Genetic predisposition, the intestinal microbiota (IM) and environmental factors, such as sedentary lifestyle and inadequate diet, should be considered as critical factors for the development of nonalcoholic fatty liver disease (NAFLD). Recently, some studies have demonstrated an association between dysbiosis and NAFLD; however, the exact mechanisms that lead to intestinal membrane damage, bacterial translocation and inflammation are not well elucidated. Due to the relevance of this theme, the IM and its metabolites have received special attention in recent years in an attempt to better understand the mechanisms related to the prevention, physiopathology, and treatment of NAFLD. In this paper, we provide a review of the human IM and its role in diet, obesity, and the development/progression of NAFLD/NASH, as well as the use of prebiotics and probiotics in the modulation of IM.
The human intestinal epithelium has an absorptive surface comparable to 300–400m2, with approximately 1014 resident bacterial species. Bacterial colonization occurs before birth and continues for life. Most of the bacteria that colonize the intestine are of the anaerobic type and comprise approximately 36,000 different species [1,2], constituting a variety of species of microorganisms that, quantitatively and qualitatively, differ between individuals [3].
The main functions of the intestinal microbiota (IM) include metabolic and immunological activities. The prominent metabolic function of the IM is the fermentation of nondigestible components of the diet. The products of this complex metabolic activity are a spectrum of organic acids, which are an important source of energy for the host. The epithelial cells of the gut participate in immunological processes. The signaling pathways of these cells are highly regulated to avert uncontrolled inflammatory responses [4]. The gut epithelium is a major barrier between the host and the environment. This barrier is formed by tight junctions that act as a highly regulated entry that open and close depending on signals, such as those from cytokines and bacterial components from the lumen, and excessive contact with bacteria may cause an increase in the proinflammatory immune response [5]. Therefore, the IM supports immune regulation and ensures that effector immune responses are activated in response to invading pathogens [6]. Another important function of the IM is protection against exogenous microorganisms. These bacteria can inhibit the growth of pathogenic microorganisms through the synthesis of antimicrobial substances or by competing for nutrients and reducing the attachment and penetration of the enteroinvasive pathogen into epithelial cells [7].
The IM has a heterogeneous distribution, with the colon being the place with the greatest density due to the favorable conditions for the proliferation of microorganisms, characterized by slow peristalsis, absence of intestinal secretions and presence of a nutritional supply [8]. Dysbiosis generates bacterial overgrowth, toxin production and increased intestinal permeability, resulting in changes in immunological and hormonal effects. Thus, lifestyle habits such as diet, stress and the use of antibiotics, for example, cause the transient microbiota to prevail over the resident microbiota, predisposing the host to gastrointestinal disorders. The actions that the IM exerts on the various systems of the organism also reflect the maintenance of the metabolism [9].
Approximately 98% of all bacterial species that make up the IM belong to the following phyla: Firmicutes (64%) and Bacteroidetes (23%), which represent most of the IM in addition to Proteobacteria (8%) and Actinobacteria (3%) [10]. The Firmicutes phylum contains more than 250 genera, including Lactobacillus[11]. The phylum Bacteroidetes, similar to Firmicutes, is found in the small and large intestines, especially in the colon, and encompasses several genera, such as Bacteroides, Flavobacteria and Sphingobacteriales, with Bacteroides thetaiotaomicron being one of the most abundant [12].
Although the functions of the bacteria belonging to the Firmicutes phylum have not yet been fully elucidated, evidence suggests that some members of this phylum, such as Clostridium butyricum bacteria, are among the bacteria producing butyric acid from lactic acid or lactose in a chemical reaction known as butyric fermentation, and this type of fermentation increases the energy extracted from the diet [13]. On the other hand, members of the Bacteroidetes phylum participate in the metabolism of carbohydrates and perform this metabolism by expressing enzymes similar to the glycosyltransferases, which promote glycosylation, that is, to join carbohydrates to the proteins formed in the ribosomes. Members of the Bacteroidetes phylum also express glycoside hydrolases that degrade biomass, such as cellulose and hemicellulose, in antibacterial defense strategies. Finally, they also express the polysaccharide-lyase enzyme that catalyzes the breakdown of a carbon–oxygen bond in polysaccharides, leading to an unsaturated product and the elimination of alcohol [14].
