Background and aims. Effective assessing the prognosis of patients with end-stage liver disease is always challenging. This study aimed to investigate the accuracy of different models in predicting short-term prognosis of patients with hepatitis B virus (HBV)-related acute-on-chronic liver failure (ACLF).
Material and methods. We retrospectively evaluated survival of a cohort of patients with at least 3-month follow up. The receiver-operating-characteristic curves (ROC) were drawn for Child-Turcotte-Pugh (CTP) classification, King’s College Hospital (KCH) criteria, model for end-stage liver disease (MELD), MELD combined with serum sodium (Na) concentration (MELDNa), integrated MELD (iMELD) and logistic regression model (LRM).
Results. Of the 273 eligible patients, 152 patients (55.7%) died within 3-month follow up. In cirrhotic patients (n = 101), the AUCs of LRM (0.851), MELDNa (0.849), iMELD (0.845) and MELD (0.840) were all significantly higher than those of KCH criteria (0.642) and CTP (0.625) (all p < 0.05), while the differences among LRM, MELD, MELDNa and iMELD were not significant, and the most predictive cutoff value was 0.5176 for LRM, 30 for MELDNa, 47.87 for iMELD and 29 for MELD, respectively. In non-cirrhotic patients (n = 172), the AUC of LRM (0.897) was significantly higher than that of MELDNa (0.776), iMELD (0.768), MELD (0.758), KCH criteria (0.647) and CTP (0.629), respectively (all p < 0.05), and the most predictive cutoff value for LRM was-0.3264.
Conclusions. LRM, MELD, MELDNa and iMELD are with similar accuracy in predicting the short-term prognosis of HBV-ACLF patients with liver cirrhosis, while LRM is superior to MELD, MELDNa and iMELD in predicting the short-term prognosis of HBV-ACLF patients without liver cirrhosis.
Chronic hepatitis B virus (HBV) infection is a global public health problem. There are about 350400 million persons infected worldwide.1 Abnormal liver function or even liver failure occurred in some patients during the long process of the chronic HBV infection.2 Hepatitis B virus (HBV)-related acute-on-chronic liver failure (ACLF) is characterized by acute deterioration in liver function due to the effects of the precipitating event such as infection/sepsis, reactivation of hepatitis B or hepatotoxic drugs in a patient on the basis of the chronic HBV infection, and accompanied by the clinical syndromes such as jaundice, coagulopathy, ascites and/or hepatic ence-phalopathy (HE), etc.3 ACLF leads to a poor prognosis and a high short-term mortality (50-90%).4 Nowadays, liver transplantation remains the most effective treatment for patients to sustain life, however it is hampered by lack of liver donors world-wide.5 It is important to timely, accurately and objectively assess the disease severity and short-term prognosis of the patients with HBV-ACLF in the face of a critical shortage of liver donors, and the emergent preparation of the liver transplantation for the patients based on the assessment will be helpful to improve the survival and the quality of life of the patients.
To evaluate the disease severity and assess the prognosis of patients with end-stage liver disease (ESLD) is a topic always challenging the clinician. Over the years, researchers have been looking for a prognostic model that could objectively and accurately reflect the disease severity and prognosis of patients with (ESLD), and various prognostic models or criteria of liver transplantation have been proposed. Child-Turcotte-Pugh (CTP) classification,6 King’s College Hospital (KCH) criteria,7 model for end-stage liver disease (MELD),8 MELD combined with serum sodium (Na) concentration (MELDNa),9 MELD with the incorporation of Na (MELD-Na),10 MELD to Na ratio index (MESO index)11 and integrated MELD (iMELD)12 are commonly used to assess the prognosis of the patients with ESLD. Among those models, CTP, MELD, MELDNa and MESO index are widely used to evaluate the prognosis of the patients with liver cirrhosis,13–18 while KCH criteria is mainly used to assess the disease severity of patients with acute liver failure (ALF).19,20 At present, someone also reported those models also could be applied in assessing the prognosis of the patients with ACLF;21–23 however the relevant literatures are comparatively few. Recently, a new logistic regression model (LRM) has been established and applied in predicting prognosis of HBV-ACLF patients.24 However, its prognostic accuracy still need to be validated in large-sample cohorts.
ObjectiveIn this study, our aim was to investigate the accuracy of the six models including CTP, KCH criteria, MELD, MELDNa, iMELD and LRM in predicting the short-term (3-month) prognosis of patients with HBV-ACLF.
