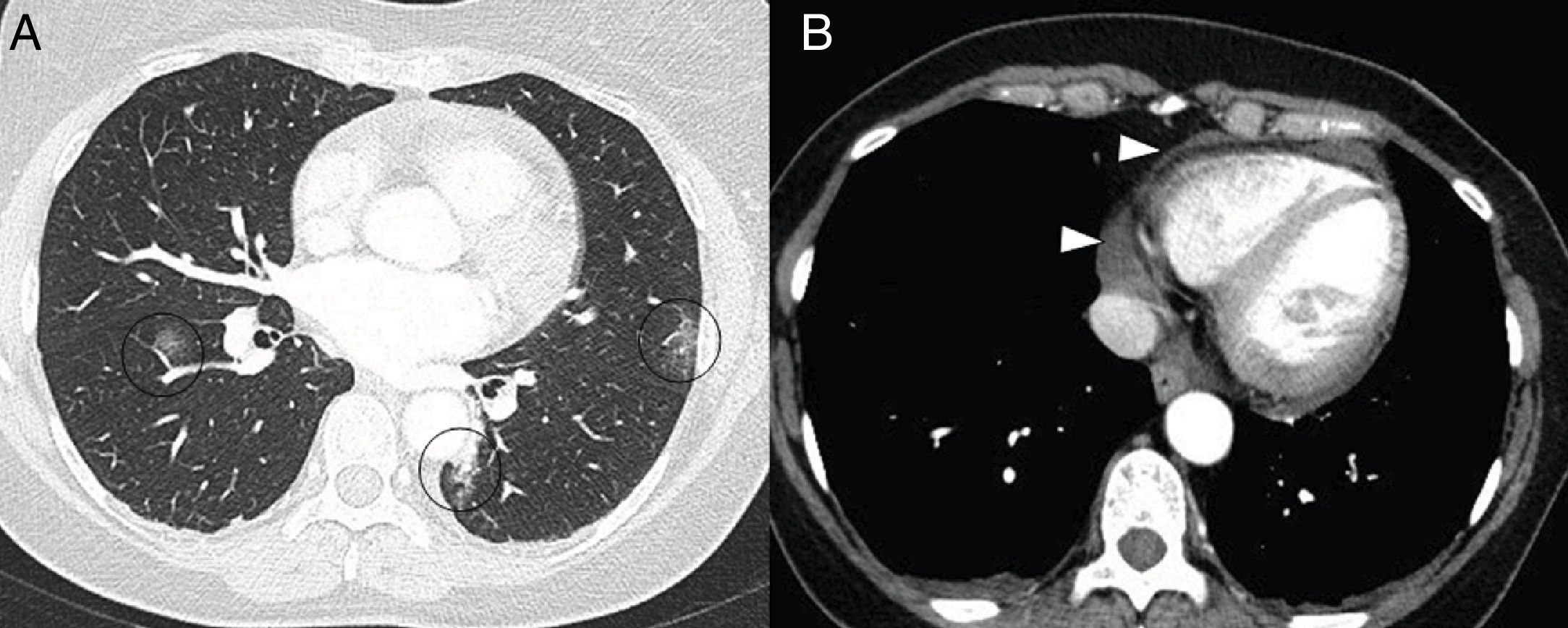
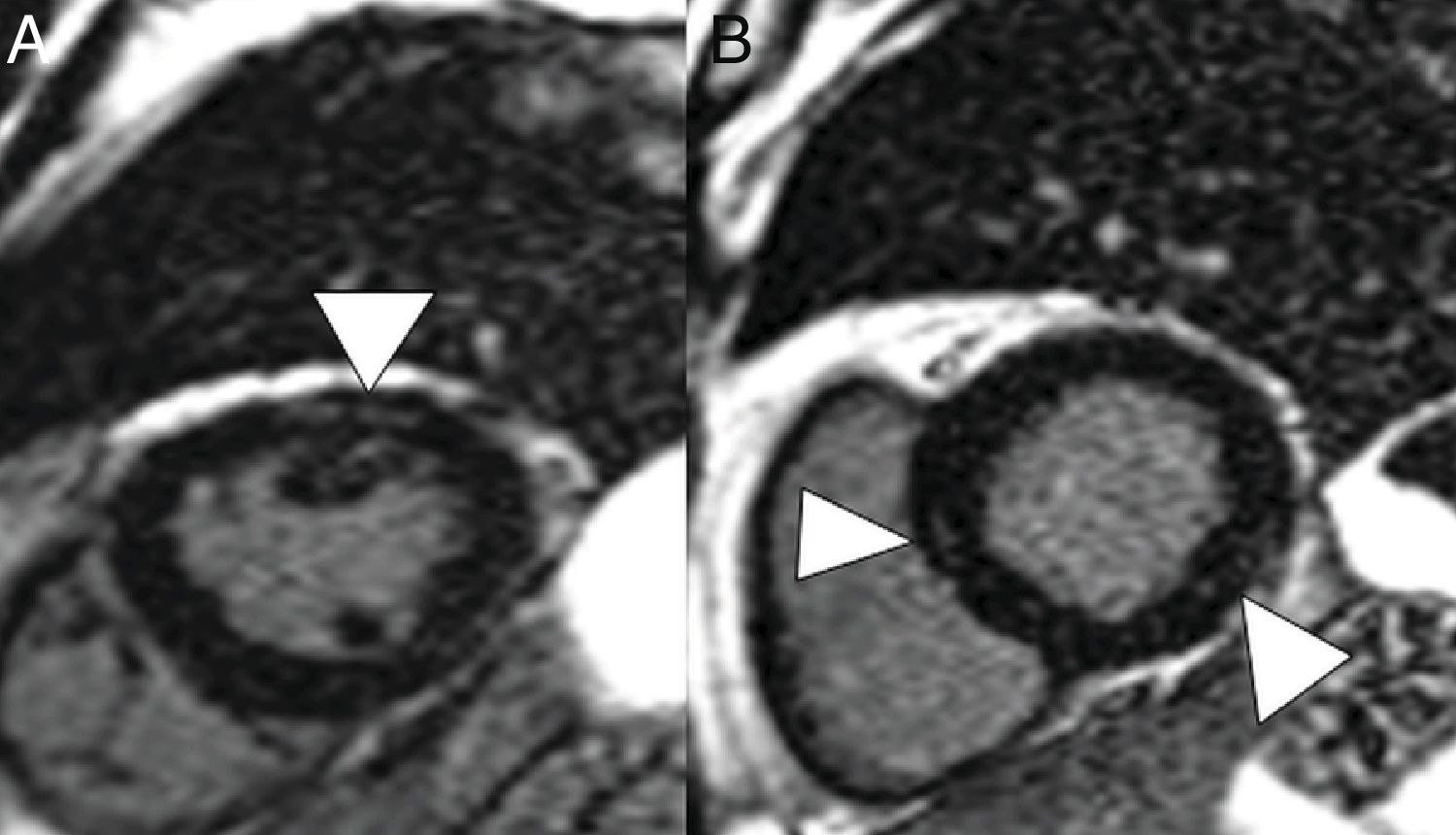
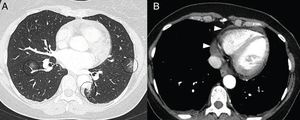
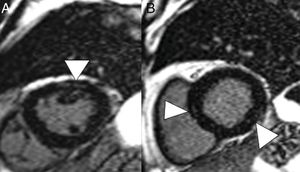
On CT, pericarditis manifests with the presence of pericardial fluid accompanying the ground-glass opacities typical of COVID-19 infection (Fig. 11). Cardiac magnetic resonance imaging may also have some useful applications, since myocarditis (Fig. 12) and cardiomyopathy have been reported in patients with COVID-19.64,65
A 59-year-old man with no personal history of note, with mild COVID-19 pneumonia. He presented chest pain and serial troponin elevation, with suspicion of inferior akinesia on echocardiography. Short-axis reconstructions and a late enhancement sequence showed hyperintense linear images in the thickness of the myocardium (arrow tips in A and B) consistent with myocarditis.
This chapter has reviewed the clinical course of patients with COVID-19, with an emphasis on those who develop severe disease with prolonged intensive care unit admission, and the associated radiological patterns of organising pneumonia and diffuse alveolar damage, as well as the characteristics of the most common thoracic complications.
Authorship- 1
Responsible for study integrity: MLPG, GBW.
- 2
Study concept: MLPG, GBW, MGG, GAC.
- 3
Study design: MLPG, GBW, MGG, GAC.
- 4
Data collection: MLPG, GBW, MGG, GAC.
- 5
Data analysis and interpretation: MLPG, GBW, MGG, GAC.
- 6
Statistical processing: N/A.
- 7
Literature search: MLPG, GBW, MGG, GAC.
- 8
Drafting of the paper: MLPG, GBW, MGG, GAC.
- 9
Critical review of the manuscript with intellectually significant contributions: MLPG, GBW, MGG, GAC.
- 10
Approval of the final version: MLPG, GBW, MGG, GAC.
All the authors declare that they have no conflicts of interest.
Please cite this article as: Parra Gordo ML, Buitrago Weiland G, Grau García M, Arenaza Choperena G. Aspectos radiológicos de la neumonía COVID-19: evolución y complicaciones torácicas. Radiología. 2021;63:74–88.