Common variable immunodeficiency (CVID) is a heterogeneous group of primary antibody deficiencies defined by marked reductions in serum IgG, IgA and/or IgM levels and recurrent bacterial infections. Some patients are associated with defects in T cells and regulatory T cells (Tregs), resulting in recurrent viral infections and early-onset autoimmune disease.
MethodsWe analyzed whether there is an association between Tregs cells (CD4+CD25+CD127low and CD4+CD25+FoxP3+); memory T cells (CD4+CD45RO+); memory B cells (CD19+CD27-IgD-); and CD21low B cells (CD19+CD38lowCD21low); as well as autoimmune manifestations in 36 patients with CVID (25 women and 11 men, mean age 24 years), all by flow cytometry.
ResultsFourteen patients presented with autoimmune diseases (AI) (39%), including 11 with autoimmune thrombocytopenia (ITP) (31%); two with vitiligo (6%); one with systemic lupus erythematosus (LES) (3%); and one with multiple sclerosis (MS) (3%). CVID patients with AI had a reduced proportion of Tregs (both CD4+CD25+CD127low and FoxP3+ cells) compared with healthy controls. CVID patients with AI had expanded CD21low B cell populations compared with patients who did not have AI. A correlation between increased CD4+CD45RO T cell populations and reduced Tregs was also observed.
ConclusionsOur results showed that 39% of patients with CVID had AI and reduced Tregs populations. Research in this area might provide noteworthy data to better understand immune dysfunction and dysregulation related to CVID.
Common variable immunodeficiency (CVID) is a heterogeneous syndrome characterized by failed B cell differentiation, defective immunoglobulin production, absent specific antibody production, which lead to recurrent sino-pulmonary and gastrointestinal infections. Additionally, patients may suffer from chronic gastroenteropathies, autoimmunity and even malignancy. The incidence of CVID is 1:10,000 to 1:50,000, making it the most frequent primary antibody deficiency. In addition, CVID requires life-long treatment.1
Genetic mutations account for only 2–10% of patients, whereas for most patients the underlying molecular defect associated with CVID remains unknown.2 Several defects in B cell differentiation, as well as T cell abnormalities, have been reported in some studies. In the majority of CVID patients, B cells fail to differentiate, which alters the peripheral blood B cell subsets; in 75% of cases, there is a loss of class-switched memory B cells.3 Several lines of evidence support disturbed T cell immunity in patients with CVID, including impaired cell proliferation and cytokine production, as well as reduced T cell numbers.4,5 Recent reports have indicated reduced numbers of regulatory T cells (Tregs) in CVID patients and the correlation of this finding with chronic inflammation, splenomegaly and autoimmune manifestations.6–8 Tregs have an essential role in controlling immune responses and maintaining peripheral self-tolerance; these cells are characterized by surface expression of CD4 and CD25 in combination with intracellular expression of the transcription factor forkhead box P3 (FoxP3) and are arguably the best-characterized Tregs subset. Tregs lymphocytes are dysfunctional in some autoimmune diseases but are also responsible for the control of immune responses to foreign antigens.9 These cells develop in the thymus and constitute a variable percentage of CD4+ T cellsin the human circulation; some studies report that Tregs represent up to 5–10% of peripheral blood lymphocytes. Low expression of the interleukin (IL-7) receptor alpha chain (CD127) on CD4+/CD25+ T cells has been suggested as a reliable marker of the Tregs population and correlates well with FoxP3 expression; these cells express CTLA4, a ligand that suppresses T cell activation upon interaction with CD80/CD86.10
Autoimmune diseases are present in 10–30% of CVID patients; of these diseases, autoimmune cytopenias are highly common.11 Autoimmune cytopenias are characterized by the production of antibodies against blood cells and include autoimmune hemolytic anemia (AIHA), autoimmune neutropenia (AIN), and autoimmune thrombocytopenia (ITP) or combinations of these conditions.12 Systemic lupus erythematosus, rheumatoid arthritis, autoimmune thyroid disease, pernicious anemia, psoriasis, and vitiligo are also observed in CVID patients at lesser frequencies.13 The pathogenesis of autoimmune complications among patients with CVID is not well defined. Reduction of switched memory B cells and expansion of CD21low B cells are associated with autoimmunity in patients with CVID.14 The number of circulating CD21low B cells is increased in systemic lupus erythematosus (SLE) and rheumatoid arthritis (RA)15; however, patients with SLE and RA have normal numbers of memory B cells.16 T cell dysregulation is also associated with the development of autoimmunity in CVID. Lymphopenia affects mostly CD4+ cells and especially naïve CD4+ cells, and these effects seem to be stronger in patients with autoimmune diseases.4,17 Many reports have described reduced numbers of Treg in patients with CVID.18
The number of Tregs is affected in CVID patients with specific gene deficiencies, such as TACI,19 LRBA,20 RAC221 and CTLA-422; however, in recent years, there have also been studies of Treg and the correlation with subsets T and B cells in CVID patients without known mutations.23,24 Here, we investigated the frequency of Tregs in CVID patients and correlated Tregs frequency with the frequencies of T and B cell subsets and the occurrence of autoimmune manifestations. We observed reduced levels of Tregs in patients with autoimmune diseases who also presented with altered frequencies of T and B cell subsets.