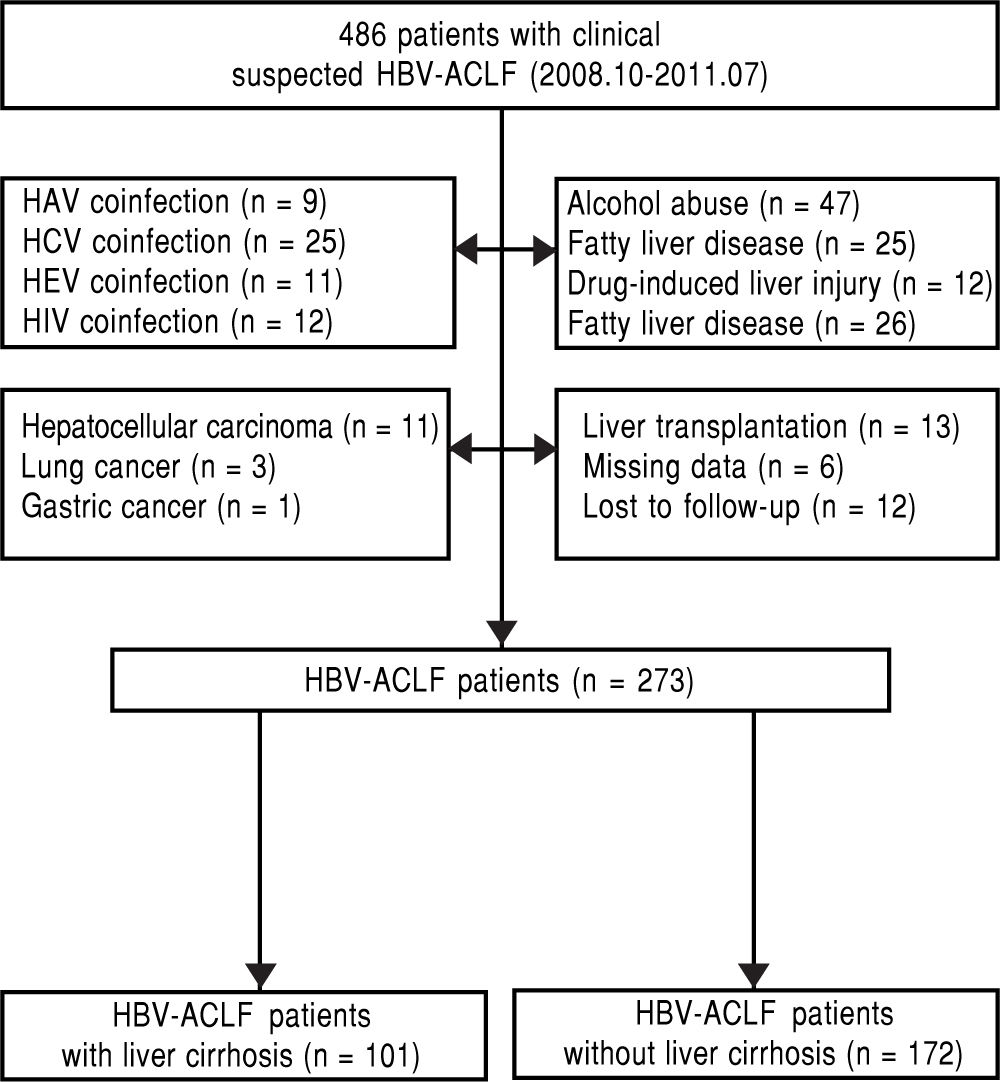
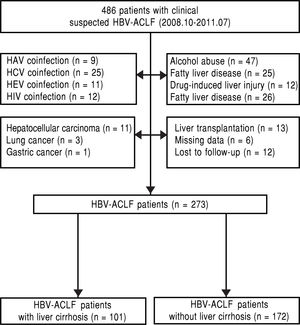
Material and MethodsPatientsThis is a retrospective single-center study on HBV-ACLF patients admitted to Department of Infectious Disease, West China Hospital of Sichuan University from October 2008 to July 2011. The inclusion criteria were defined as follows: chronic HBV infection (hepatitis B surface antigen positive for at least 6 months); ACLF; known the survival status at follow up after 3-month. Patient who met one of the following criteria was excluded: coexisting hepatitis A, C, E or human immunodeficiency virus (HIV) infection; alcohol abuse; drug-induced liver injury, autoimmune liver disease, or other causes that might lead to liver failure; past or current malignant tumors; liver transplantation; severe systemic or mental diseases (Figure 1).
Clinical diagnosis and definitionsHBV-ACLF was defined as an acute hepatic insult manifesting as jaundice (serum bilirubin ≥ 5 x upper limit of normal), coagulopathy [international normalized ratio (INR) ≥ 1.5 or prothrombin activity [PTA] < 40%) and complicated within 4 weeks by ascites and/or HE in a patient with chronic HBV infection according to consensus recommendations of the Asian Pacific Association for the Study of the Liver (APASL) in 2009.3
The diagnosis of liver cirrhosis was mainly based on the computed tomography or ultrasonography findings of a nodular liver surface, blunt liver edge, coarsened echogenicity of liver parenchyma, enlarged spleen and combined the following findings (1) medical history (2) the presence of hepatic stigmata, spider angiomas or splenomegaly on physical examination; (3) laboratory abnormalities (e.g. decreased serum albumin, increased serum globulin levels, low platelet count).
HE was classified into I-IV stages according the neuropsychiatric symptoms and physical examina-tion.25
Study designThe HBV-ACLF patients were divided into two groups according to whether the patients had liver cirrhosis or not. Baseline clinical and laboratory data of all the patients (i.e. age, serum bilirubin, creatinine, Na, PT, INR) were collected when patients were diagnosed with HBV-ACLF after the admission and these data were retrospectively analyzed in this study.
