Tuberculosis (TB) is a global public health issue. Peritoneal tuberculosis amounts to 2% of all the extrapulmonary forms. It has a difficult clinical and bacterial diagnosis.
ObjectiveTo establish the usefulness of different methods for the diagnosis of peritoneal TB in Mexican adult patients with abdominal pathology.
MethodologyA total of 44 patients with chronic abdominal pathology, clinically suspected of having peritoneal tuberculosis, were studied. All patients with peritoneum laparoscopic biopsy and ascites were studied. Studies were performed using Ziehl–Neelsen stain, cultures for microbacteria by Löwestein–Jensen (L–J) and tube of the growing indicator of Mycobacterium BACTEC (MGIT-960), real-time polymerase chain reaction (RT-PCR) with the 1S6110 insertion sequence for the Myocobacterium tuberculosis complex. Antibodies were determined by immunoenzymatic assay (ELISA) and Western blot (WB) in peripheral blood.
ResultsTuberculosis was confirmed in 22 (50%) patients through histology. In peritoneum biopsy, Ziehl–Neelsen stain was positive for AAFB (acid–alcohol-fast bacilli) in three cases (13%), in Löwestein–Jensen's cultures in five cases (22%) and in MGIT-960 in ten cases (45%). The RT-PCR was positive in twelve cases (54%). In ascites, the Ziehl–Neelsen stain was positive in one case (4%), the Löwestein–Jensen culture in nine cases (40%), in MGIT-960 culture in eight cases (36%), and in RT-PCR was positive in eleven cases (50%). The immunological methods recorded a low positivity.
ConclusionsThe bacterial and RT-PCR methods had low performance, probably due to the low bacillary load of lesions. The histopathological study with characteristic tuberculosis lesions turned out to be the most useful, and it must be jointly performed with bacteriological and molecular studies in suspected cases of tuberculosis with unknown cause ascites.
La tuberculosis (TB) es un problema de salud pública mundial. La tuberculosis peritoneal corresponde al 2% de todas las formas extrapulmonares y es de difícil diagnóstico clínico y bacteriológico.
ObjetivoEstablecer la utilidad de los diferentes métodos para el diagnóstico de TB peritoneal en pacientes adultos mexicanos con patología abdominal.
MetodologíaSe estudiaron 44 pacientes con patología abdominal crónica, clínicamente sospechosos de tuberculosis peritoneal. Todos los pacientes se estudiaron con biopsia laparoscópica de peritoneo y líquido de ascitis; se realizaron tinción de Ziehl-Neelsen, cultivos para micobacterias por Löwestein-Jensen (L-J) y tubo del indicador del crecimiento de Mycobacterium BACTEC (MGIT-960), reacción de cadena de polimerasa en tiempo real (PCRTR) con la secuencia de inserción 1S6110 para el complejo de Mycobacterium tuberculosis; en sangre periférica, se determinaron anticuerpos por ensayo inmunoenzimático(ELISA) y Western blot (WB).
ResultadosLa tuberculosis fue confirmada en 22 (50%) pacientes por histología. En biopsia de peritoneo la tinción de Ziehl-Neelsen fue positiva para BAAR (Bacilo ácido alcohol resistente) en tres casos (13%), en cultivos de Löwestein-Jensen en cinco casos (22%) y en MGIT-960 en diez casos (45%). La PCRTR fue positiva en doce casos (54%). En líquido de ascitis, la tinción de Ziehl-Neelsen fue positiva en un caso (4%), cultivo Löwestein-Jensen en nueve casos (40%), en cultivo MGIT-960 en ocho casos (36%) y en PCRTR fue positiva en once casos (50%). Los métodos inmunológicos registraron una baja positividad.
ConclusionesLos métodos bacteriológicos y la PCRTR tuvieron bajo rendimiento, posiblemente por la escasa carga bacilar de las lesiones. El estudio histopatológico con lesiones características de tuberculosis resultó el de mayor utilidad y debe realizarse conjuntamente con los estudios bacteriológicos y moleculares en casos sospechosos de tuberculosis con ascitis de causa desconocida.