2Intestinal microbiota and obesityIntestinal microorganisms influence energy production, energy homeostasis and, potentially, the development of obesity and metabolic disorders [11]. Increasing evidence supports the importance of the composition of the IM for the development of obesity and demonstrates that the type and quantity of the bacterial population can determine some important characteristics of metabolic pathways that play an important role in obesity [15,16].
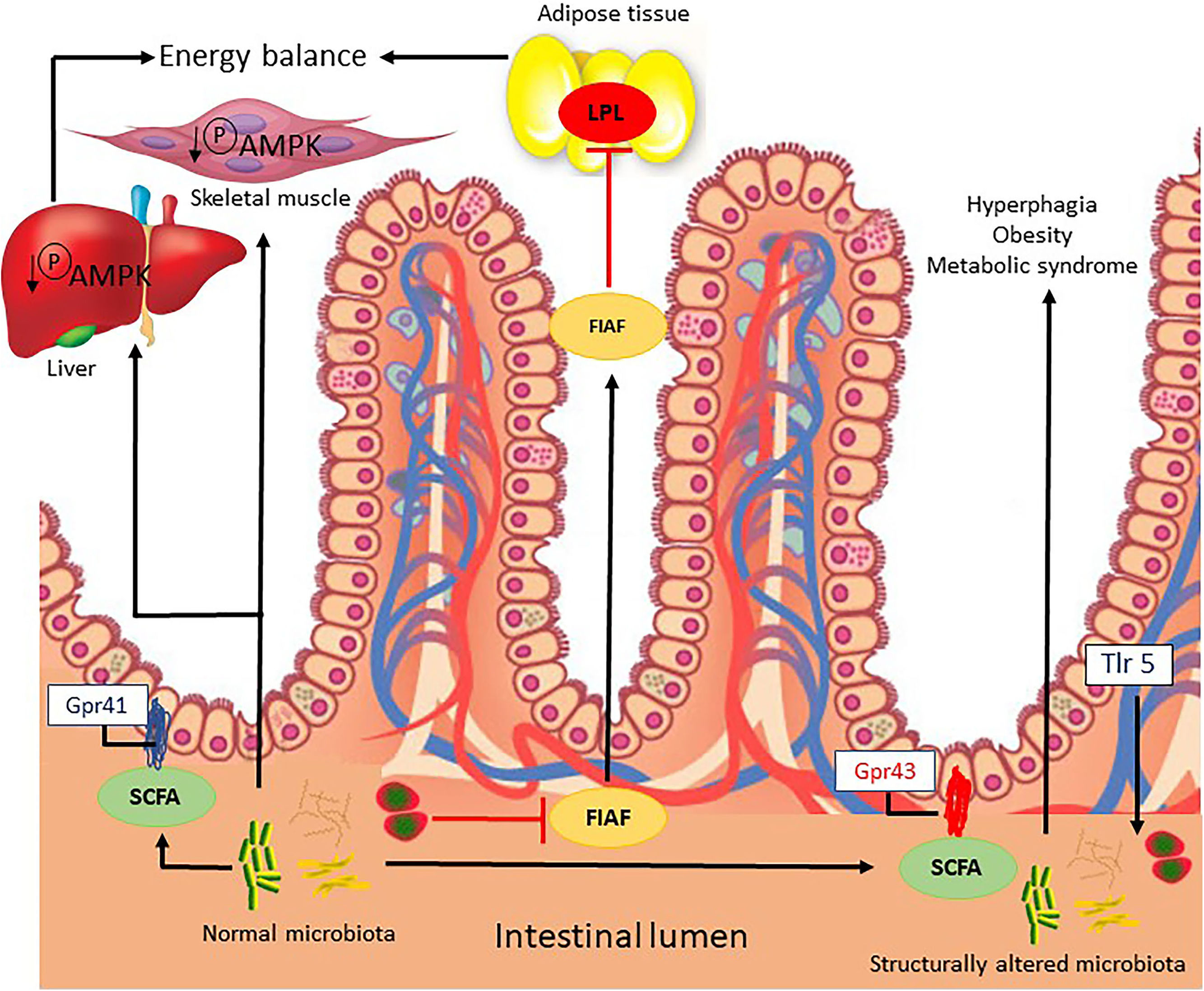
Microbial signals regulate the release of fasting-induced adipose factor (FIAF) from intestinal epithelial cells, acting as a lipoprotein lipase inhibitor and regulating the storage of peripheral fat. In addition, by unknown mechanisms, the IM regulates the energy mediator in the liver and muscle via phosphorylation of AMP-activated protein kinase (Fig. 1) [17].