All patients were followed up for at least 3-month, and the discharged patients were followed up by telephone. The prognosis of every patient (survival more than 3 months, liver transplantation or death) was recorded. All the patients underwent combined medical treatment.
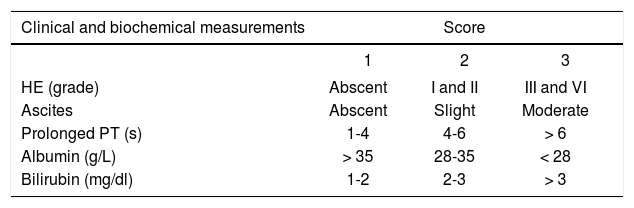
All prognostic models scores were calculated based on clinical and laboratory results obtained on the day when patients were diagnosed with HBV-as-sociated ACLF after the admission. The scoring criteria of the prognostic models were listed in tables 1 and 2.
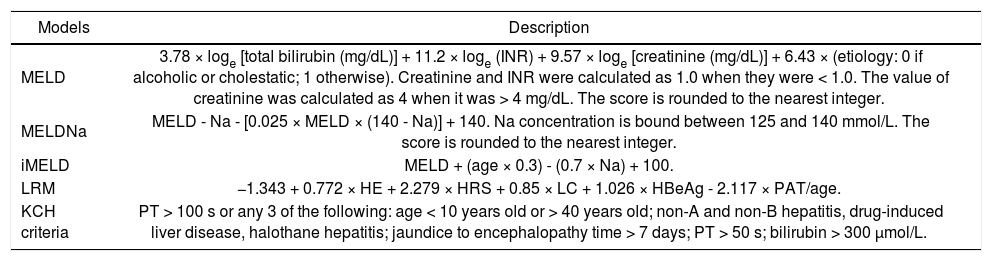
The scoring criteria of MELD, MELDNa, iMELD and KCH criteria.
| Models | Description |
|---|---|
| MELD | 3.78 × loge [total bilirubin (mg/dL)] + 11.2 × loge (INR) + 9.57 × loge [creatinine (mg/dL)] + 6.43 × (etiology: 0 if alcoholic or cholestatic; 1 otherwise). Creatinine and INR were calculated as 1.0 when they were < 1.0. The value of creatinine was calculated as 4 when it was > 4 mg/dL. The score is rounded to the nearest integer. |
| MELDNa | MELD - Na - [0.025 × MELD × (140 - Na)] + 140. Na concentration is bound between 125 and 140 mmol/L. The score is rounded to the nearest integer. |
| iMELD | MELD + (age × 0.3) - (0.7 × Na) + 100. |
| LRM | −1.343 + 0.772 × HE + 2.279 × HRS + 0.85 × LC + 1.026 × HBeAg - 2.117 × PAT/age. |
| KCH criteria | PT > 100 s or any 3 of the following: age < 10 years old or > 40 years old; non-A and non-B hepatitis, drug-induced liver disease, halothane hepatitis; jaundice to encephalopathy time > 7 days; PT > 50 s; bilirubin > 300 μmol/L. |
The study protocol was conducted in accordance with the standards of the Declaration of Helsinki and current ethical guidelines.
Data analysisStatistical analyses were performed using Med-Calc statistical software (MedCalc Software, Ma-riakerke, Belgium) and SPSS software version16.0 (SPSS Inc., Chicago, USA). Data expressed as mean ± standard deviation (SD) or median and interquartile range for continuous variables, and frequency with percentage for categorical variables. Continuous variables were compared by Student’s t-test or Mann-Whitney U test. Categorical variables were compared by Pearson Chi-squared test.
Receiver-operating-characteristic (ROC) curves were used to measure the performance of the models in predicting 3-month mortality of patients with HBV-ACLF, and validity of the model was measured by means of the area under the receiver-operating-characteristic curve (AUC). For a prognostic model, the AUC may ranges from 0 to 1. An AUC between 0.8 and 0.9 indicates excellent diagnostic accuracy and > 0.7 is generally considered useful, while an AUC of 0.9 or greater is seldom seen,8 and the comparison of AUCs was done with the method of Hanley & McNeil. The sensitivity (SS), specificity (SP), positive predictive value (PPV) and negative predictive value (NPV) were calculated according to the most predictive cutoff that had the best discriminative ability to predict mortality. For all analyses, a p value < 0.05 was considered statistically significant.
ResultsThe basic clinical features of the 273 eligible patientsA total of 273 eligible patients (251 male and 22 female, age ranged from 15 to 78 years, mean age: 40.8 ± 10.9 years) were recruited, and 101 patients had evidence of liver cirrhosis. Of those patients with HBV-ACLF, the most common complication was ascites (249 patients; 91.2%), followed by HE (102 patients; 37.4%), and HRS (84 patients; 30.8%). The overall 3-month mortality was 55.7% (152/273), and the majority of them died of the complications of ACLF such as HE, upper gastrointestinal bleeding, hepatorenal syndrome (HRS) and multi-organ failure, etc. The morality rate of HBV-ACLF patients with liver cirrhosis (60.4%) was higher than that of the HBV-ACLF patients without liver cirrhosis (52.9%), however there was no significant difference (p > 0.05).
