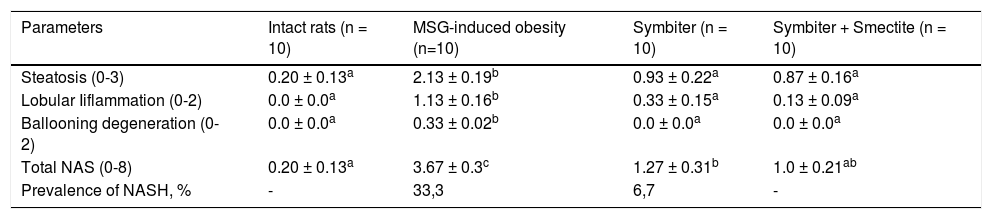Introduction and aim. Today probiotics have been suggested as a treatment for the prevention of non-alcoholic fatty liver disease (NAFLD). Smectite is a natural silicate that binds to digestive mucous and has the ability to bind endo- and exotoxins. The present study was designed to determine whether probiotics plus smectite is superior to probiotic alone on the monosodium glutamate (MSG) induced NAFLD model in rats.
Materials and methods. We included 60 rats divided into 4 groups 15 animals in each. Rats of group I were intact. Newborns rats of groups II-IV were injected with MSG. The III (Symbiter) group received 2.5 ml/kg of multiprobiotic “Symbiter” containing concentrated biomass of 14 probiotic bacteria genera. The IV (Symbiter+Smectite) groups received “Symbiter Forte” combination of probiotic biomass with smectite gel (250 mg).
Results. In both interventional groups reduction of total NAS score as compared to MSG-obesity was observed. Indeed similar values of steatosis score (0.93 ± 0.22 vs. 0.87 ± 0.16) in both treatment groups, we observed that lower total score for Symbiter+ Smectite are associated with more pronounced reduction of lobular inflammation (0.13 ± 0.09 vs. 0.33 ± 0.15) as compared to administration of probiotic alone. This data accompanied with significant reduction of IL-1 and restoration of IL-10 between these 2 groups.
Conclusions. Additional to alive probiotic administration of smectite gel due to his absorbent activity and mucus layer stabilization properties can impact on synergistic enhancement of single effect which manifested with reduction of lobular inflammation and at list partly steatohepatitis prevention.
Non-alcoholic fatty liver disease (NAFLD) is currently a leading cause of chronic liver disease1,2 affecting approximately 20-40% of adults in developed countries3 and recognized as the accumulation of lipids within the hepa-tocytes exceeding 5% of liver weight in the absence of excessive alcohol intake and secondary causes of liver diseases.4 NAFLD ranges from simple steatosis to non-alcoholic steatohepatitis (NASH) that can have different degrees of fibrosis and progress to liver cirrhosis and hepatocellular carcinoma,5 which has resulted in significant health concerns such as morbidity, mortality, and liver transplants.6
Current conception of NAFLD pathogenesis evolved from “two hit” 7 to recent “multiple hit” theory.8 According to formerly proposed two hit model insulin resistance acts as first hit and can promote intra-hepatic fat accumulation and lipotoxicity; second, development of cellular insults which caused by oxidative stress, lipid oxidation, chronic inflammation, apoptosis and fibrogenesis that determine the progression of the disease.7,9
As for nowadays the “two hit” hypothesis is outdated, as it does not fully account for several molecular and metabolic changes that occur in NAFLD development. The “multiple hit” hypothesis considers multiple insults acting together on genetically predisposed subjects to induce NAFLD and provides a more accurate explanation of NAFLD pathogenesis. Such hits include insulin resistance, disbalance of adipose tissue hormonal activity, low-grade chronic systemic inflammation, increased intestinal permeability with subsequent “metabolic endotoxemia”, nutritional factors, gut microbiota and genetic and epigenetic factors.8,10
Among multiple hits, increasing clinical and experimental evidence indicates that alteration of gut-liver axis is implicated in NAFLD onset and progression, and the gut microbiota has been recognized as the key player in the gut-liver cross talk.11,12 Thus, the modification of intestinal bacterial flora by prebiotics and specific probiotics has been proposed as a therapeutic approach for the treatment and/or prevention of NAFLD.13,14
Smectite is a natural silicate clay belonging to the dioctahedral smectite class and consists of a double aluminium and magnesium silicate arranged in parallel leaflets.15 Smectite has recently been recommended by several reputable organization for treatment of acute diarrhea.16,17
It is well known that “metabolic endotoxemia” due to “multiple hit” theory is one of the most powerful triggers strongly associated with NAFLD development. Metabolic endotoxemia is defined as increase in plasma bacterial lipopolysaccharide (LPS) concentration increasing by two to three times.18 We proposed that smectite, due to its ability to bind endo- and exotoxins and capacity to restore the barrier properties of human intestinal cell monolayers, may be beneficial when supplemented with probiotics for NAFLD/NASH development.
Therefore, the present study aims to determine whether probiotics plus smectite is superior to probiotic alone on the monosodium glutamate (MSG) induced NAFLD model in rats.
Material and MethodsStudy designThis study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health and the general ethical principles of animal experiments, approved by the First National Congress on Bioethics Ukraine (September 2001). The protocol was approved by the Committee on the Ethics of Animal Experiments of the Taras Shevchenko National University of Kyiv (Protocol number: 19/2015). The rats were kept in collective cages under controlled conditions of temperature (22 ± 3 °C), light (12 h light/dark cycle) and relative humidity (60 ± 5%). The animals were fed laboratory chow (PurinaW) and tap water ad libitum.