Tuberculosis (TB) is an infectious disease produced by Mycobacterium tuberculosis considered as a global pandemia. Most of the cases are pulmonary; however, its haematogenous and lymphatic dissemination can result in extrapulmonary forms that correspond to approximately 20% of the total cases.1–3 Extrapulmonary TB has increased in the world and is linked to malnutrition, diabetes mellitus, pandemia of the infection caused by the human immunodeficiency virus (VIH)4 and the appearance of resistant strains to specific drugs.5 In 2008 in Mexico, over 18,000 new cases were registered, 82% pulmonary and 28% extrapulmonary, with a mortality rate of 1.7 per 100,000 inhabitants.6 Abdominal TB is rare and affects the peritoneum and abdominal organs. It can match with hepatic cirrhosis,7 carcinomatosis,8 sarcoma and in patients with peritoneal dialysis,9 it is thought that it has reappeared since 1990.10 In the United States, it takes the sixth place after the lymphatic, genitourinary, osseous, articular, milliary and meningeal locations.11
Abdominal TB appears as consequence of the reactivation of latent TB disseminated by contaminated food intake or sputum intake of pulmonary TB. The symptoms are unspecific, chronic abdominal pain, fever, loss of weight, decreased appetite, nocturnal diaphoresis, deterioration of the general status, malnutrition with weight loss, diarrhoea and sometimes poor intestinal absorption.12,13 It can be confused with Crohn's disease, amoebic dysentery, histoplasmosis, and appendicular abscesses.14 In most of the abdominal TB cases, the peritoneum is involved due to its anatomical direct relation with the organs and abdominal wall.15 Frequently, there is formation of ascites,16 fibrous bands, adhesions among intestinal loops, and there can be complications such as intestinal obstruction, abscesses and haemoperitoneum.17 The ascites is yellow-serum fibrinous or of unclear nature with high proteins, leukocytosis with lymphocytis, and there can be high figures of adenosin deaminase (ADA). However, some authors have considered this of less value for TB diagnosis.18–20
Clinical diagnosis of abdominal TB is difficult and requires a high degree of caution. Despite the fact that M. tuberculosis culture is the gold standard for the pulmonary tuberculosis diagnosis, its results are poor in the extrapulmonary forms due to the sparse bacillary load. Therefore, the lack of the M. tuberculosis pathogen does not eliminate the possibility of the disease.21 For this reason, it is important to use several complementary diagnostic methods.22–24
The aim of this study is to ascertain the frequency of peritoneal tuberculosis and the usefulness of each of the different diagnostic methods in Mexican adult patients who live in the metropolitan area.
MethodologyStudy design: This was a cross-sectional, observational, consecutive-case series type study with abdominal pathology suggesting the presence of peritoneal TB performed between January 2008 and January 2010, at a third-level concentration hospital.
Selection criteria: Patients who had chronic abdominal pain, fever, ascites, temperature higher than 37.5°C, loss of weight, peritoneal thickening data, decreased appetite and malnutrition non-attributable to any evident cause, without diagnosis and prior treatment with antituberculosis drugs.
Procedure and diagnostic studies: A full clinical history of patients was made, including haematological studies, HIV by Enzyme-Linked Inmunosorbent Assay (ELISA) and immunoelectrotransference (Western-blot), chest X-ray and high-resolution computed axial tomography scan (CAT scan).
A peritoneum biopsy was taken from every patient by laparoscopy, and a sample of ascites was taken during the intervention.25 The following blind and separate diagnostic tests were performed on each specimen.
Histopathological study of the biopsy: the fragments were fixed in formaldehyde 10%, extended in paraffin, sectioned and tinted with eosin–haematoxiline for microscopic examination. They were considered suggestive of tuberculosis when they showed the typical formation of granulomas with caseous necrosis focus and around the hyalin capsule with liquefaction, fibrosis and presence of epithelial cells. Other histopathological changes non-compatible with tuberculosis were studied in the same way.
The bacteriological studies were performed in the specimens homogenised in isotonic saline solution, buffer-phosphate, and decontaminated with 4% sodium hydroxide solution. After the neutralisation and centrifugation, each homogenised specimen was suspended again in distilled water. The AAFB (acid–alcohol-fast bacilli) were identified by the Ziehl–Neelsen stain. For cultures, 0.2mL aliquot parts of the homogenised biopsy were innoculated in bottles with half fluorescent liquid BACTEC MGIT-960 (Mycobacteria Growth Indicator Tube, Becton Dickinson, San Jose, CA, USA)® and Löwenstein–Jensen (LJ) solid medium. Cultures were incubated at 37°C for eight weeks and were inspected weekly to assess growth. A real-time polymerase chain reaction (RT-PCR) amplification was performed of an IS6110 fragment (98% sensitivity and specificity del 97%), with IS11 (5′-CACGCTAATTACCCGCTTCATCG-3′) eIS12 (5′-ATCGCGCAGCTCGCGGCGG-3′), 50μL of the reaction mix containing 0.67M Tris–HCl (pH 8.8), 0.016M ammonium sulphate, 0.01M 2-mercaptoethanol MgCl2, 2U of the Taq polymerase, 200μM each of the dATP, dCTP, dGTP and TTP, and 50pmol from every card (final concentrations) was subjected to 40 cycles of the amplification at 94°C for 30s and 67°C for 2min. An aliquot part 10-μL of amplified DNA was analysed by electrophoresis in agarose geles 2%. The 175-bp product DNA was tinted with the Gel-star (Bio Whittaker®, Walkersville®, MD, USA). The HR37Ry DNA of M. tuberculosis was employed as a positive control.26
5mL of peripheral blood were obtained to collect serum, total M, G and A anti-M. tuberculosis antibodies were determined by indirect immunoenzymatic assay (ELISA), a M. tuberculosis soluble extract (MBTSE) was used as antigen, and optic density values ≤0.239 were considered as positive values.
