Endoscopic submucosal dissection is a technique developed in Japan for en bloc resection with a lower rate of recurrence. It is considered technically difficult and performed only in specialized centers. This study sought to report the initial experience from the Gastrocentro – Campinas State University for the treatment of gastric and colorectal lesions by endoscopic submucosal dissection.
MATERIALS AND METHODS:The guidelines of the Japanese Association of Gastric Cancer were used as evaluative criteria. For colorectal lesions, the recommended standards proposed by Uraoka et al. and Saito et al. were employed. The practicability of the method, the development of complications and histological analysis of the specimens were evaluated.
RESULTS:Sixteen patients underwent endoscopic submucosal dissection from June 2010 to April 2011; nine patients were treated for gastric lesions, and seven were treated for colorectal lesions. The average diameter of the gastric lesions was 28.6 mm, and the duration of resection was 103 min without complications. All lesions presented lesion-free margins. Of the seven colorectal tumors, four were located in the rectum and three were located in the colon. The average size was 26 mm, and the average procedure time was 163 min. Two complications occurred during the rectal resection procedures: perforation, which was treated with an endoscopic clip, and controlled bleeding. One of the lesions presented a compromised lateral margin without relapse after 90 days. Depth margins were all free of lesions.
CONCLUSION:Endoscopic submucosal dissection at our institution achieved high success rates, with few complications in preliminary procedures. The procedure also made appropriate lesion staging possible.
Endoscopy is a useful tool for the diagnosis, staging and treatment of early cancers of the gastrointestinal tract. In minimal invasion to superficial lesion, mucosal-restricted lesions can receive endoscopic treatment with a curative intent.
Endoscopic mucosectomy resection (EMR) is the most widely used technique because it is easy to use and has a low complication rate (1). However, for larger lesions, en bloc resection is not possible, which makes histological analysis difficult and increases the rate of local recurrence (2).
Endoscopic submucosal dissection (ESD) is a technique developed in Japan that enables en bloc resection, thereby facilitating a histological study with appropriate analyses in both lateral and deep margins (3,4). However, because this is a complex procedure, global application of the technique is difficult.
The Gastrocentro Endoscopic Unit at the Campinas State University started preparations to employ this technique in 2009 after the acquisition of accessories and equipment, as well as team training on a daily basis. This study aimed to report the procedures from the Gastrocentro Unit in the treatment of gastric and colorectal lesions by ESD.
MATERIALS AND METHODSFor ESD to be indicated, the macroscopic aspect and histological diagnosis, which had previously been obtained by endoscopic biopsies, were considered. The Japanese Association of Gastric Cancer criteria (5,6) were used for gastric resections, and the criteria of Uraoka et al. and Saito et al. (7,8) were used for colorectal lesions. Endoscopic with digital cromoscopy and magnification (EG 590ER and EC 590 ZWL, Fujinon Inc., Japan) were used.
Extended criteria and indications for gastric ESD.
| Extended criteria and indications for gastric ESD |
|---|
| Adenocarcinoma differentiated from the mucosa without ulcer, independent of the size |
| Adenocarcinoma differentiated from the mucosa with ulcer <3 cm |
| Adenocarcinoma undifferentiated from the mucosa with ulcer smaller than 2 cm |
Criteria and indications for ESD in the colon.
| Criteria and indications for ESD of the colon |
|---|
| Endoscopic aspects of non-invasion determined by cromoendoscopy and magnification are criteria for ESD and EMR |
| Definite indications for ESD: |
| LST – non-granular >20 mm |
| Relative indications for ESD: |
| LST - granular > = 40 mm |
| Large villous tumors |
| Intramucosal lesions |
| Residual or recurrent lesions showing no lifting signs after EMR |
| Definite indications for EMR and EMR piece-meal |
| Lesions smaller than 20 mm |
| LST-G larger than 20 mm but smaller than 40 mm |
The accessories used for resection included a needle knife (Olympus Inc. Japan), IT Knife2 (Olympus Inc., Japan) and a flush knife (Fuji Inc., Japan), as well as a transparent tip hood attached to the endoscopes and a high-frequency electric scalpel (Olympus Inc., Japan).