Patients and methodsPatientsThirty-six patients with a diagnosis of CVID, based on the standard criteria suggested by the European Society for Immunodeficiency ESID and Pan-American group for Immunodeficiency (PAGID), were recruited after reading, accepting and signing an informed consent letter. Briefly, CVID patients showed hypogammaglobulinemia and variable numbers of B cells (>2%), with the exclusion of other well-known, single gene defects.25 For the diagnosis of autoimmune diseases, the systematic serologic screening for autoimmunity included a direct Coomb's test and examinations of anti-nuclear antibodies (ANAs), anti-double-stranded DNA antibodies (anti-dsDNA) and antiphospholipid antibodies, according to the diagnostic criteria for autoimmune diseases of the American College of Rheumatology (ACR) and European League Against Rheumatism (EULAR).
ITP, AIHA and Evans syndrome were defined according to the criteria suggested by the American Society of Hematology Guidelines. The diagnosis was based mainly on clinical history, physical examination, and analysis of peripheral blood smears, which excluded other causes of thrombocytopenia.26 All patients received replacement therapy every 21 days with intravenous immunoglobulin (IVIG) at a dose of 400–600mg/kg/dose. Peripheral blood samples were obtained before IVIG infusion in patients who were not suffering from any severe infection. Samples from 13 healthy, age-matched control subjects were also included in all analyses described below. This study was approved by the local ethics committee of Instituto Nacional de Pediatría, SSA (project number 26/2016).
Lymphocyte population and subpopulation phenotypingTo assess the different lymphocyte populations and subpopulations, we performed flow cytometric analysis of controls and CVID patients before IVIG infusion. Lymphocyte populations were enumerated in whole-blood samples and stained with the following mixtures of monoclonal antibodies (mAbs): anti-CD45-FITC/anti-CD14-PE, anti-CD3-FITC/anti-CD19-PE/anti-CD45-PerCP, anti-CD4-FITC/anti-CD8-PE/anti-CD3-PerCP, anti-CD3-FITC/anti-CD19-PE/anti-CD45-PerCP, and anti-CD3-FITC/anti-CD16+56PE/anti-CD45-PerCP. The following antibodies were used to detect naïve (CD45RA+) and memory (CD45RO+) T cells: anti-CD45-RO-FITC/anti-CD45-RO-PE/anti-CD3-PerCP/anti-CD4-APC. In addition, γ1-FITC/γ2-PE/anti-CD45-PerCP were used for isotype controls. All antibodies were purchased from BD Biosciences, San Diego, CA, USA. Samples were incubated for 20min at room temperature in the dark. After incubation, erythrocytes were lysed by suspending the cells in 500μl FACS lysing solution (BD Biosciences) for 10min. Cells were then washed with PBA (1% bovine serum albumin in PBS) and fixed using 1% formalin in PBS. To identify B cell populations, peripheral mononuclear cells (PMBCs) were isolated using density gradient centrifugation with Histopaque®-1077 (Sigma–Aldrich, St. Louis, MO, USA). Isolated PMBCs were then stained with a mixture of anti-CD19-APC/anti-IgD-FITC/anti-CD27-PE, anti-CD19-FITC/anti-CD21-PE/anti-CD38-APC, and anti-CD19-FITC/anti-CD24-PE/anti-CD38-APC (BD Biosciences, San Diego, CA, USA); samples were incubated for 20min at room temperature in the dark. Next, the samples were washed using PBA and fixed in PBS containing 1% formalin. The Freiburg classification was used to identify groups of patients based on PBMC staining with the following antibodies, as described in Re. 27. Group Ia represents patients with an isotype-switched memory B cell fraction (CD19+/CD27+/IgD-) less than 0.4% of the total lymphocyte population, along with a proportion of CD21low B cells equal to or exceeding 20% of total B cells. Group Ib included patients with a low percentage of switched memory B cells as detailed above, but with a normal proportion of CD21low B cells; and group II included patients with normal B cell subsets.
Tregs were detected by staining PMBCs with anti-human FoxP3-Alexa-Fluor 647 (BD Biosciences) CD25-PE (eBioscience, San Diego CA, USA), CTLA-4-PECy5 and CD4-FITC (BD Biosciences); the frequency of Tregs (FoxP3+/CD25+ cells) was calculated from CD4 T cells, and the frequency of CTLA-4+ cells was calculated from the total FoxP3+/CD25+ cell population. Additional analysis included cell surface staining using anti-CD25-PE (eBioscience), anti-CD4-PerCP (BD Biosciences) and anti-CD127-APC (BD Biosciences) antibodies were conducted using the blood cell lysis technique. Cells that were CD25+/CD127low were determined and expressed as a percentage of CD4 cells. Isotype control was used for FOXP3 and CTLA-4, with the following reagents: Alexa-Fluor 647 mouse IgG1 isotype control, cloneMOC-21; and PECy5 mouse IgG1 isotype control, clone MOC-21(BioLegend) (Fig. 2a).
Statistical analysisStatistical analyses were performed using GraphPad Prism, version 5.0 (GraphPad Software Inc., La Jolla, CA, USA). Two-group comparisons were performed using the Mann–Whitney test. Stratified data were expressed as the mean value, and classified variables were evaluated by Fisher's exact test to demonstrate independence from clinical parameters. Spearman's coefficient (R2) was used to determine the significance of the correlation between two variables. The results are expressed as the mean, and p-values <0.05 were considered to be significant.
