In a recent article published in your journal, Rubio et al.1 reported their experience with sentinel node biopsy (SNB) in women diagnosed with ductal carcinoma in situ (DCIS). The article justified this diagnostic procedure because a group of these patients presented metastatic involvement of the sentinel lymph node (SLN), which modified staging and therapeutic planning. Like these authors, our group concurs with the indication of SNB in women with DCIS because the identification of an underestimated infiltrative component in the initial biopsy is justified by the importance of this finding and the low morbidity of the technique. Nevertheless, the introduction of the one-step nucleic acid amplification (OSNA) method for the intraoperative study of SLN has significantly increased the incidence of micrometastases in this group of patients in whom, from an oncological standpoint, metastasisation is not possible as it is a non-infiltrating process. We therefore believe we should report our experience with SLN micrometastases in patients with DCIS to discuss the possible causes and repercussions in therapeutic planning.
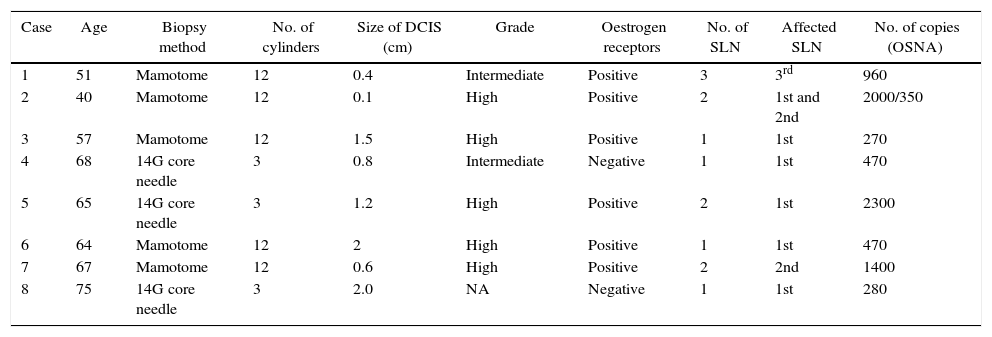
In our experience, a total of 8 patients diagnosed with DCIS presented micrometastases in the SNB since we began implementing the OSNA method in February 2011 until January 2015, without the revisions of the diagnostic biopsies and surgical specimens showing any infiltration/microinfiltration through the basal membrane (Table 1). This represents an incidence of lymph node involvement of 18.4% out of a total of 33 patients diagnosed with DCIS during this time period. The majority of the DCIS (5 out of 8) were high grade, and in 2 patients the micrometastases affected SLN numbers 2 and 3, without evidence of involvement of the first SLN. In the majority of the patients (7 out of 8), the micrometastatic involvement of the SLN did not modify the therapeutic approach, while in one patient the existence of 2 SLN with micrometastases resulted in axillary lymph node dissection (ALND), and the indication of chemotherapy treatment after surgery. In this last patient, no infiltration was observed in the previous biopsies, surgical specimens, or in the axillary fat.
Case Reports Analysed.
| Case | Age | Biopsy method | No. of cylinders | Size of DCIS (cm) | Grade | Oestrogen receptors | No. of SLN | Affected SLN | No. of copies (OSNA) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 51 | Mamotome | 12 | 0.4 | Intermediate | Positive | 3 | 3rd | 960 |
| 2 | 40 | Mamotome | 12 | 0.1 | High | Positive | 2 | 1st and 2nd | 2000/350 |
| 3 | 57 | Mamotome | 12 | 1.5 | High | Positive | 1 | 1st | 270 |
| 4 | 68 | 14G core needle | 3 | 0.8 | Intermediate | Negative | 1 | 1st | 470 |
| 5 | 65 | 14G core needle | 3 | 1.2 | High | Positive | 2 | 1st | 2300 |
| 6 | 64 | Mamotome | 12 | 2 | High | Positive | 1 | 1st | 470 |
| 7 | 67 | Mamotome | 12 | 0.6 | High | Positive | 2 | 2nd | 1400 |
| 8 | 75 | 14G core needle | 3 | 2.0 | NA | Negative | 1 | 1st | 280 |
DCIS: ductal carcinoma in situ; SLN: sentinel lymph node; NA: not assessed.
The presence of lymph node micrometastases in patients with DCIS poses 3 possible pathogenic mechanisms. The first would involve undiagnosed infiltration in the breast lesion with metastatic invasion of the SLN detected by the OSNA method. In this situation, the patient would be staged as IB, and adjuvant therapies should be considered. The therapeutic limitation of this situation is centred around the absence of an infiltrating histological component to carry out a immunohistochemistry study that would indicate this treatment. Furthermore, micrometastatic involvement of the SLN requires assessing the indication of ALND, even though currently the ACOSOG Z0011 study2 and the IBCSG 23-01 trial3 do not justify it, either with conservative surgery or with mastectomy. In our experience, ALND was done in one patient who had undergone mastectomy and presented microinvolvement of 2 SLN at a date prior to the publication of the Galimberti et al. study.3
The second theory attributes the micrometastatic involvement of the SLN to factors unrelated with the invasion of a carcinoma. This situation would represent a false positive for the neoplastic involvement of the SLN. Several authors4,5 have provided information about different mechanisms that can lead to an erroneous reading of the OSNA method for the detection of lymph node metastases. Piñero et al.4 have recently published an experience similar to ours, in which they suggest that the main mechanism is the displacement of epithelial cells during diagnostic biopsy, which correlated with the number of cylinders obtained during the biopsy as well as other data. In our case series, 5 patients were biopsied by means of vacuum-assisted biopsy with the extraction of 12 cylinders for the histological study, which is considerable trauma for the breast and can lead to the transport of epithelial component that will induce an erroneous interpretation of the actual stage of the process. Other theories propose the contamination of the SLN by shedding skin, eyebrows, eyelashes, hair, etc., during surgical manipulation or histological preparation. In these circumstances, if after careful assessment of the surgical specimen there is no infiltrative component in the breast lesion, breast cancer specialists should assess whether to maintain the final stage of 0, meaning that the process would be labelled as DCIS in spite of the lymph node microinvolvement. Finally, a third hypothesis is related with false positives of the OSNA technique due to a cut-off point that is too low. Currently, this cut-off point, based on previous studies, is arbitrary and is set at 250 copies of CK19 mRNA, but an increase in this level would significantly decrease the diagnosis of micrometastasis. In our experience, 3 patients presented less than 500 copies of mRNA.
In conclusion, the presence of micrometastatic involvement of the SLN in women with DCIS make it necessary for the pathologist to thoroughly review the surgical specimen in order to rule out the presence of microinfiltration in the breast lesion. Surgical decisions and the planning of systemic therapy should be assessed by a committee of breast cancer specialists in order to avoid disproportionate treatments, especially LA and chemotherapy.
Please cite this article as: Acea-Nebril B, García-Novoa A, Santiago-Freijanes P, Mosquera-Oses J. Micrometástasis del ganglio centinela en mujeres con carcinoma ductal in situ. Falsos positivos del método OSNA (One-step nucleic acid amplification)? Cir Esp. 2016;94:121–122.