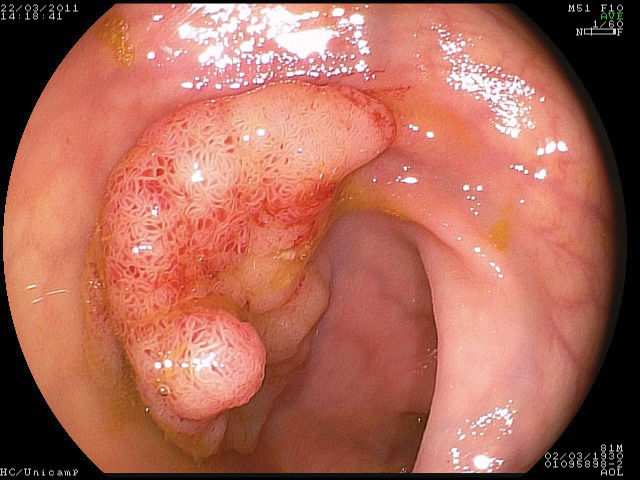
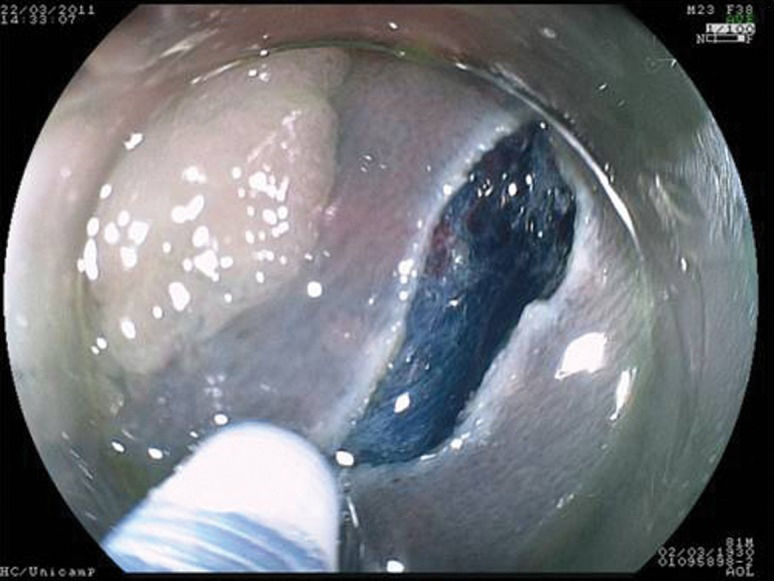
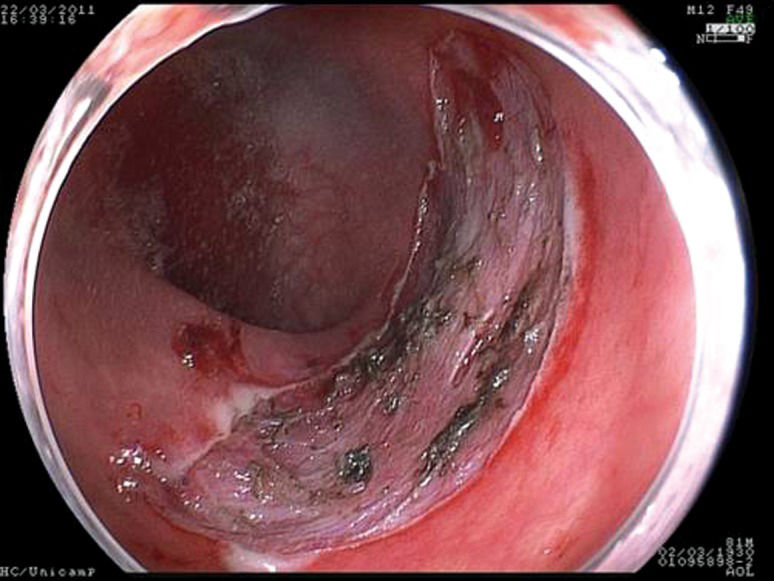
The following steps were standardized and followed in all procedures: identification of the lesion; cromoscopy with 0.4% indigo carmine; demarcation of the periphery of the lesion with a needle knife; submucosal injection of a solution containing 250 ml of 10% mannitol, 1 ml of epinephrine and 2 ml of 0.4% indigo carmine or hyaluronic acid; and completion of small incisions circumferentially to the lesion made with the needle knife and submucosal dissection with the IT knife and/or flush knife. After resection, hemostasis was conducted when necessary with electrical cauterization or endoscopic clips. The investigation period spanned from the beginning of the demarcation to the end of the resection.