ResultsClinical and demographic data of CVID patientsThe demographic details of the subjects included in this study are shown in Table 1. This study included 36 patients with CVID (11 males and 25 females), and the median age was 24 years. A group of 30 healthy individuals was included as controls. At diagnosis, 96% of the patients had decreased serum IgG (two standard deviations below the reference levels according to age); the mean value was 397mg/dl for IgG, and 55mg/dl and 173mg/dl for IgA and IgM, respectively (Table 1). Only one patient had a family history of consanguinity. The main manifestations of the patients were recurrent infections among which pneumonia was present in 61% of patients, otitis media in 44% and sinusitis in 50%. Other non-infectious clinical manifestations included splenomegaly in 19% of patients, lymphadenopathy in 14%, and granulomas 6%. Autoimmune diseases were observed in 14 patients (37%), including 11 with autoimmune thrombocytopenia (ITP) (31%); two with vitiligo (6%); one with systemic lupus erythematosus (SLE) (3%), and one with multiple sclerosis (MS) (3%). Two groups of patients with or without AI were defined for analysis (n=14 and n=22, respectively). Additionally, CVID patients were also classified according to Freiburg criteria. Seven patients had low levels of memory B cells and increased frequencies of CD21low B cells (Group Ia), 11 patients had low levels of memory B cells and normal frequencies of CD21low B cells low (Group Ib), and 13 patients had normal levels of memory B cells (Group II) (Table 1). CD21low B cells were not determined in five patients, and therefore, these patients were not analyzed according to the Freiburg classification.
Clinical characteristics of 36 CVID patients.
| Total patients (n=36) | Patients with autoimmune manifestations (n=14) | Patients without autoimmune manifestations (n=22) | |
|---|---|---|---|
| Sex (female/male) | 25/11 | 8/6 | 17/5 |