Clinical and biochemical characteristics of the 273 eligible patients subdivided according to 3-month survivalThe patients were further divided into the survival and death groups according to the 3-month follow up results. The characteristics of the patients who survived and who died are shown in table 3.
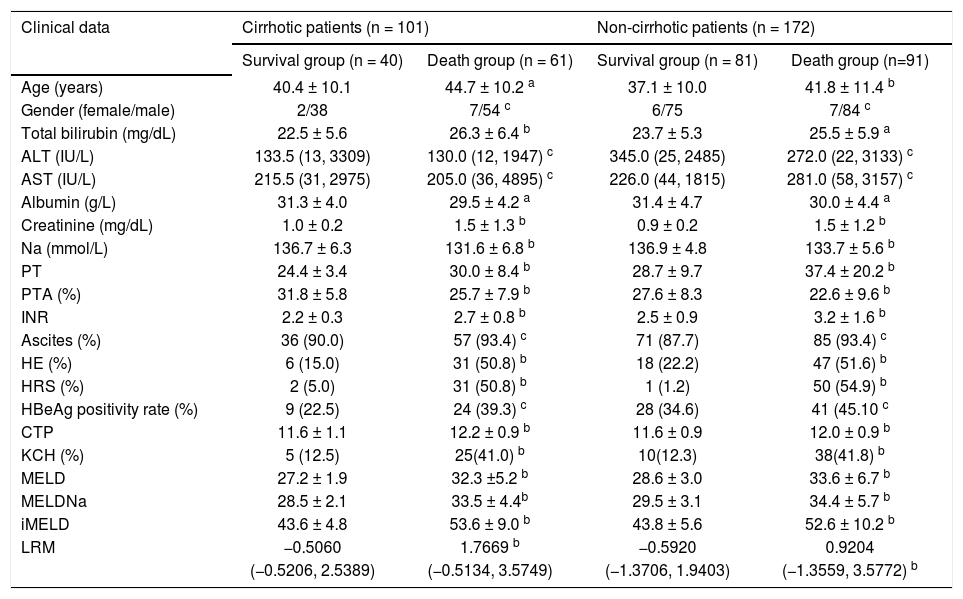
Clinical and biochemical characteristics of the 273 HBV-ACLF patients subdivided according to 3-month survival.
| Clinical data | Cirrhotic patients (n = 101) | Non-cirrhotic patients (n = 172) | ||
|---|---|---|---|---|
| Survival group (n = 40) | Death group (n = 61) | Survival group (n = 81) | Death group (n=91) | |
| Age (years) | 40.4 ± 10.1 | 44.7 ± 10.2 a | 37.1 ± 10.0 | 41.8 ± 11.4 b |
| Gender (female/male) | 2/38 | 7/54 c | 6/75 | 7/84 c |
| Total bilirubin (mg/dL) | 22.5 ± 5.6 | 26.3 ± 6.4 b | 23.7 ± 5.3 | 25.5 ± 5.9 a |
| ALT (IU/L) | 133.5 (13, 3309) | 130.0 (12, 1947) c | 345.0 (25, 2485) | 272.0 (22, 3133) c |
| AST (IU/L) | 215.5 (31, 2975) | 205.0 (36, 4895) c | 226.0 (44, 1815) | 281.0 (58, 3157) c |
| Albumin (g/L) | 31.3 ± 4.0 | 29.5 ± 4.2 a | 31.4 ± 4.7 | 30.0 ± 4.4 a |
| Creatinine (mg/dL) | 1.0 ± 0.2 | 1.5 ± 1.3 b | 0.9 ± 0.2 | 1.5 ± 1.2 b |
| Na (mmol/L) | 136.7 ± 6.3 | 131.6 ± 6.8 b | 136.9 ± 4.8 | 133.7 ± 5.6 b |
| PT | 24.4 ± 3.4 | 30.0 ± 8.4 b | 28.7 ± 9.7 | 37.4 ± 20.2 b |
| PTA (%) | 31.8 ± 5.8 | 25.7 ± 7.9 b | 27.6 ± 8.3 | 22.6 ± 9.6 b |
| INR | 2.2 ± 0.3 | 2.7 ± 0.8 b | 2.5 ± 0.9 | 3.2 ± 1.6 b |
| Ascites (%) | 36 (90.0) | 57 (93.4) c | 71 (87.7) | 85 (93.4) c |
| HE (%) | 6 (15.0) | 31 (50.8) b | 18 (22.2) | 47 (51.6) b |
| HRS (%) | 2 (5.0) | 31 (50.8) b | 1 (1.2) | 50 (54.9) b |
| HBeAg positivity rate (%) | 9 (22.5) | 24 (39.3) c | 28 (34.6) | 41 (45.10 c |
| CTP | 11.6 ± 1.1 | 12.2 ± 0.9 b | 11.6 ± 0.9 | 12.0 ± 0.9 b |
| KCH (%) | 5 (12.5) | 25(41.0) b | 10(12.3) | 38(41.8) b |
| MELD | 27.2 ± 1.9 | 32.3 ±5.2 b | 28.6 ± 3.0 | 33.6 ± 6.7 b |
| MELDNa | 28.5 ± 2.1 | 33.5 ± 4.4b | 29.5 ± 3.1 | 34.4 ± 5.7 b |
| iMELD | 43.6 ± 4.8 | 53.6 ± 9.0 b | 43.8 ± 5.6 | 52.6 ± 10.2 b |
| LRM | −0.5060 | 1.7669 b | −0.5920 | 0.9204 |
| (−0.5206, 2.5389) | (−0.5134, 3.5749) | (−1.3706, 1.9403) | (−1.3559, 3.5772) b | |
a
p < 0.05.
bp < 0.01.
cp > 0.05 vs. survival group.