We included 60 newborn Wistar male rats divided into 4 groups 15 animals in each: intact (I), MSG-obesity (II), Symbiter (III) and (Symbiter+Smectite) (IV) groups. Newborns rats of intact group were subcutaneously (s.c.) injected with saline in the volume of 8 µL/g and animals of groups II-IV received MSG solution (4.0 mg/g of body weight) s.c. at 2nd, 4th, 6th, 8th and 10th postnatal days. Neonatal administration of MSG causes the significant accumulation of abdominal fat with subsequent development of NAFLD and severe visceral obesity in adult rats.19,20 This happens because of the neurotoxicity effects on the arcuate and ventromedial nuclei of the hypothalamus.21
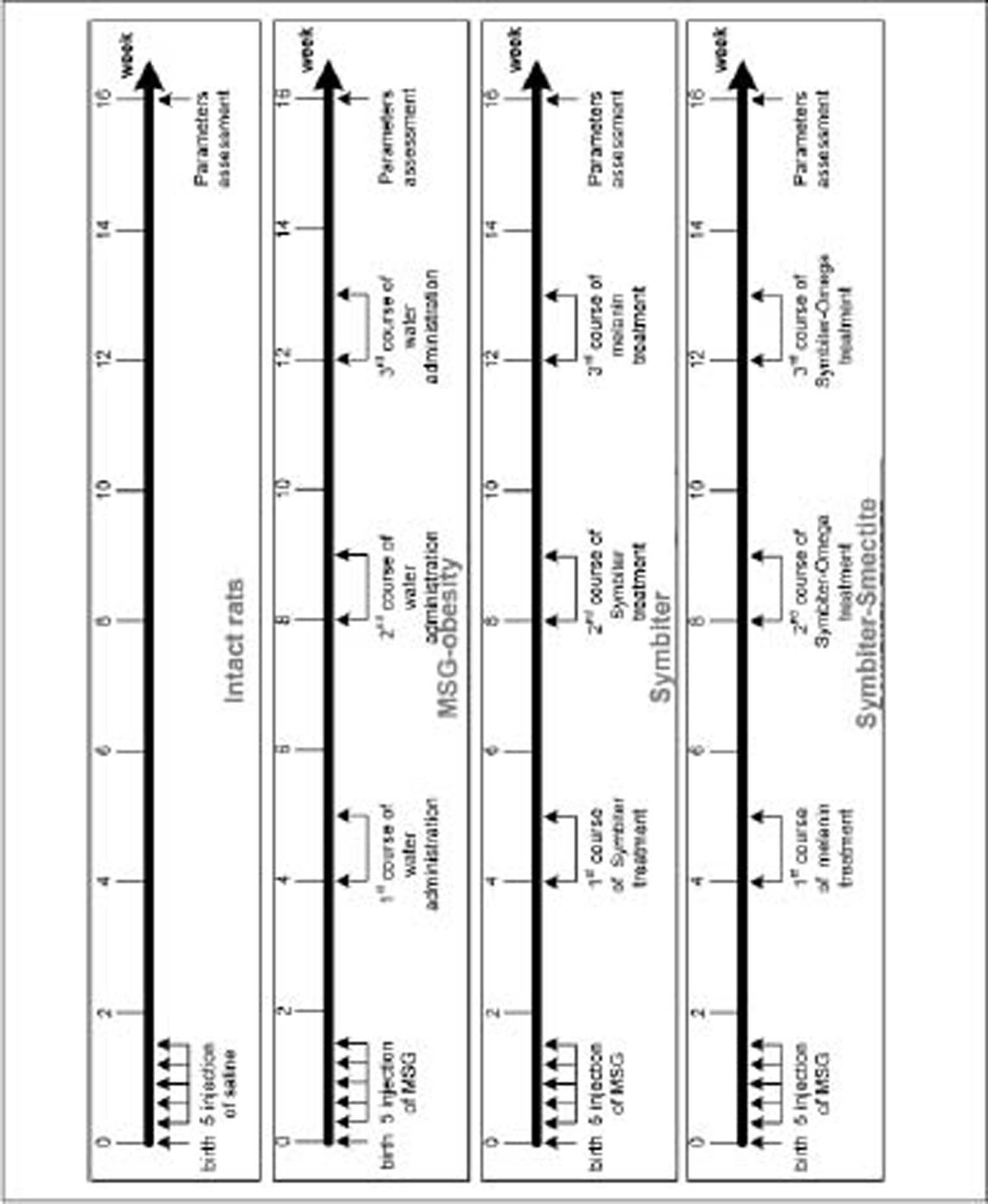
During the first 4 months after birth rats had a normal diet. The III (Symbiter) group received 2.5 mL/kg of multiprobiotic “Symbiter” containing concentrated biomass of 14 probiotic bacteria genera Bifidobacterium, Lactobacillus, Lactococcus, Propionibacterium. The IV (Symbiter+Smectite) groups received “Symbiter Forte” combination of probiotic biomass with smectite gel (250 mg). Administration was started at the age of 4 weeks just after the wean and continued for 3 month intermittently alternating two-week course of introduction with two-week course of break (Figure 1).