| Mean age | 24 | 21 | 25 |
| Pneumonia | 22 (61%) | 8 (57%) | 14 (64%) |
| Sinusitis | 18 (50%) | 8 (57%) | 10 (45%) |
| Otitis | 16 (44%) | 7 (50%) | 9(41%) |
| Chronic diarrhea | 5 (14%) | 1 (7%) | 4 (18%) |
| Gastroenteritis | 4 (11%) | 1(7%) | 3 (14%) |
| Lymphoma | 3(8%) | 0(0%) | 3(14%) |
| Bronchiectasis | 4 (11%) | 0 (0%) | 4 (18%) |
| Splenomegaly | 7 (19%) | 3 (21%) | 4 (18%) |
| Lymphadenopathy | 5 (14%) | 0 (0%) | 5 (23%) |
| Granulomas | 2 (6%) | 1 (7%) | 1 (5%) |
| Mean IgG (mg/dl) | 397 | 412 | 387 |
| Mean IgM (mg/dl) | 173 | 76 | 230 |
| Mean IgA (mg/dl) | 55 | 44 | 62 |
| Freiburg's classification: | |||
| Ia | 7 | 5 | 2 |
| Ib | 11 | 2 | 13 |
| II | 13 | 5 | 8 |
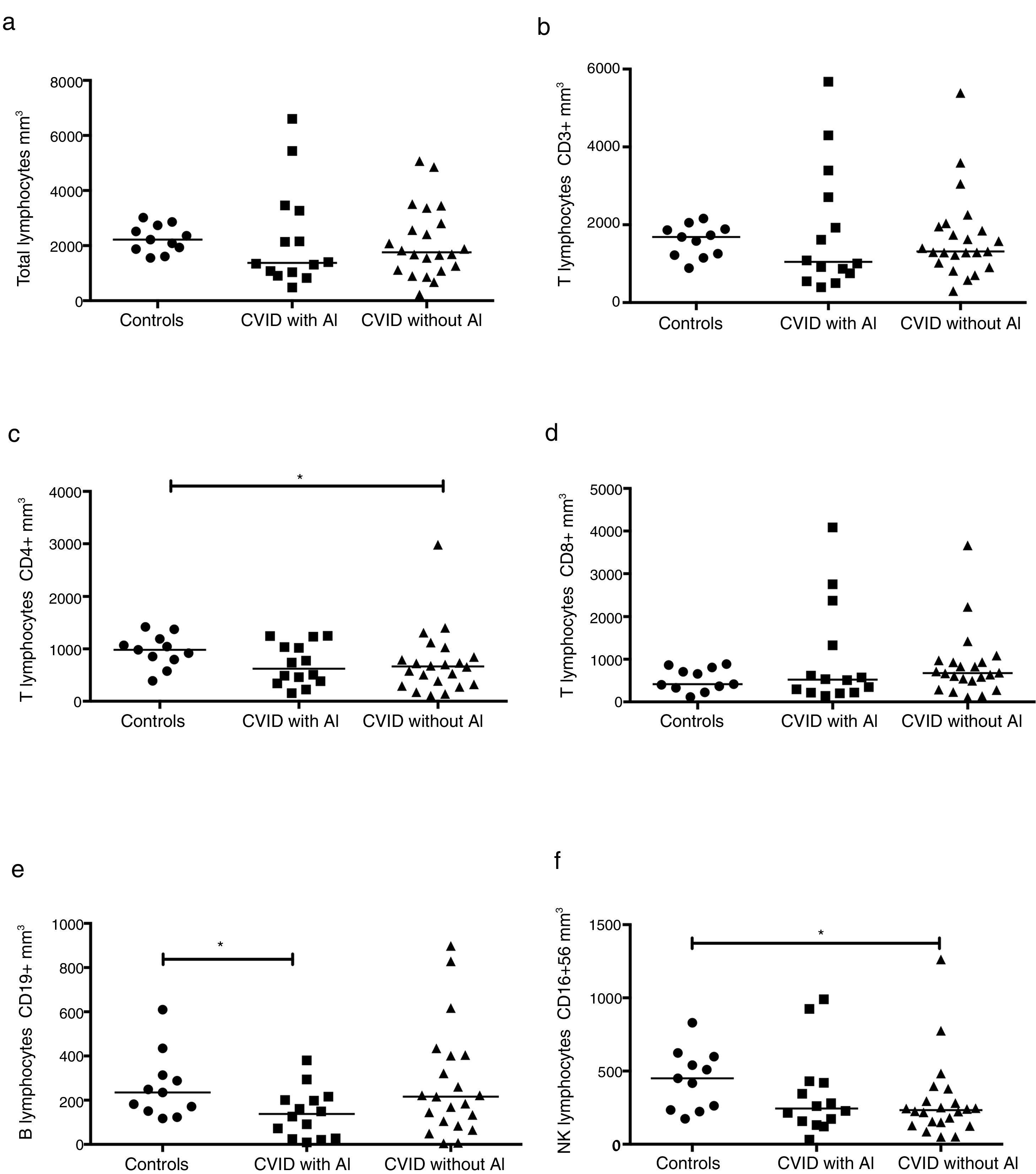
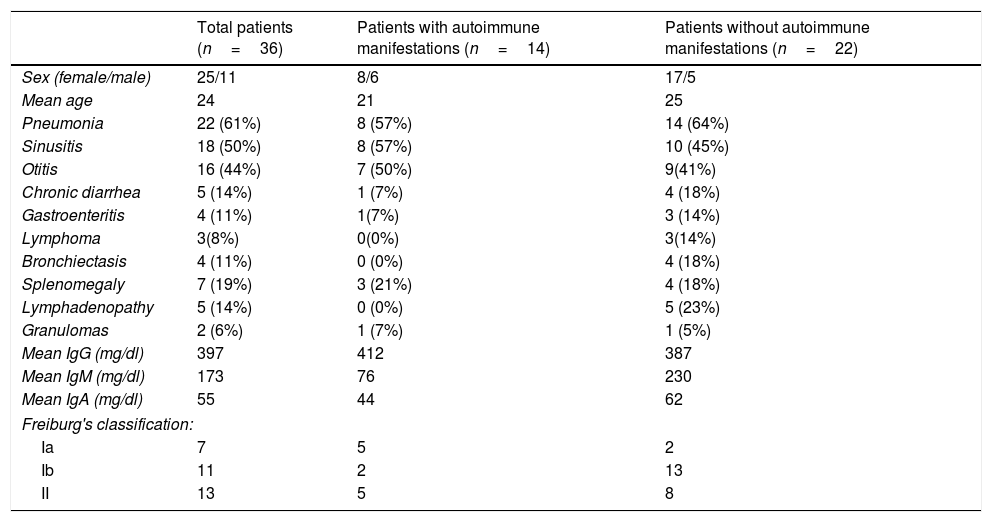
The absolute numbers of different cell populations were compared in patients with and without AI. As shown in Fig. 1, levels of total lymphocytes, CD3+ T cells, and CD3+CD8+ T cells were similar in both groups of patients compared with the healthy controls (Fig. 1a, b, and d). CD4+ T cells and NK cells were significantly reduced in the group of CVID patients without AI compared with the healthy controls (p=0.0264 and p=0.0196, respectively); patients with AI, however, showed levels similar to those of the control group (Fig. 1c and f). CVID patients with AI also showed significantly reduced numbers of CD19+ B cells in comparison with the healthy controls (p=0.0383).
Analysis of lymphocyte populations. Flow cytometric analysis of total lymphocytes, T cells, B cells and NK cells in absolute numbers (cells/mm3) from CVID patients with autoimmune diseases (AI) (n=14) (■) and CVID patients without AI (n=22) (▴). These groups were compared with healthy controls (n=13) (●). Differences between patients and controls were compared using the Mann–Whitney U-test. (*) Significant, p=<0.05; (**) very significant, p=<0.01; and (***) highly significant, p=<0.001.