Histopathological analysisSpecimens were prepared in Styrofoam plaque, measured at the major axis and fixed in 10% formaldehyde. The histopathological study (Pathological Unit Gastrocentro) followed the norms recommended by the Japanese Association of Gastric Cancer (9) and Japanese Society and Research of Colon and Rectum Cancer (10). The following aspects were considered: tumor differentiation, depth of invasion and lateral and depth margins.
Pre-procedure and follow-up protocolsMannitol (10%) was used for bowel cleansing, and cephalosporin was used as a prophylactic. The patients remained in the hospital for 24 hours. For gastric resection, patients received 40 mg of proton bomb inhibitors for 56 days. Endoscopic follow-ups were performed at three and six months after the procedure for an evaluation of macroscopic relapse. Biopsies were performed in the resectioned areas. After six months, control exams were conducted annually.
RESULTSBetween June 2010 and March 2011, 16 lesions were resected. Of these, nine were gastric lesions, and seven were lesions of the colon or rectum. Three endoscopists (Gastrocentro) with previous training in ESD performed the procedures. All lesions were resected en bloc.
Gastric resectionsThe mean age of the patients was 65 (58-73) years, and all patients were male. Four patients were carriers of hepatic cirrhosis. The pre-procedure diagnosis was adenoma in four patients and well-differentiated adenocarcinoma in five patients. The average size of the lesions was 28.6 (20-45) mm; five lesions were located in the body, and four were located in the antrum. Regarding endoscopic aspects, five lesions were type IIa, and four were type IIa+IIc. The average duration of gastric resections was 103 (60-240) min, with no immediate complications. There was one complication at follow-up in a lesion located in the pyloric region. Asymptomatic stenosis was identified three months after the procedure with successful endoscopic dilatation.
The post-procedure diagnoses were as follows: adenoma (n = 4), with one patient who had a lesion with a focal area of intra-mucosal carcinoma; slightly differentiated adenocarcinoma with SM1 invasion (n = 1); and well-differentiated adenocarcinoma (n = 4), with one patient demonstrating M2 invasion and vascular invasion, one patient demonstrating M3 invasion with vascular invasion and two patients with SM1 invasion without vascular or lymphatic invasion. All gastric lesions presented lateral margins and depths free of lesion.
Colorectal resectionsThe mean age of the patients was 54 (45-60) years, and the four patients were male (57%). Four lesions were located in the rectum, one was in the sigmoid colon, one was in the hepatic flexure and one was in the ascending colon. All were lateral-spreading tumors, with a medium size of 26 (20-50) mm. Four were granular, one was non-granular and two were of the mixed type. The average duration of resection was 163 (80-240) min. There were two complications, both in the rectal resections, which were treated during the procedure: hemorrhaging without the need for blood transfusion, which was controlled by cauterization, and one perforation that was treated with an endoscopic clip. The latter patient remained in the hospital for five days. The histopathological study of the resected specimens demonstrated the presence of a hyperplasic lesion (n = 1), adenoma associated with carcinoma in situ (n = 1) and five adenomas. One lesion presented a compromised lateral margin without relapse 30 days after resection. In all of cases, the depth margins were free of lesions, and there were no subsequent complications.