Sample collection and blood biochemistry analysisRats of all groups were fasted for approximately 12 h prior sacrifice by cervical dislocation under urethane anesthesia. Blood was drawn from the apex of the cardiac ventricle and few blood drops were collected into a microcentrifuge tube containing a mixture of NaF and EDTA at a 2:1 (w/w) ratio. Blood sample was collected into a sterile tube and centrifuged at 3,500 rpm (2260 g) for 15 min. After centrifugation serum supernatant for further analysis was aliquoted into microcentrifuge tubes and stored at −80 °C. Bilirubin, activity of alanine (ALT) and aspartate aminotransferase (AST) in serum were determined by the standard biochemical methods.
The contents of interleukins (ILs) 1ß, 4, 10, 12B p40, interferon (INF) γ, and transforming growth factor (TGF) β in rat serum were measured by ELISA using specific mono- and polyclonal antibodies (Sigma) to these proteins. Monoclonal antibodies to ILs 4, 10 and TGF-ß were mice-produced. Polyclonal antibodies to IL-12B p40 were produced in rabbits and polyclonal antibodies to IL-1 ß and INF-y were produced in goats. Studied molecules were immobilized in 96-well plates with sorption surface. Then primary and secondary antibodies labeled with enzyme were added to wells. Adding of substrate led to the development of colored reaction. The optical density of the solution in each well after the addition of substrate is proportional to the content of studied cytokines. The content was expressed in absorbance units of optical density.
Liver histology assessmentLiver tissue samples from both the right and left hepatic lobes were taken (sample size 0.5 x 0.5 cm) for histological analysis. After being fixed for 24 h in a liquid Buena, fragments of liver were dehydrated in alcohols of increasing concentrations (from 70° to 96°), embedded in paraffin and then sliced into 5-6 micron thick pieces and stained with hematoxylin-eosin. A pathologist blinded to group distributions performed the histological analyses of slides using light microscopy (“Olympus”, Japan). To assess morphological changes in liver we used NAS (NAFLD activity score), which includes histological features and has been defined as unweighted sum of scores for steatosis (0-3), lobular inflammation (0-3) and ballooning (0-2). According to NAS scores, ≥ 5 are diagnosed as non-alcoholic steatohepatitis (NASH), and cases with a NAS < 3 are mentioned as not NASH.22 Lipid extraction from the liver was performed according to Folch, et al.23
Statistical analysisStatistical analysis performed by using SPSS-21 software. All data were expressed as mean ± standard error (M±SEM) or %. Data distribution was analyzed using the Kolmogorov-Smirnov normality test. Continuous variables with parametric distribution were analyzed using Analysis of Variance (one-way ANOVA) and if the results were significant, a post-hoc Turkey’s test was performed. For data with non-parametric distribution Kruskall-Wallis and post-hoc Tukey’s test were conducted for multiple comparisons. For comparisons of categorical variables we conducted χ2 test. The difference between groups was defined to be statistically significant when a p-value was less than 0.05.
ResultsWe didn’t find any significant changes in liver serum (ALT, AST, bilirubin) function parameters between intact, MSG-obesity and both interventional groups (Table 1).
Liver function tests of rats under MSG-induced obesity and after administration of probiotic and their combination with smectite.
| Parameters | Intact rats (n = 15) | MSG-induced Obesity (n = 15) | Symbiter (n = 15) | Symbiter + Smectite (n = 15) |
|---|---|---|---|---|
| Total bilirubin, umol/l | 12.9 ± 0.73a | 12.6 ± 0.58a | 13.4 ± 0.63a | 12.4 ± 0.47a |
| Indirect bilirubin, umol/l | 8.3 ± 0.55a | 8.2 ± 0.35a | 8.3 ± 0.53a | 8.3 ± 0.33a |
| Direct bilirubin, mmol/l | 4.6 ± 0.33a | 4.4 ± 0.3a | 5.1 ± 0.23a | 4.1 ± 0.27a |
| ALT, mkkat/l | 0.231 ± 0.011a | 0.243 ± 0.015a | 0.212 ± 0.007a | 0.211 ± 0.014a |
| AST, mkkat/l | 0.386 ± 0.007a | 0.397 ± 0.011a | 0.381 ± 0.018a | 0.373 ± 0.016a |
Data are presented as the M ± SEM. One-way ANOVA or Kruskall-Wallis test were performed for data analysis. All differences were not significant (p > 0.05).