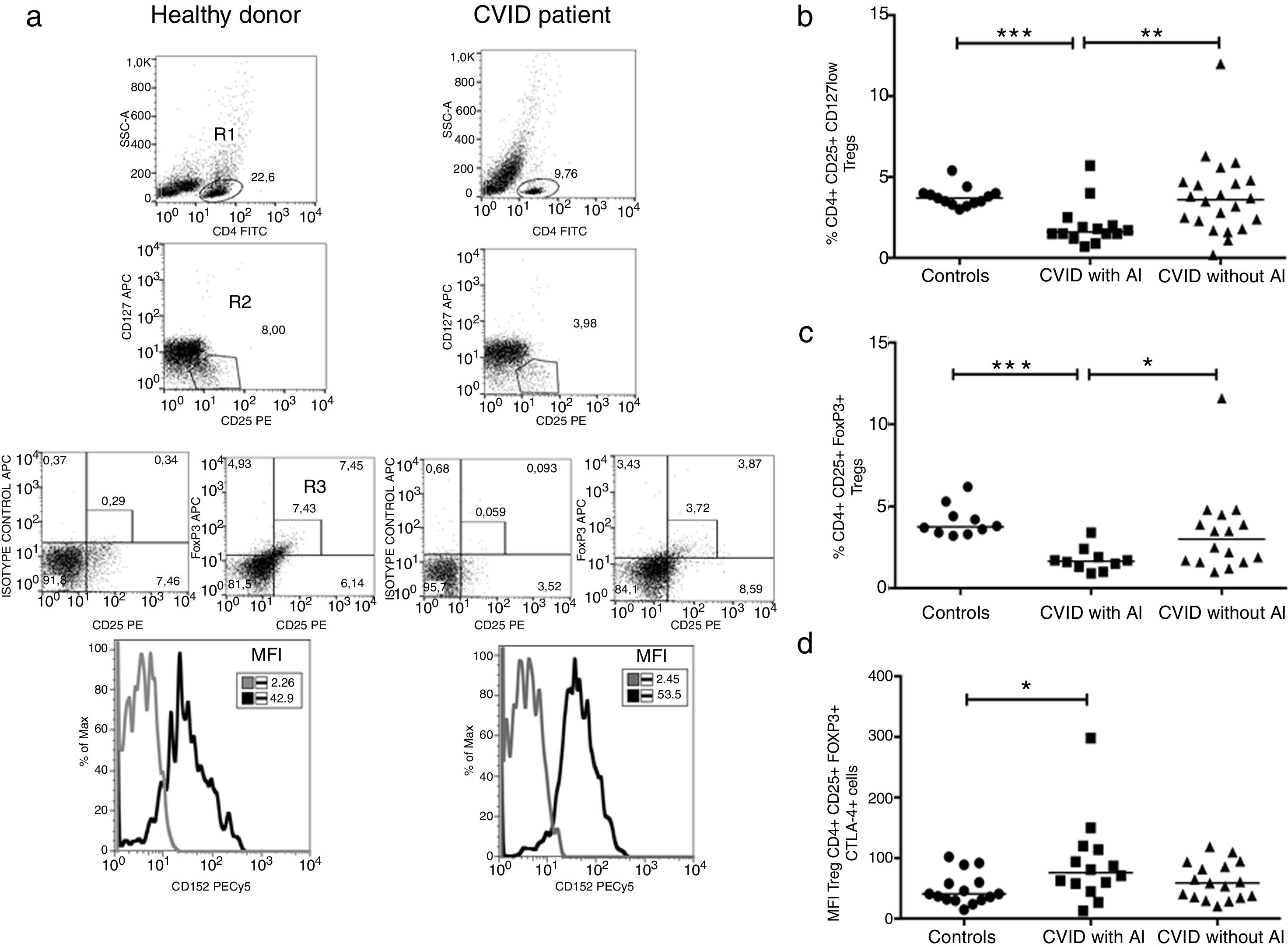
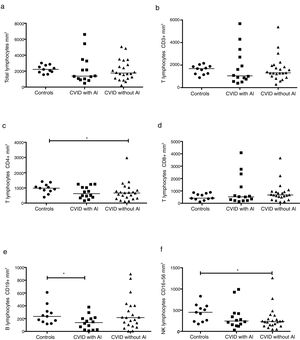
Tregs were identified by two strategies; they were defined as CD3+CD4+CD25+FoxP3+ or CD3+CD25+CD127low T cells or according to CTLA-4 expression (Fig. 2a). The frequency of CD4+CD25+FoxP3+ Tregs was significantly reduced in CVID patients with AI compared with CVID patients without AI and with healthy individuals (p=0.0219 and p=<0.0001, respectively) (Fig. 2b). Similar results were observed when Tregs were detected by CD3+CD25+CD127low staining (p=0.0057 and p=0.0005, respectively) (Fig. 2c). The median fluorescence intensity for CTLA-4 was analyzed in CD4+CD25+FoxP3+ Tregs. We observed that CTLA4 expression is slightly increased in CVID patients with AI (p=0.0283), whereas patients without AI show no differences compared with the healthy control group (Fig. 2d).
Comparison of Treg cells between healthy controls and CVID subgroups. (a) Gating strategy for the identification and of Treg cells and CTLA4 expression. CD4+ T lymphocytes were gated (R1), and CD25+CD127low cells (R2) were sub-gated; in a second strategy, CD25+Foxp3+ (R3) cells were determined from a CD4+ T lymphocyte (R1). CTLA4 expression (CD152) MFI was calculated from CD4+CD25+Foxp3+ (R3) gate (black line) and compared with the isotype control (gray lines). Treg cells and CTLA4 expression comparison between healthy controls and CVID subgroups. (b) CD4+CD25+CD127low and (c) CD4+CD25+Foxp3 T cells percentages comparison between controls (n=13) (●) and CVID with (n=14) (■) and without autoimmunity (AI) (n=22) (▴); (d) CTLA4 (CD152) MFI expression on Treg cells (CD4+CD25+Foxp3+). Median values are represented with a horizontal line. Statistical differences between patients and controls were compared using the Mann–Whitney U-test. (*) Significant, p=0.05; (**) very significant, p=0.01; and (***) highly significant, p=0.001. MFI, mean fluorescent intensity.