In the cirrhotic patients:
- •
The death group had significantly higher age (44.7 ± 10.2 vs. 40.4 ± 10.1).
- •
Total bilirubin (26.3 ± 6.4 vs. 22.5 ± 5.6).
- •
Creatinine (1.5 ± 1.3 vs. 1.0 ± 0.2).
- •
PT (30.0 ± 8.4 vs. 24.4 ± 3.4).
- •
INR (2.7 ± 0.8 vs. 2.2 ± 0.3).
- •
CTP score (12.2 ± 0.9 vs. 11.6 ± 1.1).
- •
MELD score (32.3 ± 5.2 vs. 27.2 ± 1.9).
- •
MELDNa score (33.5 ± 4.4 vs. 28.5 ± 2.1).
- •
iMELD score (53.6 ± 9.0 vs. 43.6 ± 4.8), and
- •
LRM score (1.7669 vs. −0.5060).
And rate of patients met the KCH criteria (41.0 vs. 12.5%) than the survival group (all p < 0.05), while the death group had significantly lower Na (131.6 ± 6.8 vs. 136.7 ± 6.3) and PTA (0.257 ± 0.079 vs. 0.318 ± 0.058) (all p < 0.05). However, AST, ALT, and HBeAg positivity rate did not differ statistically between the survival group and death group.
In the non-cirrhotic patients:
- •
The death group had significantly higher age (41.8 ± 11.4 vs. 37.1 ± 10.0).
- •
Total bilirubin (25.5 ± 5.9 vs. 23.7 ± 5.3).
- •
Creatinine (1.5 ± 1.2 vs. 0.9 ± 0.2).
- •
PT (37.4 ± 20.2 vs. 28.7 ± 9.7).
- •
INR (3.2 ± 1.6 vs. 2.5 ± 0.9).
- •
CTP score (12.0 ± 0.9 vs. 11.6 ± 0.9).
- •
MELD score (33.6 ± 6.7 vs. 28.6 ± 3.0).
- •
MELDNa score (34.4 ± 5.7 vs. 29.5 ± 3.1).
- •
iMELD score (52.6 ± 10.2 vs. 43.8 ± 5.6), and
- •
LRM score (0.9204 vs. −0.5920).
And rate of patients met the KCH criteria (41.8 vs. 12.3%) than the survival group (all p < 0.05), while the death group had significantly lower Na (133.7 ± 5.6 vs. 136.9 ± 4.8) and PTA (0.226 ± 0.096 vs. 0.276 ± 0.083) (all p < 0.05). However, AST, ALT and HBeAg positivity rate did not differ statistically between the survival group and death group.
Predictive accuracy of the prognostic modelsThe AUC was:
- •
0.851 (95%CI: 0.766-0.914) for LRM.
- •
0.849 (95%CI: 0.764-0.912) for MELDNa.
- •
0.845 (95%CI: 0.760-0.910) for iMELD.
- •
0.840 (95%CI: 0.753-0.905) for MELD.
- •
0.642 (95%CI: 0.541-0.735) for KCH criteria, and
- •
0.625 (95%CI: 0.523-0.719) for CTP in evaluating the short-term prognosis of the HBV-ACLF patients with liver cirrhosis, respectively.
The AUCs of LRM, MELD, MELDNa and iMELD were all significantly higher than those of CTP and KCH criteria (all p < 0.05), while the differences among LRM, MELD, MELDNa and iMELD were not significant.