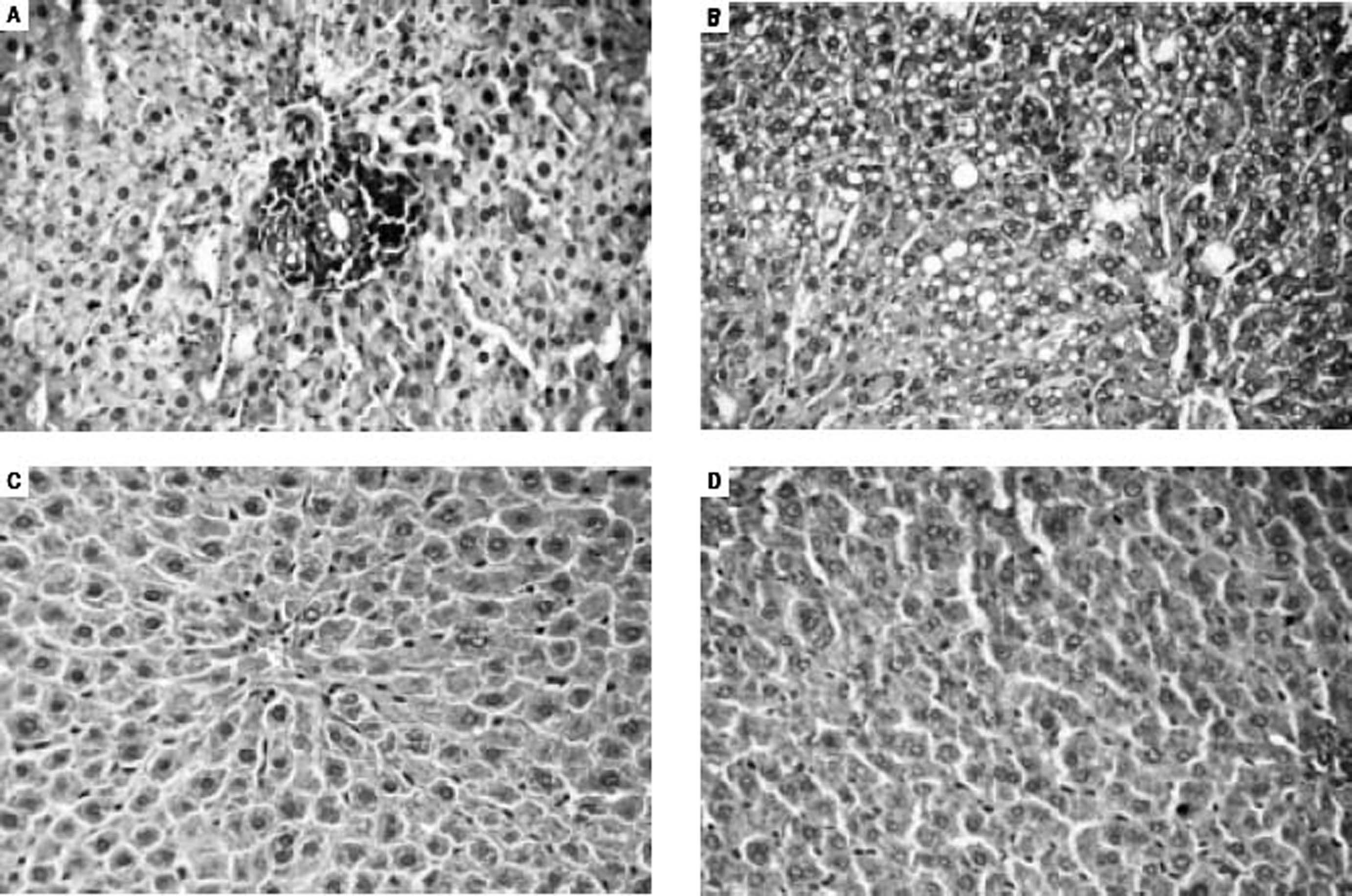
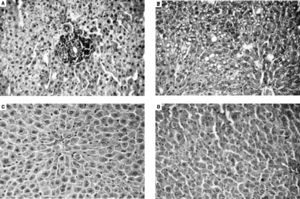
Histological analysis of liver micropreparations confirmed the development of NAFLD in rats. It was registered in MSG-obesity group typical for NAFLD histological pattern: microvesicular steatosis (Figure 2A), mild lobular inflammation in zone 3 and ballooning degeneration (Figure 2B), which were also confirmed by total NAS score increasing approximately by 18 times (p < 0.001) compared with values of intact rats (Table 2). The degree of steatosis was increased by 10 times (p < 0.001) in MSG-rats as compared with intact rats (Table 2). Intact rats did not display inflammation in liver, at the same time lobular inflammation in MSG-group reached 1.13 ± 0.16 points (Table 2). Similar results were recorded for ballooning degeneration (Table 2).
Light microscopic micrographs of the rat liver tissue stained with hematoxylin and eosin, x400. A, B. MSG-induced obesity group (II). In micrographs observed perivascular leukocyte infiltration at zone 3 (mild lobular inflammation) (A) and predominantly microvesicular pronounced total steatosis (B). In micrographs mainly observed focal mild microvesicular steatosis (C, D). C. Symbiter group (III). D. Symbiter-Smectite (IV) group.
Morphological changes of the liver tissue under MSG-induced obesity and after administration of probiotic and their combination with smectite.
| Parameters | Intact rats (n = 10) | MSG-induced obesity (n=10) | Symbiter (n = 10) | Symbiter + Smectite (n = 10) |
|---|---|---|---|---|
| Steatosis (0-3) | 0.20 ± 0.13a | 2.13 ± 0.19b | 0.93 ± 0.22a | 0.87 ± 0.16a |
| Lobular Iiflammation (0-2) | 0.0 ± 0.0a | 1.13 ± 0.16b | 0.33 ± 0.15a | 0.13 ± 0.09a |
| Ballooning degeneration (0-2) | 0.0 ± 0.0a | 0.33 ± 0.02b | 0.0 ± 0.0a | 0.0 ± 0.0a |
| Total NAS (0-8) | 0.20 ± 0.13a | 3.67 ± 0.3c | 1.27 ± 0.31b | 1.0 ± 0.21ab |
| Prevalence of NASH, % | - | 33,3 | 6,7 | - |
Data are presented as the M ± SEM. One-way ANOVA with post hoc Tukeys test for multiple comparisons were performed for data analysis. a,b,c Values at the same row with different superscript letters show significant differences at p < 0.05.