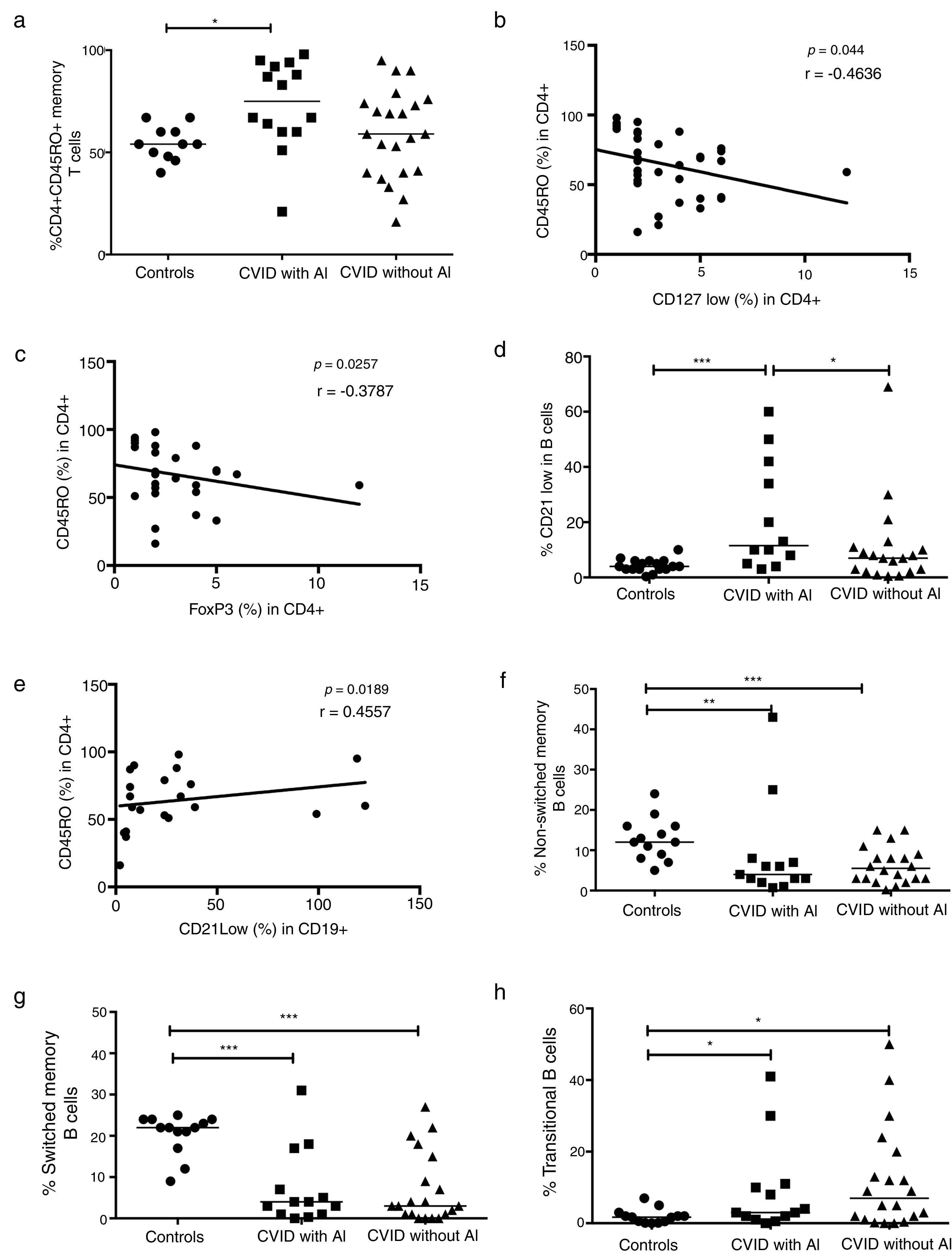
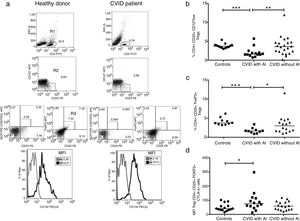
When the CD4+CD45RO+ memory T cells were determined in CVID patients, we observed that this subpopulation was significantly increased in patients with AI compared with healthy controls (p=0.0056) (Fig. 3a). Negative correlations between the frequencies of memory T cells and CD4+CD25+CD127low and CD4+CD25+FoxP3+ Tregs were also observed (p=0.0044, r=−0.4636; p=0.0257, r=−0.3787, respectively) (Fig. 3b and c). The frequency of CD21low B cells was significantly increased in CVID patients with AI compared with both the controls and the patients without AI (p=0.0006 and p=0.0500; respectively) (Fig. 3d). A positive association between CD4+CD45RO+ memory T cells and CD21low B cells was also observed (p=0.0189; r=0.4557). Additionally, we observed significant differences in B cell subpopulations between CVID patients with and without AI and the controls. The results showed reduced CD27+ non-switched memory B cells in CVID patients with and without AI (p=0.0057 and p=0.0009, respectively). Switched memory B cell frequencies remained low in both group of patients with and without AI (p=0.0003 and p=<0.0001, respectively) (Fig. 3f and g). Finally, transitional B cell frequencies were significantly increased in both groups of patients (p=0.0199, p=0.0447; respectively) (Fig. 3h).
Analysis of T and B cell subpopulations. Flow cytometric analysis of T and B cell subpopulation percentages from CVID patients with autoimmune diseases (AI) (n=14) (■) and CVID patients without AI (n=22) (▴). These groups were compared with controls (n=13) (●). (a) CD4+CD45RO+T cell percentage; (b) correlation analyses between CD4+CD45RO+ T cells and Tregs CD127low cells; (c) correlation analyses between CD4+CD45RO+ T cells and Tregs FoxP3+ cells; (d) CD21low B cells (CD19+CD21-CD38-); (e) correlation analyses between CD4+CD45RO+ T cells and CD21low B cells (CD19+CD21-CD38-); (f) non-switched memory B cells (CD19+CD27+IgD+); (g) switched memory B cells (CD19+CD27+IgD-); (h) transitional B cells (CD19+CD24+CD38+). Differences between patients and controls were compared using the Mann–Whitney U-test. (*) Significant, p=<0.05; (**) very significant, p=<0.01; and (***) highly significant, p=<0.001. AC=autoimmune cytopenia.