As a confirmation test, anti-M. tuberculosis antibodies were determined by Western blot using the H37Ra M. tuberculosis strain of five-week growth in half liquid, which was considered positive with the presence of a 4kDa-band.27,28
The ADA test was performed in 5mL of ascites. It was considered positive with a 39IU/L value. In 12 patients, a high-resolution computed axial tomography scan (CAT scan) was performed.29,30
The strictly supervised treatment with isoniazid, rifampicin, pirazinamide and ethambutol (SST) was started in those cases in which tuberculosis was documented by any method. In VIH-positive patients, the combination of zidovudine and lamivudine with efavirenz or neviparine was also indicated [28]. The clinical response was assessed during the first year.
Statistical analysis. Through the use of 2×2 contingency tables, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and Kappa index were estimated. The gold standard test was through the peritoneum biopsy. In addition, the ascites was considered as a golden standard proxy using the same diagnostic tests as those of the peritoneum biopsy.
ResultsFrom the 44 studied cases, 22 patients with histological peritoneal tuberculosis diagnosis were found, nine women and 13 men, ranging from 19 to 74 years. In eight patients lesions in the abdomen and lungs coexisted, whereas ten cases were found HIV positive (Table 1).
Cases with abdominal pathology and ascites.
| Results of the laparoscopic biopsy n=44 | |
|---|---|
| • Lesions compatible with TB | 22 |
| • Chronic inflammatory fibrosis | 9 |
| • Metastasic adenocarcinoma | 4 |
| • Fibrosis | 3 |
| • Chronic inflammatory fibrosis and peritonitis | 2 |
| • Hepatic cirrhosis | 2 |
| • Sinus hyperplasia | 1 |
| • Hodgkin's lymphoma | 1 |
The most common symptomatologies in the TB cases were abdominal pain (100%), fever (80%), malnutrition (80%), poor general state (70%) and lack of appetite (60%).
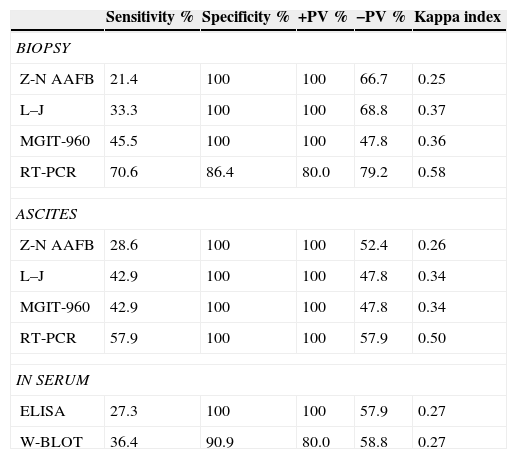
Bacteriological studiesIn peritoneum biopsies, the Ziehl–Neelsen stain was positive for acid–alcohol-fast bacilli (AAFB) in three cases (13%) with a 21.4% sensitivity. In Löwenstein–Jensen cultures (22%), a low positivity of M. tuberculosis development was present in five cases with a 33.3% sensitivity, and in MGIT-960 medium there were 10 positive cases (45%) with a 45.5% sensitivity.
In ascites from the TB cases, only one case had positive Ziehl–Neelsen stain (4%) with 28.6% sensitivity, nine cases (40%) were positive in Löwenstein–Jensen cultures for M. tuberculosis, with 42.9% sensitivity, and eight cases (36%) were positive in MGITR960 with 42.9% sensitivity.
RT-PCR amplificationIn the peritoneum biopsies, 12 cases (54%) were positive for RT-PCR with70.6% sensitivity, 86.4% specificity, 80% PPV, and 79.2% NPV.
In ascites, the RT-PCR study was positive in 11 cases (50%) with a 57.9% sensitivity, 100% specificity, 100% PPV, and 57.9% NPV. In non-TB, there three positive cases were observed, two with fibrosis and one with sinus hyperplasia.