Possible mechanisms related to the intestinal microbiota that lead to energy imbalance and consequently obesity.
FIAF, fasting-induced adipose factor; Gpr41/43, G-protein coupled receptor; LPL, lipoprotein lipase; SCFAs, short chain fatty acids; AMPK, AMP-activated protein kinase; TRL5, Toll-like receptor-5.
Microbial metabolites, such as short chain fatty acids (SCFAs), bind to conjugate receptors of G protein on intestinal epithelial cells by regulating energy from gut-derived hormones, such as the intestinal peptide hormone, and controlling the inflammatory response of the host. The activation of Toll-like receptor-5) in the epithelia or myeloid cells affects the structural composition of the intestinal microbiota, which regulates appetite, weight gain and insulin sensitivity (Fig. 1) [18].
It is suspected that the intestinal microbiota of obese individuals presents peculiarities that may induce chronic inflammation. The induction of endotoxemia in obesity, diabetes mellitus and insulin resistance causes the expression of inflammatory factors that resemble those present in a diet rich in fat [19].
Obesity and diet composition can cause sensitivity to the intestinal barrier and result in the malfunction of the intestine–liver axis, allowing the indiscriminate passage of bacterial components into circulation. The intestinal barrier is a complex functional unit composed of luminal and mucosal elements, such as the epithelial cell layer, the mucosal barrier, and innate and acquired immune components. This barrier plays a key role in protecting against enteric organisms, potentially harmful toxins and bacterial bioproducts that are closely associated with health and disease susceptibility [20,21]. The intestine–liver axis refers to the anatomical and functional relationship between the gastrointestinal (GI) tract and the liver. The interaction between the two organs, whether healthy or diseased, includes the transfer of the IM and its potential hepatotoxic products to the liver [22]. Alterations in this axis (consisting of the intestinal barrier, the IM and the liver) may lead to changes in intestinal permeability favoring excessive pathogenic bacterial growth (dysbiosis). In particular, the excessive growth of Gram-negative bacteria increases the production of hepatotoxic products such as lipopolysaccharides (LPS), which in turn stimulate Toll-like receptor-4 and ultimately nuclear factor kappa B (NF-kβ) and proinflammatory cytokines, such as interleukin-6, transforming growth factor-beta and tumor necrosis factor-alpha (TNF-α) [23,24].
3Intestinal microbiota and dietThe relationship between the diet and the composition and function of the IM is evident. Indeed, bacteria in the gut are responsible for the digestion and production of many essential vitamins and minerals. The dietary components provide nutrients for bacteria, which then produce metabolites involved in energy balance, metabolism, and immune response, but these metabolites can also result in obesity and metabolic diseases [25,26]. The high caloric content of the Western diet (high in fat and carbohydrates) is a major contributor to weight gain, which is associated with an altered IM [27,28]. A recent study showed that the IM promotes intestinal absorption and the transfer of monosaccharides to the portal circulation by increasing the expression of the glucose-1 transporter in the small intestine. This increased carbohydrate flow stimulates lipogenesis in the liver and adipose tissue [15]. A study comparing a control diet vs. diets high in nondigestible carbohydrates (fibers) demonstrated that nondigestible carbohydrates produce significant changes in the species composition (E. rectale and Ruminococcus bromii) of the IM within a number of days [29]. The link between diet, IM, and health has been shown in animal models that were switched from low-fat/fiber rich plant diets to high-fat/high-sugar diets and exhibited significant increases in Bacilli and Erysipelotrichia from the Firmicutes phylum, which were associated with a significant decrease in the abundance of members of the Bacteroidetes phylum [30]. The GI transit time could be affected by the IM according to Kashyap et al. They proposed an animal model to evaluate the interactions between diet, IM, and GI transit time in the host. Diet can independently affect both GI transit time and IM composition and function (as determined by metabolite profiles). However, diet-induced changes in microbial composition may be mediated in part by changes in GI transit time, and the effect of diet on GI transit time may be a result of altered functionality of the IM community caused by the dietary change [31].
4Intestinal microbiota and NAFLD/NASHNonalcoholic fatty liver disease (NAFLD) is one of the most common chronic liver disorders worldwide due to the epidemic increase in the prevalence of obesity in affluent societies [32]. NAFLD includes a wide spectrum of hepatic pathological characteristics ranging from simple steatosis to nonalcoholic steatohepatitis (NASH) with or without fibrosis, cirrhosis, and hepatocellular carcinoma [33]. The development and progression of NAFLD involve complex pathophysiological processes [32] that are influenced by host factors such as genetic polymorphisms, diet, and, as reported more recently, the composition of the IM [34,35].