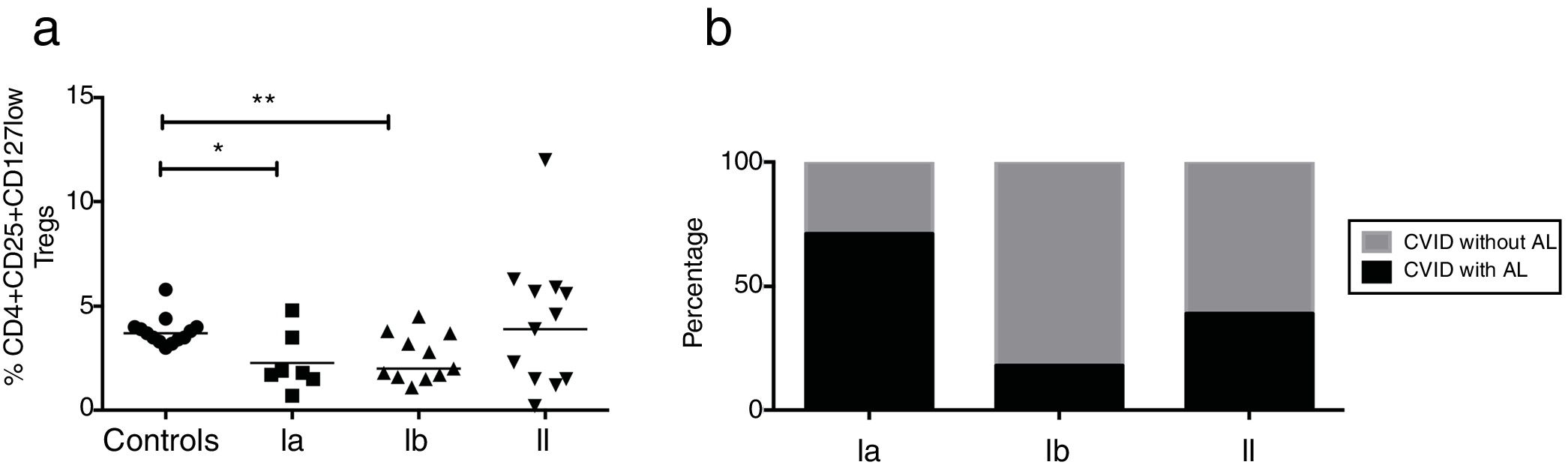
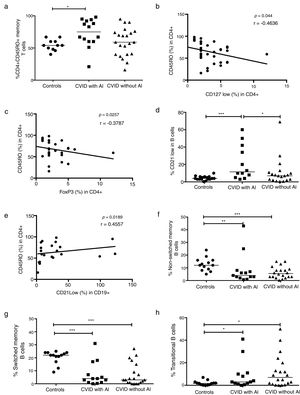
CVID patients were organized according to the Freiburg classification (27). Patients from groups Ia and Ib had reduced frequencies of CD4+CD25+CD127low Treg cells (p=0.0224 and p=0.0092, respectively); whereas group II patients did not present significant differences relative to healthy controls (Fig. 4a). We additionally determined the frequency of AI in each of the three groups of CVID patients. Among the 14 patients who presented with AI, five were in group Ia (71%), two were in group Ib (18%), and five were in group II (39%). Patients without AI (n=22) included two patients in group Ia (29%), nine in group Ib (82%), and nine in group II (69%) (Fig. 4b).
Expression of Treg cells in patients with CVID divided according to Freiburg classification. (a) Expression Tregs according to the CD4+CD25+CD127low phenotype in CVID patients divided according to Freiburg classification; Ia (n=7) (■); Ib (n=11) (▴); II (n=13) (▾) and controls (n=13) (●). (b) The frequency of each group of patients according to the Freiburg classification, in patients with or without autoimmune cytopenia. Differences between patients and controls were compared using the Mann–Whitney U-test. (*) Significant, p=<0.05; (**) very significant, p=<0.01; and (***) highly significant, p=<0.001.
CVID is considered the most common symptomatic PID. Clinically, CVID is a heterogeneous disorder where patients suffer from recurrent respiratory tract infections and B and T cells defect. B cell defects are common in all individuals with CVID, as evidenced by the reduced ability of B cells to differentiate into memory and plasma cells. Sera immunoglobulin levels in these patients are usually below normal ranges. T cell defects include reduced CD4 T cell numbers, impaired cytokine production, low percentages of Tregs and other deficiencies.5 Moreover, autoimmune manifestations occur in 20–25% of patients and include autoimmune hemolytic anemia, thrombocytopenia, rheumatoid arthritis, and pernicious anemia. In most patients, autoimmune manifestations occur before the diagnosis of CVID.28
In our cohort of CVID patients, 14 had AI (39%), the most common of which was ITP (31%). There was no significant difference between the infectious and non-infectious clinical manifestations among patients with and without AI. In other reports, autoimmune cytopenia is the most commonly occurring AI in CVID patients and occurs in 4–20% of patients who have some form of autoimmunity. An association of AI with granulomatous diseases and splenomegaly has been reported.29 However, in our cohort of patients, this association was not evident among patients with AI; this may be due to a very low percentage of our patients with splenomegaly and granulomatous diseases.