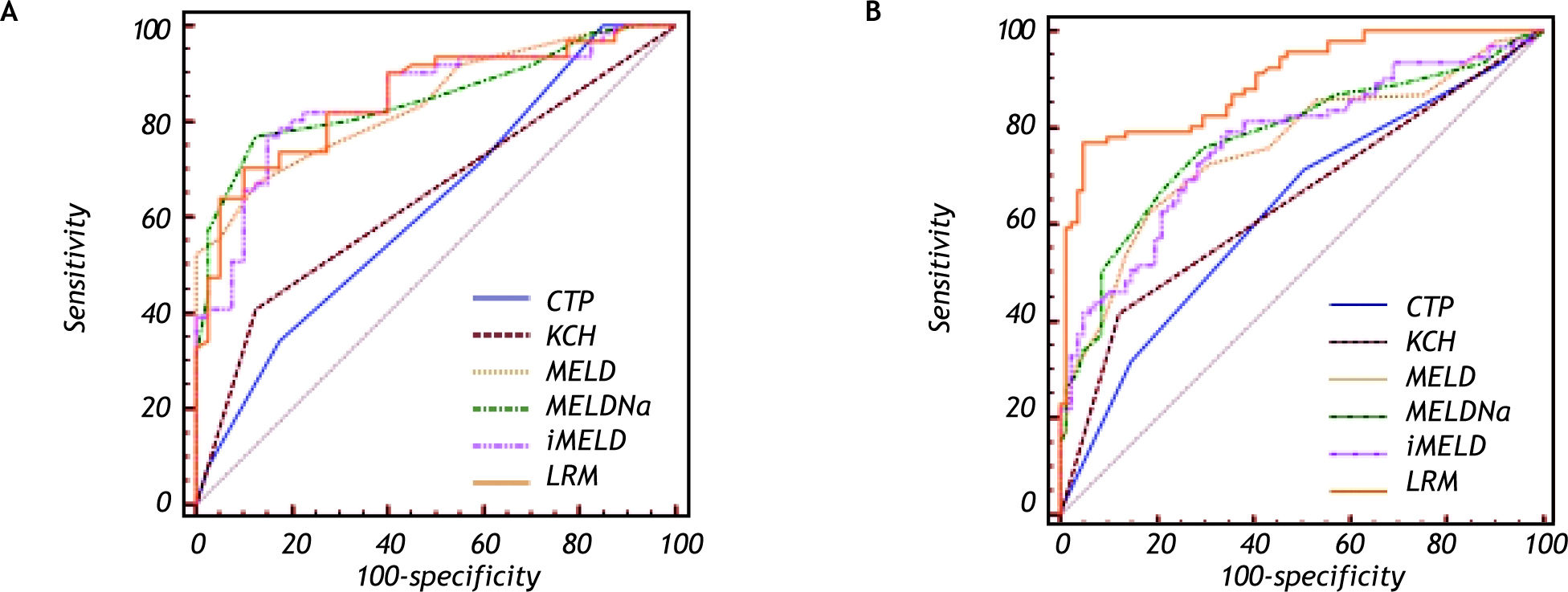
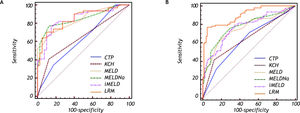
The best predictive cutoff value was (Table 4, Figure 2A):
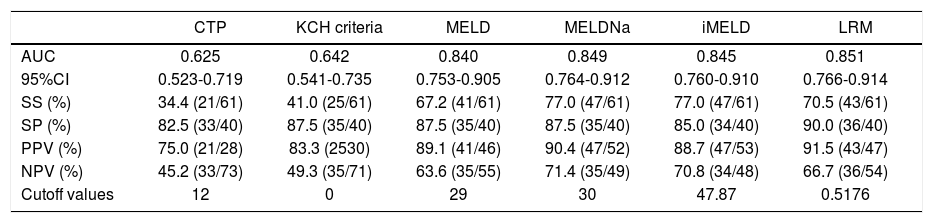
Predictive accuracy of the six models in predicting HBV-ACLF patients with liver cirrhosis.
| CTP | KCH criteria | MELD | MELDNa | iMELD | LRM | |
|---|---|---|---|---|---|---|
| AUC | 0.625 | 0.642 | 0.840 | 0.849 | 0.845 | 0.851 |
| 95%CI | 0.523-0.719 | 0.541-0.735 | 0.753-0.905 | 0.764-0.912 | 0.760-0.910 | 0.766-0.914 |
| SS (%) | 34.4 (21/61) | 41.0 (25/61) | 67.2 (41/61) | 77.0 (47/61) | 77.0 (47/61) | 70.5 (43/61) |
| SP (%) | 82.5 (33/40) | 87.5 (35/40) | 87.5 (35/40) | 87.5 (35/40) | 85.0 (34/40) | 90.0 (36/40) |
| PPV (%) | 75.0 (21/28) | 83.3 (2530) | 89.1 (41/46) | 90.4 (47/52) | 88.7 (47/53) | 91.5 (43/47) |
| NPV (%) | 45.2 (33/73) | 49.3 (35/71) | 63.6 (35/55) | 71.4 (35/49) | 70.8 (34/48) | 66.7 (36/54) |
| Cutoff values | 12 | 0 | 29 | 30 | 47.87 | 0.5176 |
In cirrhotic patients: the AUCs of LRM, MELD, MELDNa and iMELD were all significantly higher than those of CTP and KCH criteria (all p < 0.05), while the differences among LRM, MELD, MELDNa and iMELD were not significant (all p < 0.05).
- •
0.5176 (SS = 70.5%; SP = 90.0%; PPV =91.5%; NPV = 66.7%) for LRM.
- •
30 (SS = 77.0%; SP = 87.5%; PPV = 90.4%; NPV =71.4%) for MELDNa.
- •
47.87 (SS = 77.0%; SP = 85.0%; PPV = 88.7%; NPV = 70.8%) for iMELD, and
- •
29 (SS = 67.2%; SP = 87.5%; PPV =89.1%; NPV = 63.6%) for MELD, respectively.
The AUC was:
- •
0.897 (95%CI: 0.841-0.938) for LRM.
- •
0.776 (95%CI: 0.707-0.836) for MELDNa.
- •
0.768 (95%CI: 0.689-0.829) for iMELD.