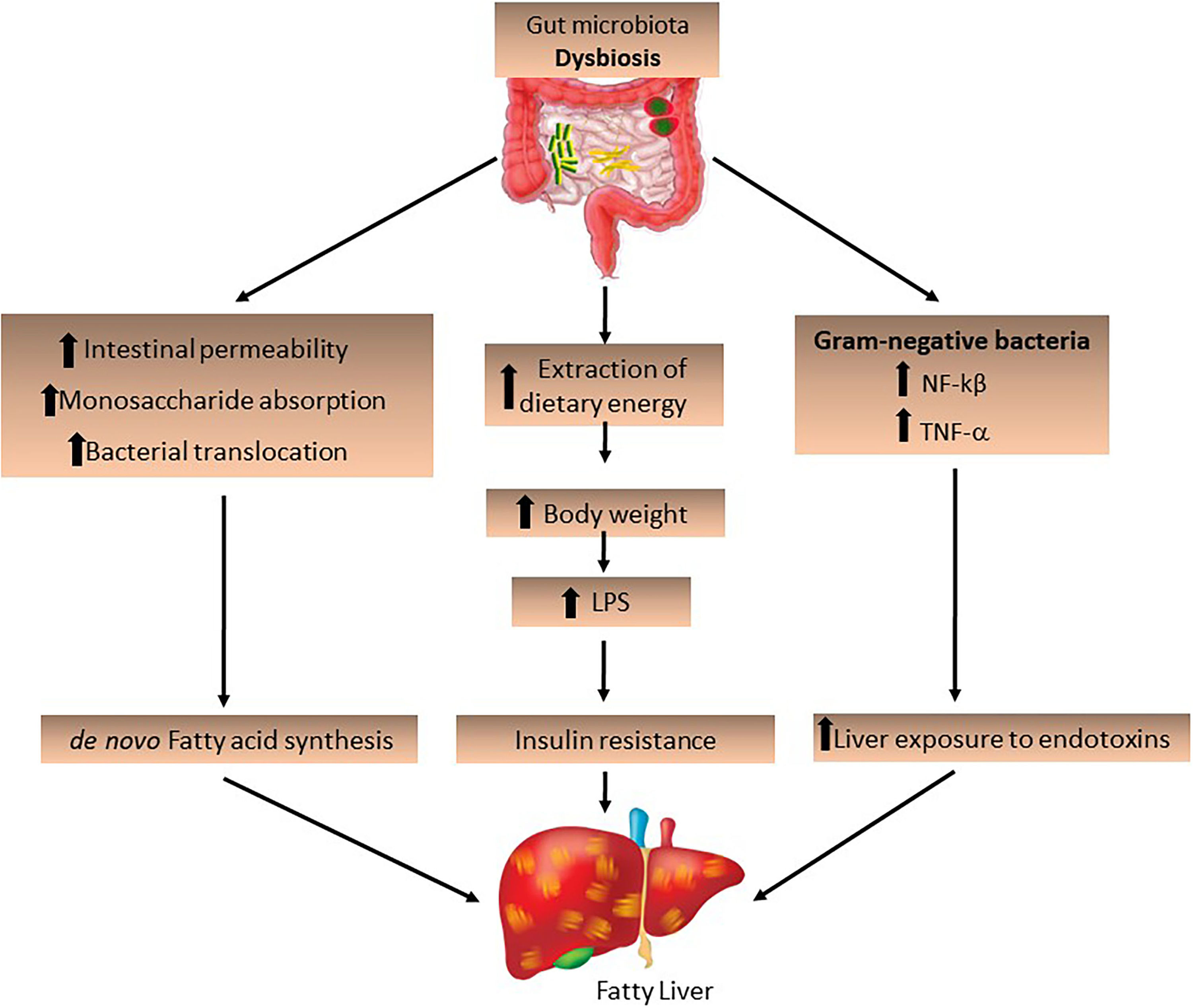
The IM and its derived metabolites may also contribute to the development of NAFLD. Increased intestinal permeability stimulates monosaccharide absorption from the intestinal lumen, leading to bacterial translocation. The endotoxins produced by these bacteria penetrate the portal vein and decrease FIAF secretion by increasing LPL activity, promoting de novo fatty acid synthesis and triglyceride production [9] and activating inflammatory Toll-like receptors in hepatocytes [23]. A permeable intestinal barrier and small intestine bacterial overgrowth, which is a condition in which elevated levels of small intestinal bacteria lead to abdominal distension and other common symptoms related to irritable bowel syndrome, are frequent in obese patients and induce liver injury by increasing LPS production derived from Gram-negative bacteria in the gut, which activates NF-kβ and TNF-α production. This suggests that IM increases liver exposure to endotoxins, which play an important role in the progression of NASH (Fig. 2) [36,37].