We compared the absolute numbers of T, B and NK cells among patients with validated CVID diagnoses. The patients were divided according to the presence or absence of AI and then compared with age-matched healthy controls. CD4+ T cells in CVID patients without AI were significantly lower compared with the control group. In a previous report, abnormalities in the number and function of CD4+ T cells in group Ib CVID patients (according to the Freiburg classification) were demonstrated.30 The B cells were diminished in patients with AI, and 40% of these patients were within group Ia. Group Ia of the Freiburg classification is characterized by a very low percentage of memory B cells, expanded CD21low cells and decreased peripheral blood B cells in both percentage and absolute numbers.27 On the other hand, NK cells were diminished in the group without AI. In a previous report, it was demonstrated that low NK cell counts correlate with invasive infections (pneumonia, septicemia, and meningitis) and granulomas but not with autoimmune diseases.31
We determined Tregs frequencies using two different phenotyping approaches, one with CD4+CD25+FoxP3+CTLA-4+ and the other with CD4+CD25+CD127low. We compared CVID patients with AI, CVID patients without AI and healthy controls; the low percentage of Tregs in CVID patients with AI correlated with the presence of autoimmunity. Fevang et al. first demonstrated a lower frequency of Tregs in patients with CVID7; other studies also demonstrated reduced Tregs in CVID patients with AI. The results of these studies showed that Tregs do play an essential role in the pathogenesis of CVID with a strong association with autoimmunity and splenomegaly.6,8,32 It has been suggested from murine and in vitro experiments using human cells that CTLA4 is essential for FoxP3-mediated immunosuppression.33 Although the patients with AI in our study had low Tregs frequencies, they had normal CTLA-4 expression. A key feature of CTLA-4 deficiency in humans is an impaired Tregs compartment. As Tregs express the highest levels of CTLA-4, it may be that deficiency in one allele is sufficient to compromise the high levels of expression required for adequate function.34 In the future, functional studies of Tregs should be conducted to identify patients with CTLA-4 deficiencies.
In our study, the CVID patients with AI had increased CD4+CD45RO+ memory T cell populations compared with healthy controls, and there was a negative correlation between memory CD4+ T cells and Tregs. It was also observed that CVID patients with AI had increased CD21low B cell populations that were significantly greater than those of the CVID patients without AI and the healthy controls; we also observed a positive correlation between the expansion of CD21low B cells and increased memory CD4+ T cells. CD45 is a protein tyrosine phosphatase and is expressed on all hematopoietic cells. CD45 can be expressed as one of several isoforms. CD45RA is a marker for naive T cells; after antigen experience, central and effector memory T cells increase expression of CD45RO.35
On the other hand, CD21, which is also known as complement receptor 2 (CDR2), is a type I membrane glycoprotein that binds split C3 molecules and forms a complex with CD19 and CD81 to act as a coreceptor. CD21low B cells show signs of the previous activation and express decreased amounts of homeostatic chemokine receptors but increased levels of an inflammatory chemokine receptor. The expansion of CD21low cells has been shown in autoimmune diseases, mainly SLE.14
Phenotypic and functional defects in CD4+ T cells in CVID patients have been reported during the last decade. These studies showed that the severity and prognosis of CVID are reflected in a parallel loss of naive CD4+ T cells.4,36 Few reports have demonstrated increased CD4+CD45RO T cells in CVID patients. In a study by Carbone et al., activated CD4+ T cells were more common in patients with suspected IgG hypercatabolism and patients with clinical complications including lymphoid proliferation, splenomegaly and autoimmune disease.37 Another study showed that CVID patients with autoimmune cytopenia had increased numbers of activated CD4+ T cells, decreased numbers of naïve T cells, and increased proportions of CD21low B cells.14,24 Our findings prove that T and B cell abnormalities in CVID are partially related to each other and also suggest that the coexistence of autoimmunity and immunodeficiency is a result of dysfunction in multiple immune pathways. The CVID patients within this cohort demonstrated impaired B cell differentiation into the memory compartment, inadequate class switch recombination and expanded transitional B cell populations, regardless of whether they had an autoimmune disease.
Finally, several clinical and immunological classifications have been proposed in an attempt to stratify and maybe even simplify the complex and heterogeneous phenotypes observed in CVID.27,38,39 We used the Freiburg classification, which is based on B cell phenotype. According to this schema, our patients in groups Ia and Ib had significantly lower percentages of Tregs compared with patients in group II and healthy controls. The frequency of autoimmune disease was higher in group Ia patients, which was in agreement with data reported in other patient cohorts. These results explain that abnormalities in peripheral blood B-cell subpopulations, such as those observed in group Ia and Ib patients, are accompanied by a significant reduction in the proportion of Tregs. Consequently, T cells may play a key role in the pathogenesis of CVID.
ConclusionIn conclusion, we have shown that Tregs are reduced in CVID patients compared with healthy controls and that this reduction is higher in patients with AI. The expansion of CD21low B cells in our patients with AI has also been previously reported. Regarding the Tregs abnormalities in CVID patients, it could be concluded that cellular dysregulation, including an increased memory CD4 T cell population, may be a consequence of lower numbers and reduced Tregs suppressive capacity, especially in patients with autoimmune manifestations. These observations suggest that the coexistence of autoimmunity and immunodeficiency is a result of dysfunction among multiple immune pathways.
Conflicts of interestThe authors declare no conflicts of interest.
The authors wish to thank Jimena Cerafino Martínez for technical assistance. This study was partially supported by grants from Consejo Nacional de Ciencia y Tecnología (CONACyT), project #CB-2016-256471 and #FOSISSS-161089.