- •
0.758 (95%CI: 0.687-0.820) for MELD.
- •
0.647 (95%CI: 0.571-0.718) for KCH criteria, and
- •
0.629 (95%CI: 0.552-0.701) for CTP in evaluating the short-term prognosis of the HBV-ACLF patients without liver cirrhosis, respectively.
The AUC of LRM was significantly higher than that of MELDNa, iMELD, MELD, KCH criteria and CTP, respectively (all p < 0.05) and the best predictive cutoff value for LRM was-0.3264 (SS = 76.9%; SP = 95.1%; PPV = 94.6%; NPV = 78.6%) for LRM (Table 5, Figure 2B).
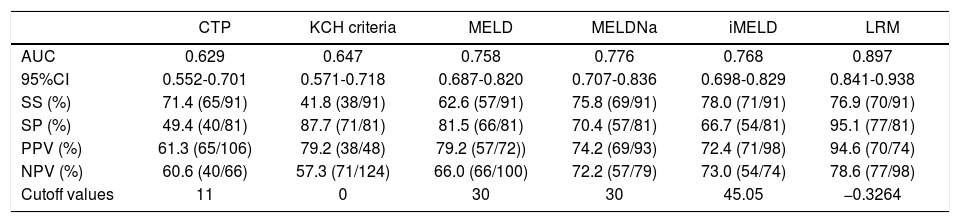
Predictive accuracy of the six models in predicting HBV-ACLF patients without liver cirrhosis.
| CTP | KCH criteria | MELD | MELDNa | iMELD | LRM | |
|---|---|---|---|---|---|---|
| AUC | 0.629 | 0.647 | 0.758 | 0.776 | 0.768 | 0.897 |
| 95%CI | 0.552-0.701 | 0.571-0.718 | 0.687-0.820 | 0.707-0.836 | 0.698-0.829 | 0.841-0.938 |
| SS (%) | 71.4 (65/91) | 41.8 (38/91) | 62.6 (57/91) | 75.8 (69/91) | 78.0 (71/91) | 76.9 (70/91) |
| SP (%) | 49.4 (40/81) | 87.7 (71/81) | 81.5 (66/81) | 70.4 (57/81) | 66.7 (54/81) | 95.1 (77/81) |
| PPV (%) | 61.3 (65/106) | 79.2 (38/48) | 79.2 (57/72)) | 74.2 (69/93) | 72.4 (71/98) | 94.6 (70/74) |
| NPV (%) | 60.6 (40/66) | 57.3 (71/124) | 66.0 (66/100) | 72.2 (57/79) | 73.0 (54/74) | 78.6 (77/98) |
| Cutoff values | 11 | 0 | 30 | 30 | 45.05 | −0.3264 |
In non-cirrhotic patients: the AUC of LRM was significantly higher than that of CTP, KCH criteria, MELD, MELDNa and iMELD, respectively (all p < 0.05).
HBV-ACLF is one of the most dangerous ESLDs with a poor prognosis and high short-term mortality. An objective, accurate, simple and convenient prognostic model can help the doctors to evaluate the disease severity and short-term prognosis and select an appropriate treatment program such as combined medical treatment or liver transplantation.
The CTP classification was first proposed by Child and Turoctte and then modified by Pugh.6 Because the five variables (bilirubin, albumin, PT, as-cites and HE) are easy to obtain and calculation of the CTP scores is simple, it has become the most common model in assessing the reserve liver function capacity and the prognosis of patients with liver cirrhosis.26,27 However, over many years, the limitations came out gradually during the application, such as subjective judgments on HE and ascites and limited discriminant ability.28 In order to overcome these limitations of CTP, Kamath, et al.8 proposed a new scoring system (MELD) which was initially developed and validated to assess the short-term prognosis of cirrhotic patients undergoing the transjugular intrahepatic portosystemic shunt (TIPS). Compared with the CTP, The variables (bili-rubin, creatinine and INR) of the MELD scoring system were objective, and the renal function was taken as an independent factor influencing the prognosis of patients with liver disease. Meanwhile, MELD scores were continuous, which could distinguish the patient’s disease severity better. MELD also could reflect the pathological changes of hyper-bilirubinemia, coagulation disorders, kidney failure and other pathological changes better. In recent year, MELD has been widely applied to predict mortality across a broad spectrum of liver diseases in many studies. Since February 2002, MELD has been a tool used to predict mortality risk and to assess disease severity so as to determine organ allocation priorities by the United Network for Organ Sharing (UNOS) in the United States.29 However, there is still controversy about whether MELD is better than CTP.30–32
Na has been considered to be an important independent factor that affects the short-term prognosis of patients with ESLD. Hyponatremia is associated with complications of liver cirrhosis, such as ascites and HRS.10 Kim, et al.9combined the MELD and Na, and proposed the new scoring system (MELD-Na) on the basis of MELD, and showed that it could enhance the predictive accuracy of MELD. iMELD was generated on the basis of 3 clinical variables (MELD, age and Na), and the predictive accuracy of the iMELD was reported in another independent report, which showed that it had better predictive accuracy than that of the MELD in evaluating the prognosis of patients with liver cirrhosis.13 LRM was developed to assess the short-term prognosis of HBV-ACLF patients and Zheng, et al.24 reported that LRM was with greater accuracy than MELD and CTP in predicting the prognosis of patients with HBV-ACLF, however its usefulness still need to be validated in other cohorts. The KCH criteria are mainly used for listing patients with ALF for liver transplantation, however KCH criteria are rarely used to predict the prognosis of patients with ACLF. In our study, we compare the