Serological studiesThe antibody search by ELISA was positive in eight (36%) of the 22 TB cases. In both ELISA and WB methods, six positives in the confirmed TB cases were detected. For both methods, sensitivity had low values in contrast with the specificity, which was 100 and 90 respectively (Table 2).
Analysis of diagnostic tests in peritoneal tuberculosis n=44.
| Sensitivity % | Specificity % | +PV % | −PV % | Kappa index | |
|---|---|---|---|---|---|
| BIOPSY | |||||
| Z-N AAFB | 21.4 | 100 | 100 | 66.7 | 0.25 |
| L–J | 33.3 | 100 | 100 | 68.8 | 0.37 |
| MGIT-960 | 45.5 | 100 | 100 | 47.8 | 0.36 |
| RT-PCR | 70.6 | 86.4 | 80.0 | 79.2 | 0.58 |
| ASCITES | |||||
| Z-N AAFB | 28.6 | 100 | 100 | 52.4 | 0.26 |
| L–J | 42.9 | 100 | 100 | 47.8 | 0.34 |
| MGIT-960 | 42.9 | 100 | 100 | 47.8 | 0.34 |
| RT-PCR | 57.9 | 100 | 100 | 57.9 | 0.50 |
| IN SERUM | |||||
| ELISA | 27.3 | 100 | 100 | 57.9 | 0.27 |
| W-BLOT | 36.4 | 90.9 | 80.0 | 58.8 | 0.27 |
Z-N AAFB: acid–alcohol-fast bacilli. L–J: Löwestein–Jensen, MGIT-960. Mycobacterium Grow Indicator Tube. W.BLOT: Western blot. RT-PCR: real-time polymerase chain reaction. ELISA. +PV: positive predictive value. −PV: negative predictive value.
The ADA determination in ascites was only performed in 13 patients with TB, reporting average figures of 20IU/L with a range from 6IUL to 66IU/L.
High-resolution computed axial tomography scan (CAT scan)A high-resolution CAT scan was performed in 12 patients with TB, six men and six women, three with VIH-infection and three with chronic malnutrition. The presence of ascites was proven in the 12 (100%) cases. In 10 cases (83%), a thickening of the intestinal loop wall, blindgut and omentum was observed, and in seven cases (58%), an increase of the peripheral fat density was observed. In six cases (50%), a significant increase of the size of the lymphatic, abdominal and retroperitoneal nodes was identified, with small mesenteric nodules, some of them calcified. Pulmonary involvement was found in 66%, pleural involvement in 50%, and pericardial involvement in 16% (Fig. 1, Table 3).
(a) Adenomegaly at the common iliac chains with hypodense nodes and hyperdense wall. (b) Thickening of the peritoneum, abundant ascites, thickening of the intestinal loop wall and evident lymphatic nodes. (c) Diffuse increase in the attenuation of the peritoneal fat. Calcified mesenteric lymph nodes of granulomatous aspect. (d) Ascites of perihepatic distribution, discreet increase of peripheral fat density and small nodules. (e) Important thickening of the blindgut wall, hypodense lymph nodes. Increased density in the mesenteric fat. (f) Wide hypodense retroperitoneal adenomegaly with well-defined precaval and paraaortic walls adjacent to the caeliac artery and portal hepatic region.
Peritoneal tuberculosis and high-resolution computed tomography scan (cat scan).
| General imaging (high-resolution cat scan) findings n=12 | ||
|---|---|---|
| Abdominal | ||
| • Ascites | 12 | 100% |
| • Peritoneal thickening | 10 | 83% |
| • Fat attenuation peritoneal and mesenteric | 7 | 58% |
| • Mesenteric lymph nodes | 6 | 50% |
| • Retroperitoneal lymph nodes | 6 | 50% |
| • Abdominal wall thickening | 6 | 50% |
| • Bands | 4 | 33% |
| • Mesenteric nodules | 4 | 33% |
| • Thickening | ||
| • Of the blindgut wall | 2 | 16% |
| • Thickening of bowel loops | 2 | 16% |
| Thoracic | ||
| • Lung involvement | 8 | 66% |
| • Pleura involvement | 6 | 50% |
| • Pericardium involvement | 2 | 16% |
Note. Different alterations variably match in the different cases. The most important are mentioned.
In this study, 22 cases with non-tuberculosis pathology were found, 15 men and seven women, from 22 to 65 years. The most frequent non-tuberculosis pathology was chronic inflammatory fibrosis with 9 cases (41%) and the rest with varied pathology (see Table 1).