In both interventional groups reduction of total NAS score as compared to MSG-obesity was observed. However only in Symbiter+Smectite group we didn’t find significant changes as compared to intact rats (1.0 ± 0.21 vs. 0.20 ± 0.13, p = 0.223). Indeed similar values of steatosis score (0.93 ± 0.22 vs. 0.87 ± 0.16, p = 0.994) in both treatment groups, we observed that lower total score for Symbiter+Smectite are associated with more pronounced reduction of lobular inflammation (0.13 ± 0.09 vs. 0.33 ± 0.15, p = 0.695) as compared to administration of probiotic alone. Furthermore, in both interventional groups, lobular inflammation was remarkably reduced, we determined predominantly focal steatosis, the fat droplet number significantly decreased and the hepatic lobules were clearly delineated as compared with MSG group (Figures 2C, D).
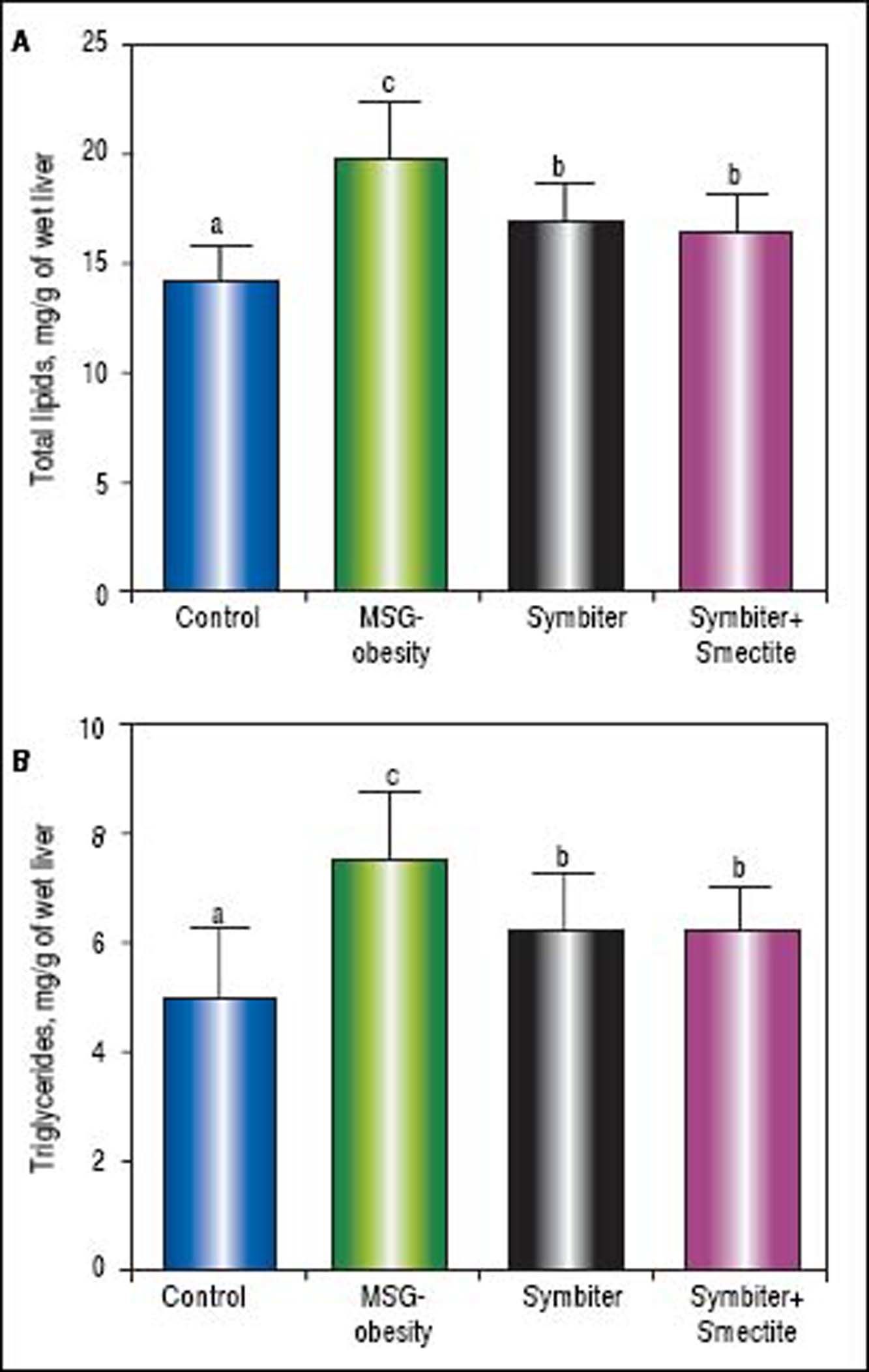
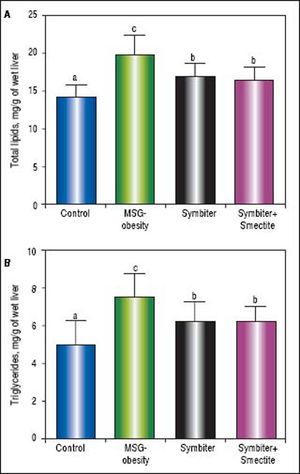
Administration of both Symbiter+Smectite or probiotic mixture alone led to significant decrease of total lipids and triglycerides content in liver as compared to the MSG-obesity group (Figures 3A, B). Nevertheless, the differences of liver lipid contents between both interventional groups were insignificant.