accuracy of the six models in predicting the short-term prognosis of 273 patients with HBV-ACLF. Moreover, whether the incorporation of age and /or Na (MELDNa and iMELD) to MELD could enhance accuracy of MELD in predicting the patients with HBV-ACLF is also investigated. Firstly, LRM, MELD, MELDNa and iMELD are all superior to CTP and KCH criteria in predicting the short-term prognosis of the HBV-ACLF patients. LRM, MELD, MELDNa and iMELD are with similar predictive accuracy in predicting the short-term prognosis of HBV-ACLF patients with liver cirrhosis, while LRM is superior to MELD, MELDNa and iMELD in predicting the short-term prognosis of HBV-ACLF patients without liver cirrhosis. Secondly, our result also shows that incorporation age and/or Na into the MELD (MELDNa and iMELD) in the AUC analysis did not improve the predictive accuracy of the MELD in patients with HBV-ACLF. Thirdly, From the ROC analysis, we have determined the best predictive cutoffs for the models in predicting the short-term prognosis of the HBV-ACLF patients. MELD, MELDNa, iMELD and LRM had a consistently high SS (67.2-77.0%), SP (85.0-90.0%), PPV (88.7-91.5%) and NPV (63.6-71.4%) in predicting the short-term prognosis of HBV-ACLF patients with liver cirrhosis and LRM had a consistently high SS (76.9%), SP (95.1%), PPV (94.6%) and NPV (78.6%) in predicting the short-term prognosis of HBV-ACLF patients without liver cirrhosis. Therefore, it might be helpful to doctors to evaluate the disease severity and prognosis of patients with HBV-ACLF, for example, when a HBV-ACLF patient with liver cirrhosis had a iMELD score 40.0 (< 47.87), the patient would have a 70.8% chance of survival in 3 months without liver transplantation, while with a iMELD score 50 (> 47.87), the patient would have a 88.7% chance of death in 3 months without liver transplantation. So according to the assessment, it might be helpful to doctors to choose an appropriate treatment program (for example, combined medical treatment, artificial liver treatment or liver transplantation). In addition, the morality rate of HBV-ACLF patients with liver cirrhosis (60.4%) was higher than that of the HBV-ACLF patients without liver cirrhosis (52.9%), however there was no significant difference. It indicated that the liver cirrhosis might not affect the short-term prognosis of patients with HBV-ACLF.
Although the parameters in MELD, MELDNa and iMELD were relatively objective, which excluded the potential deviation due to the subjective judgments such as HE and ascites in CTP, we should also realize the potential limitations of the three models. For example, some important factors (i.e. HE, HRS, upper gastrointestinal bleeding) affecting the prognosis of patients with HBV-ACLF have not been taken into consideration in the three mo-dels.24,33,34 In addition, treatment such as diuretics or artificial liver treatment would affect the Na, bilirubin and creatinine. Therefore, in practice, we might consider these factors.
ConclusionLRM, MELD, MELDNa and iMELD are with similar predictive accuracy in predicting the short-term prognosis of HBV-ACLF patients with liver cirrhosis, while LRM is superior to MELD, MELD-Na and iMELD in predicting the short-term prognosis of HBV-ACLF patients without liver cirrhosis. However, our findings also need to be verified in large sample size clinical trials in future.
Abbreviations- •
ACLF: acute-on-chronic liver failure.
- •
HBV: hepatitis B virus.
- •
HCV: hepatitis C virus.
- •
HEV: hepatitis E virus.
- •
HIV: human immunodeficiency virus.
- •
ESLD: end-stage liver disease.
- •
CTP: Child-Turcotte-Pugh.
- •
KCH criteria: King’s College Hospital criteria.
- •
MELD: model for end-stage liver disease.
- •
MELDNa: MELD combined with serum sodium concentration.
- •
iMELD: integrated MELD.
- •
MESO index: MELD to serum sodium ratio index.
- •
LRM: logistic regression model.
- •
ROC: receiver-operating-characteristic curve.
- •
AUC: area under the receiver-operating-characteristic curve.
- •
INR: international normalized ratio.
- •
SS: sensitivity.
- •
SP: specificity.
- •
PPV: positive predictive value.
- •
NPV: negative predictive value.
- •
PT: prothrombin time.
- •
PTA: prothrombin activity.
- •
CI: confidence interval.
- •
TIPS: transjugular intrahepatic portosystemic shunt.
- •
LC: liver cirrhosis.
- •
HE: hepatic encephalopathy.
- •
HBeAg: hepatitis B e antigen.
- •
HRS: hepatorenal syndrome.
None.
AcknowledgementsThis study was supported by National Natural Science Foundation of China (No. 30972622) and National science and Technology major project of China (No. 2012ZX10002007 and 2008ZX10002-006). We thank all patients and his families who participated in this study.