In the group of 22 tuberculosis cases, 10 died within a year, eight being HIV positive. For the remaining 12, follow-up a year from the beginning of treatment showed that five recovered, five had discreet improvement and two abandoned treatment.
DiscussionIn this study, the definitive diagnostic complexity of the peritoneal TB is proven, which, due to its unspecific clinical manifestations, does not tend to arouse suspicion of tuberculosis aetiology because it can be confused with other non-tuberculosis diseases with alterations of the abdominal structures, revealing a true clinical dilemma31–33 even in emergency cases.34 Since most of the extrapulmonary locations of the TB are of endogenous origin by reactivation of a latent infection,35 the same subject can have tuberculosis lesions in different sites, the coincidence with pulmonary TB being of diagnostic value.
The most serious consequence of the delay in diagnosis was reflected in the fact that of 44 patients who started the studies, 10 from the TB group died within a year, eight of them HIV positive, due to severe sepsis and advanced TB symptoms, and two HIV negative, due to advanced tuberculosis peritonitis and intestinal perforation. In the clinical study, the constant abdominal pain, fever and sweating of variable intensity, the deterioration of the general state and malnutrition with weight loss could speed the diagnosis.36 The high-resolution CAT scan provides helpful information. Among the most evident data, the free ascites coincident with retroperitoneal adenopathies were present. These can be of great size, uniform thickening of the peritoneum, enlarged bands with distorted vessels in the mesentery, diffuse opacities at the omentum, thickening of the intestinal wall and the blindgut.
Laparoscopy with direct biopsy sampling to perform confirmatory diagnostic studies turned to be the most useful because the different tests could be carried out in this material.37–40
The bacteriological demonstration of M. tuberculosis as a gold standard did not result in complete operation, possibly due to the sparse bacillary load of the tuberculosis lesions present in the abdominal cavity and its different structures. The negativity of the different tests does not necessarily imply the absence of TB.41 In the global result analysis, the histopathology had the best diagnostic efficiency, even it had been proposed as alternative gold standard,42 with greater compatibility with the clinical impression and imaging.43–46 Also, the disease progress can be assessed with a new laparoscopic biopsy.47,48 The lesions which best matched with TB were small white granuloma from 2 to 4mm on the parietal and visceral peritoneum associated to fibrine deposits with congestion and agglutination of intestinal loops. The positive histopathological result matching with the positive RT-PCR supports the diagnosis for TB in HIV negative and positive subjects, as occurs in pulmonary TB.49,50 The two non-TB cases with positive RT-PCR could be due to latent infection, contamination of the sample or false positives. The low positivity of ELISA and Western blot decreasing in the presence of an HIV positive could deregulate the antibody response. The ADA figures obtained in this study (20UI/L with a range of 6UIN/L to 66UI/L) are lower than the ones of other authors, with values near to the reported in cerebrospinal fluid, probably due to low protein concentration in the samples analysed as a consequence of malnutrition. Some authors consider 27U/L values useful.51
In this study, all patients had ascites in variable amounts with palpable urachus, indicating that in similar cases of non-proven aetiology, TB must be discarded as a diagnostic possibility.50,52
The limitations of this study mainly refer to the low positivity of M. tuberculosis cultures and the possibility that the histological lesions are of an aetiology different from tuberculosis. However, clinical progress and good response to treatment support the tuberculosis aetiology. It should be pointed out that the result of each test by itself would have little value for the diagnosis, which depends on the interpretation of the set of results.
ConclusionsThe isolation of M. tuberculosis in L–J and MGIT-960 media is the gold standard for the tuberculosis diagnosis. However, when it is negative, which is frequent in abdominal tuberculosis, that does not imply the absence of TB. In that case, the histopathological result of the laparoscopic biopsy with granulomas, caseosis, epithelial cells and TB-characteristic granulomas, has high reliability and has been proposed as alternative gold standard. The positive RT-PCR matching with the histopathology supports the TB diagnosis. The imaging with high-resolution CAT scan shows the extension and characteristics of the abdominal lesions suggestive of the disease. None of the bacteriological studies, AAFB search, cultures, molecular biology and serological studies was completely satisfactory on its own. A therapeutical test with good results confirms the long-term diagnosis. Peritoneal tuberculosis remains as a high-mortality clinical dilemma associated to VIH/AIDS.
Conflict of interestThe authors declare that they have no conflict of interests.
To Arturo Reding MC from the Research Division, HGM “Eduardo Liceaga” and Marco Gudiño, Computing Technician at the Clinical Research Unit, School of Medicine, and UNAM for its important contribution in the data analysis and paper writing.