Liver total lipids and triglycerides content of rats with the MSG-induced obesity and after administration of probiotic and their combination with smectite. Data are presented as the M ± SEM. One-way ANOVA with post hoc Tukeys test for multiple comparisons were performed for data analysis. a,b,c Values at the same row with different superscript letters show significant differences at p < 0.05.
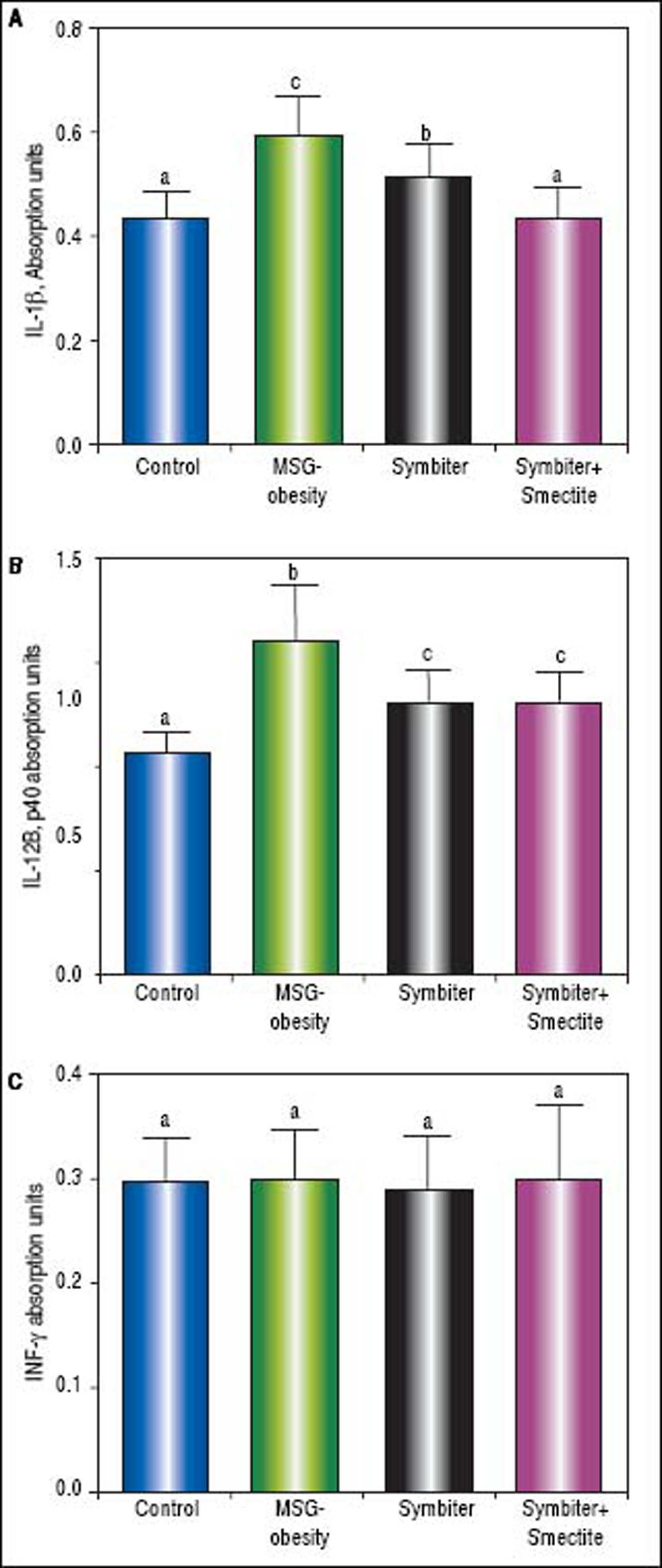
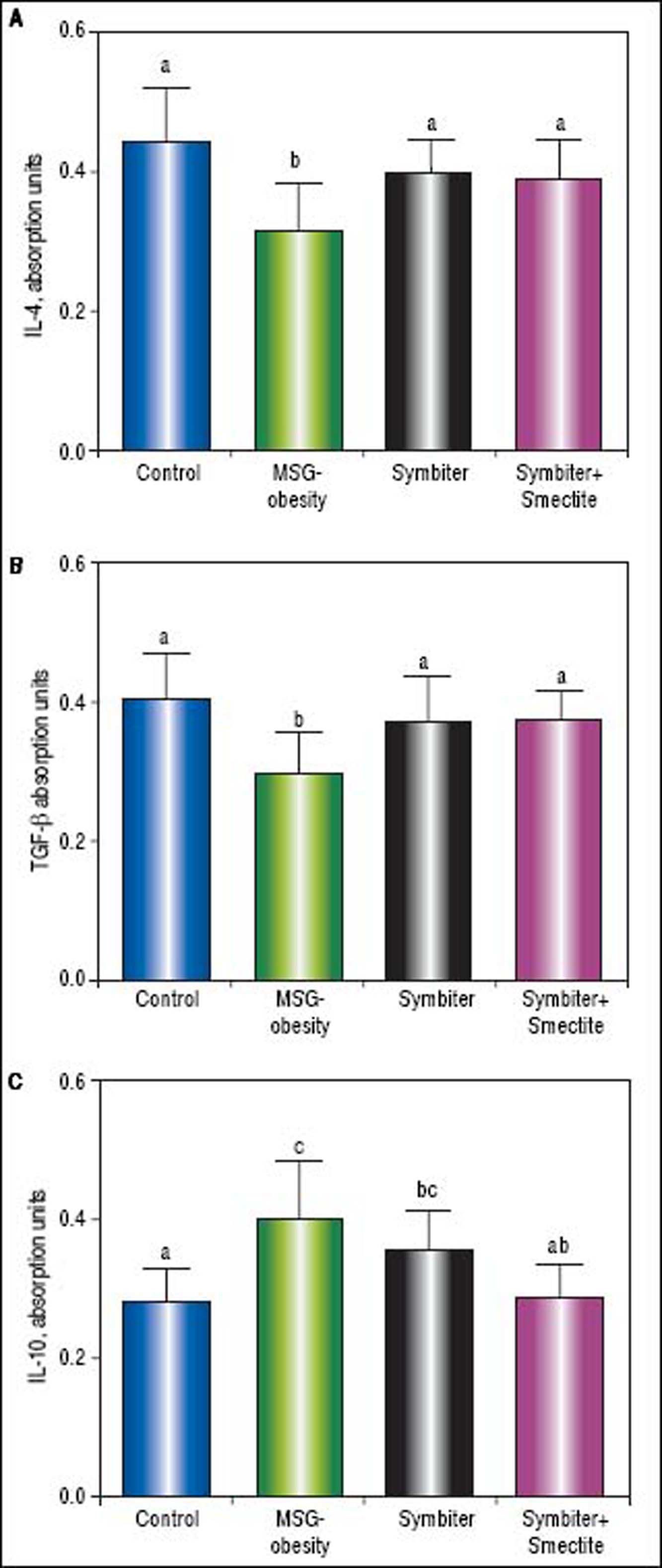
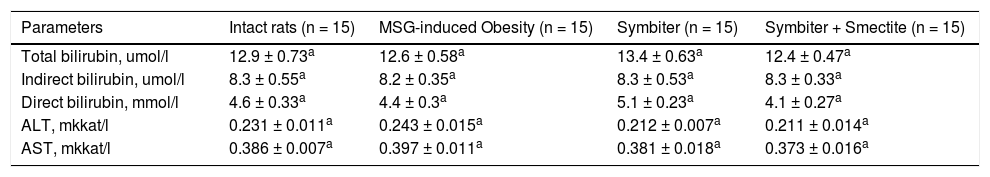
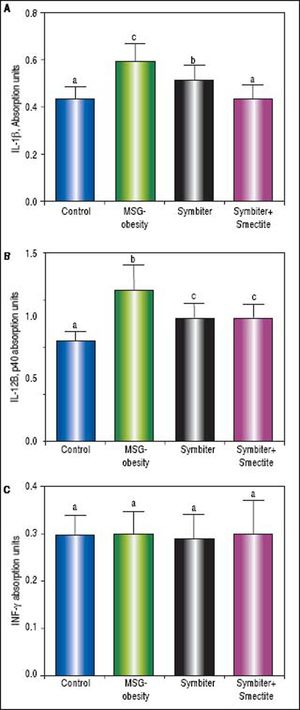
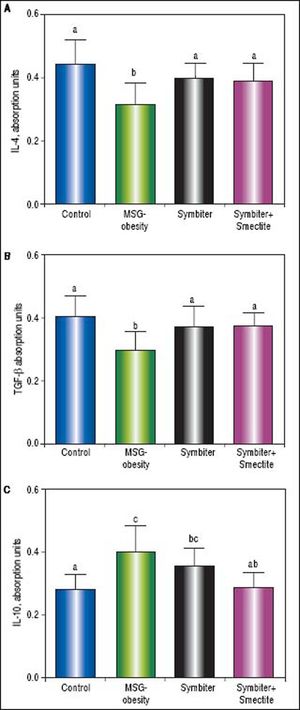
Immunoassay analysis has shown the intensification of inflammatory processes in conditions of MSG-obesity. The increase content of pro-inflammatory cytokines IL-1β and IL-12B p40 in obese rat serum was found. The IL-1ß level raised by 37.1% (p < 0.001) and IL-12B p40 - by 50.7% (p < 0.001) as compared to intact rats (Figures 4 A, B). In contrast, we did not reveal difference in the level of INF-y between obese and intact rats (Figure 4C). In the conditions of obesity, anti-inflammatory system was down-regulated. That was evident because of the reduction of IL-4 by 29.0% (p < 0.001) (Figure 5A) and TGF-β by 27.2% (p < 0.05) (Figure 5B) in MSG-group as compared to control. As opposed to this IL-10 was elevated in obese rats by 55.4% (p < 0.001) that may suggest compensatory effect of anti-inflammatory system (Figure 5 C).
Serum proinflammatory cytokines content of rats with the MSG-induced obesity and after administration of probiotic and their combination with smectite. Data are presented as the M ± SEM. One-way ANOVA with post hoc Tukeys test for multiple comparisons were performed for data analysis. a,b,c Values at the same row with different superscript letters show significant differences at p < 0.05, a.u. - absorption units.