The role of the intestinal microbiota in the development of fatty liver. Intestinal dysbiosis may be a determining factor for the development and progression of NAFLD/NASH. The increase in pathogenic bacteria and Gram-negative bacteria increases dietary energy extraction, leading to increased intestinal permeability and bacterial translocation, stimulating de novo fatty acid synthesis and increasing LPS expression, as well as the expression of NF-kβ and TNF-α.
LPS, lipopolysaccharides; NF-kβ, nuclear factor-kappa β; TNF-α, tumor necrosis factor-alpha.
Miele et al. showed that NAFLD is associated with increased intestinal permeability and SIBO, and these factors are associated with the severity of hepatic steatosis. They reported evidence of a “leaky” intestine with tight junction disruption in human NAFLD with evidence of increased small intestine bacterial overgrowth [38]. Some studies in animal models and NAFLD or NASH patients showed an association with small bowel overgrowth and increased intestinal permeability [39–42]. Brun et al. demonstrated that leptin-deficient obese mice naturally presented increased intestinal permeability, higher circulation of inflammatory cytokines and increased serum levels of LPS [40]. Another study showed that LPS in mice simulated the effects of a high-fat diet by metabolic endotoxemia, which led to dysregulation of inflammation and triggered body weight gain, IR, and the development of NAFLD [43]. Studies involving obese and NAFLD patients showed an association between metabolic endotoxemia and hepatic and systemic inflammation [41,43,44]. Verdam et al. also observed an increase in plasma antibodies against LPS in patients with NASH compared to healthy controls. This increase was directly proportional to the increased severity of liver disease [45]. A recent study showed that obese patients with NASH had a decreased abundance of Faecalibacterium[46]. Species of Faecalibacterium exhibited anti-inflammatory effects, partly due to secreted metabolites that block NF-kB activation and interleukin 8 (IL-8) secretion. In addition, the effects were associated with a decrease in proinflammatory colonic cytokine synthesis and with the induction of anti-inflammatory cytokine secretion [47].
Recently, our group demonstrated a decreased abundance of Faecalibacterium and Ruminococcus in lean NASH patients [48]. Ruminococcus belongs to the family Ruminococcaceae, which are also fermenting anaerobes that lead to the production of SCFAs, promoting the development of NAFLD [49]. Interestingly, in the same study, advanced fibrosis (stage >2) was associated with an increase in Lactobacillus in obese NASH patients [48]. The genus Lactobacillus comprises more than 180 species with important immunological functions and metabolic activities [50]. Some of these bacteria produce lactic acid from the fermentation of carbohydrates in the diet and can produce acetate and ethanol [51], which can induce liver damage, such as inflammation, leading to the progression of NASH [52].
Some studies have shown an association between Bacteroides and Escherichia and advanced fibrosis [53,54]; Bifidobacterium spp. and Akkermansia muciniphila were strongly associated with markers of lipid metabolism and negatively associated with inflammation in adipose tissue and circulating glucose, leptin, triglycerides and insulin [55]. Despite the fact that the diet is directly connected to dysbiosis, a study showed that the IM of a genetically obese human could promote the onset of liver steatosis independent of dietary factors [21].
A possible mechanism by which the MI contributes to the development of NAFLD may be related to the amount of ethanol produced by bacteria (Escherichia coli) [56]. The ethanol produced by these bacteria contributes to physiological and morphological changes in the intestinal barrier associated with small intestine bacterial overgrowth by increasing intestinal permeability and thereby increasing the passage of endotoxins from the intestinal lumen to the portal blood, leading to an increase in the production of reactive oxygen species, which stimulates hepatic inflammation [57]. Ethanol is constantly produced by the MI even in the absence of alcohol ingestion. It has been found that a diet rich in refined sugar can lead to increased levels of alcohol in the blood and that ingested ethanol is eliminated via the enzyme alcohol dehydrogenase in the liver. This enzyme converts the alcohol into acetaldehyde, which, even in small concentrations, is toxic to the body [58]. The production of ethanol by the MI in obese and eutrophic patients with NASH suggests that it may contribute to the development and progression of the disease [59].