Administration of probiotic alone or in combination with smectite gel led to similar decrease of IL-12 (Figure 4 B) and significant elevation of the IL-4 (Figure 5 A) and TGF-β (Figure 5 B) approximately on 25-35% (p < 0.001) as compared to MSG-obese rats. Furthermore, significant activation of the anti-inflammatory system was accompanied by restoration of cytokines levels to levels in intact rats. The reduction of IL-1β (Figure 4A) and IL-10 (Figure 5C) also at least partially achieve values of intact rats but was most pronounced in Symbiter+Smectite group as compared to probiotic alone and to MSG-obese rats.
Serum anti-inflammatory cytokines content of rats with the MSG-induced obesity and after administration of probiotic and their combination with smectite. Data are presented as the M ± SEM. One-way ANOVA with post hoc Tukeys test for multiple comparisons were performed for data analysis. a,b,c Values at the same row with different superscript letters show significant differences at p < 0.05, a.u. - absorption units.
The use of probiotics in the NAFLD treatment and prevention has dramatically increased over the last decade. Probiotic supplements are well known ingredients of functional foods and nutraceuticals and may provide beneficial health effects because they can influence the intestinal microbial ecology and immunity.24
The previous animal studies demonstrated strain-specific potential of probiotics for NAFLD development. The most favorable effects on NAFLD are associated with Lactobacillus and Bifidobacterium strains and appear with restoration of gut barrier function,25 improvement of insulin sensitivity,14 reduction of LPS absorption,26 serum cholesterol level,27 liver fat accumulation,25–28 prevention from lipid peroxidation, NFκB activation28 and subsequent attenuation of liver inflammation and steatosis in distinct animal models of diet- and genetically determined obesity. Cani, et al. summarized these findings and showed that at least 15 different strains of Lactobacillus and 3 strains of Bifidobacterium do not equally influence on hepatic lipids and NAFLD manifestation. Remarkably, 12 strains decreased hepatic tissue inflammation and 11 reduced the hepatic triglyceride content when given as a single treatment.29 Interesting data reported by these authors.30 They find failure of NAFLD prevention with lyophilized monoprobiotics B. animalis VKL, B. animalis VKB, L.casei IMVB-7280 strains. Nevertheless, multiprobiotic cocktails due to formation of mutualistic interactions lead to significant reduction of hepatic steatosis, total lipids and triglycerides content in the liver and finally prevent the development of NAFLD in animals as compared to MSG-obesity littermates. Remarkably that more pronounced changes were admitted after administration of probiotic mixture preferably containing alive strains as compared to lyophilized cocktails.
Smectite is a natural silicate that binds to digestive mucous and has the ability directly to absorb bacterial toxins, bacteria, viruses and bile salts.31,32 On the other hand, when administered orally, smectite is not absorbed and eliminated unchanged directly with faeces within sixteen hours. Smectite increases water and electrolyte absorption, may affect intestinal permeability, formes multilayer structure with high plastic viscosity and powerful coating properties hence preserving integrity of the mucus layer and rendering the intestinal epithelium more resistant to endogenous (bile salts) or exogenous (such as bacterial toxins and alcohol) impacts.31,33,34 Diosmectite also has a protective effect against intestinal inflammation34 hence suppressing production of cytokines such as interleukin-8 from secretory epithelial cells35in vitro and to attenuating the proinflammatory action of TNFα.36 We suggested that all these pharmacological properties may be beneficial for the treatment of NAFLD.
According to this suggestion we firstly designed the study to test the hypothesis that whether probiotics plus smectite is superior to probiotic alone on NAFLD prevention in rats. The reduction of total NAS score in both nutraceuticals groups as compared to MSG-obesity was observed. However only for Symbiter+Smectite group, indeed similar values of steatosis score, more pronounced but insignificant reduction of lobular inflammation (0.13 ± 0.09 vs. 0.33 ± 0.15) as compared to administration of probiotic alone was demonstrated. The reduction of IL-1β and restoration of IL-10 also at least partially achieved values of intact rats but was most pronounced in Symbiter+Smectite group as compared to probiotic alone and to MSG-obese rats.
ConclusionDue to smectite gel’s absorbent property and its ability to stabilize mucus layer, combining it with administration of alive probiotics can have an impact on synergistic enhancement of single effect manifested in reduction of lobular inflammation and at least partly NASH prevention.
In summary, our study creates conditions for finding new combinations of nutraceuticals that may enhance or summarize single effects of separate composition, that may be more beneficial for treatment or prevention of different metabolic disturbances.
Abbreviations- •
ALT: alanine aminotransferase.
- •
AST: aspartate aminotransferase.
- •
ILs: interleukins.
- •
INF: interferon.
- •
LPS: lipopolysaccharide.
- •
MSG: monosodium glutamate.
- •
NAFLD: non-alcoholic fatty liver disease.
- •
NAS: NAFLD activity score.
- •
NASH: non-alcoholic steatohepatitis.
- •
NFκB: nuclear factor kappa-light-chain-enhancer of activated B cells.
- •
SC: subcutaneously.
- •
TGF: transforming growth factor.
The authors express their sincere thanks to Dr. Yankovsky Dmitro Stanislavovych for the help, advice and financial support of this work.
Conflict of Interests
The authors report no conflict of interests.