Another product of the metabolism of nutrients by bacteria that may be toxic to the liver is trimethylamine N-oxide. The IM can promote the conversion of choline to trimethylamine, which will then reach the liver by the circulation gate and will be converted into trimethylamine N-oxide [42]. The increase in the production of this compound leads to a decrease in the exportation of hepatic very-low-density lipoprotein and the modulation of bile acid synthesis, which has detrimental effects on the liver, such as increased liver fat deposition and inflammatory and oxidative lesions and decreased glucose metabolism [60].
NAFLD patients are characterized by a significant increase in circulating levels of endotoxins, which may represent an important early marker of potential liver abnormality [61]. Furthermore, gut dysbiosis may be associated with changes in the level of serum metabolites, including branched-chain amino acids and aromatic amino acids. Moreover, BCAAs are increased in individuals with IR, which is considered the major risk factor for hepatic steatosis [62].
5Prebiotics and probiotics and the intestinal microbiotaCurrent studies have shown that the use of prebiotics and probiotics can modulate the MI in several diseases, including NAFLD [63–65].
Probiotics improve the composition of the MI, which is associated with a reduction in liver inflammation, a decrease in LPS concentrations and an improvement in aminotransferase concentrations [66,67]. Probiotic supplementation can improve inflammatory factors [68,69] and improve metabolic parameters of NAFLD, such as visceral fat, total cholesterol and IR [70]. In addition, the consumption of probiotics has been associated with a decrease in low-density lipoprotein levels in animals and in humans with hypercholesterolemia [71,72]. Cano et al. demonstrated that the consumption of the Bifidobacterium pseudocatenulatum probiotic bacteria improved glucose tolerance, inflammatory status and hepatic steatosis [73].
Prebiotics can modulate the IM by increasing beneficial bacteria in the intestine, particularly Bifidobacteria and Lactobacilli, and inhibit bacterial activities that are detrimental to host health [74]. In addition, in the large intestine, inulin-type fructan and resistant starches are completely catabolized by the microbiota and are transformed into SCFAs, bacterial biomass and organic acids such as lactic acid and gases (carbon dioxide, hydrogen and methane) [66]. Data suggest that altering the microbiota through the use of prebiotics leads to decreased hepatic lipogenesis and blood TG levels and increases the concentration of SCFAs; however, the ability to modulate the intestinal microbiota and liver function in patients with NAFLD should be investigated [75–77].
Recent studies have shown that the use of prebiotics and probiotics can be an alternative therapy in the treatment of NAFLD. In an experimental model of NAFLD, it was observed that the use of synbiotics can reduce hepatic aminotransferases and IL-6 expression. However, the histology showed that the synbiotic was not able to prevent the progression of the disease [78]. A randomized clinical trial demonstrated that the use of multistrain probiotics was able to modulate the IM, thereby significantly reducing fatty liver and aminotransferase activity and reducing the expression of TNF-α and IL6 in NAFLD patients [79]. Another randomized study showed that synbiotic supplementation improves hepatic steatosis, aspartate aminotransferase levels, lipid profile and homeostatic model assessment) in NAFLD patients [64]. A study in NASH patients also showed that the use of synbiotics (Bifidobacterium longum and fructooligosaccharide), in combination with lifestyle modification (diet and exercise), improved aspartate aminotransferase, low-density lipoprotein, and TNF-α, leading to decreased endotoxins, IR and steatosis [80]. In addition, a study conducted by Eslamparast et al. corroborated the conclusion that the use of synbiotics combined with physical exercise improves aminotransferase levels, TNF-α and NF-kβ [81].
In conclusion, some evidence has demonstrated the role of the IM in the pathogenesis of NAFLD/NASH; however, it is necessary to explore how the IM can be modulated to improve NAFLD/NASH progression. Future randomized studies involving diet and/or probiotics should be performed.AbbreviationsGI gastrointestinal intestinal microbiota lipopolysaccharides nonalcoholic fatty liver disease nonalcoholic steatohepatitis nuclear factor-kappa B short-chain fatty acid tumor necrosis factor-alpha
The authors have no conflicts of interest to declare.