Clinical aspects.
| Gastric lesions (n = 9) | Colorectal lesions (n = 7) | |
|---|---|---|
| Average age | 65 (58-73) | 54 (45-60) |
| Male | 9 (100%) | 4 (57%) |
| Hepatic cirrhosis | 4 | 0 |
| Localization | ||
| Antrum/body | 4/5 | |
| Rectum/sigmoid/hepatic flexure/ascending colon | 4/1/1/1 | |
| Average size (mm) | 28,6 28.6(20-45) | 26 (20-50) |
| Endoscopic type I/IIa/IIa+IIc | 0/5/4 | |
| Granular/non-granular/mixed | 4/1/2 | |
| Average duration (min.) | 103 (60-240) | 163 (80-42) |
| Histological type | ||
| Adenoma/ adenoma+adenocarcinoma | 3/10 | 5/1 |
| Adenocarcinoma well differentiated/less differentiated | 4/1 | 1/0 |
| Invasion M1/ M2/M3/Sm1/Sm2 | 0/1/1/2/0 | 1/0/0/0/0 |
| Lymphatic/vascular Invasion | 1/1 | 0 |
| Compromised lateral margins | 0 | 1 |
| Complications: bleeding/perforation/stenosis | 0/0/1 | 1/1 /0 |
| Mortality | 0 | 0 |
ESD is a superior technique because it allows en bloc resections with appropriate histological analysis. It also results in lower rates of relapse as compared to EMR (11). In the stomach, the incidence of local recurrence with EMR varies from 2.3% to 36.5% as compared to 0% to 2% with ESD (12). For en bloc resection of gastric lesions smaller than 10 mm, the success rates are similar for both techniques (13). However, for lesions larger than 20 mm, resection with the ESD technique demonstrates success rates higher than 90% (14). Similarly, in colon and rectal lesions, the success rates of en bloc resection for lesions larger than 20 mm may be 30% less with EMR (15-17).
Despite some advantages in comparison to EMR, ESD is more difficult to perform and presents a higher incidence of complications (10,11),. In Brazil, its use was postponed due to the unavailability of appropriate accessories and the lack of training for endoscopists.
Indication for local resection is based on an appropriate presurgical endoscopic evaluation because the lesion must be confined to the wall without massive submucosal invasion. The evaluation for colorectal lesions is easier because the possibility of lymph node metastasis occurs only in the presence of SM3 (10,17,18). Evaluation is more difficult with gastric lesions, however, due to the more aggressive biological behavior of adenocarcinoma in this organ. The incidence of lymph node metastasis for lesions confined to the mucosa varies from 0% to 3% and from 9% to 19% in the submucosa (21). In the stomach, we must consider tumor differentiation, size of the lesion and presence of depression (12),.
In the colon and rectum, small lesions can be resected en bloc with the EMR technique (25,26). However, difficulties associated with endoscopic treatment in larger lesions can lead to additional surgical treatment (25,27). Currently, resection with ESD has been more frequently used, as this technique enables en bloc resection. In post-EMR situations in which the presence of relapse has been identified, resection with ESD can be performed, even with the presence of fibrosis caused by a previous resection.
Saito Y et al. (16) compared EMR with ESD for colon and rectal lesions larger than 20 mm and reported a longer time for ESD (108 min as compared to 29 min; p<0.0001), in addition to a higher en bloc resection rate (84% compared with 33%; p<0.0001) and lower rate of relapse (2% compared with 14%). Furthermore, the perforation and late bleeding rates in the ESD and EMR groups were 6.2% and 1.3%, respectively, and 1.4% and 3.1%, respectively (p<0.05).
For the correct evaluation of flat lesions, which have been associated with higher rates of submucosal invasion (17), the employment of endoscopes with high definition and magnification is useful. Furthermore, high-definition endoscopes are considered essential for the identification of aspects that can be related to invasion (31-33) and the differentiation of malignant lesions (34,35). In the present study, cromoscopy and magnification were employed in all cases. Morphological study of the lesions is also important to evaluate the correct indication for endoscopic resection. Colorectal lesions of lateral growth (LST) with elevation have been associated with submucosal invasion, and approximately 30% of non-granular LST lesions present multifocal submucosal invasion with difficult evaluation when the specimen is submitted to “piece-meal” resection (8). Therefore, for appropriate histological study, en bloc resection is essential but difficult when employing EMR (16,17,20,36,37) (Figures 1–3).
There are some technical details that are specific for performing ESD in the colon and rectum; for example, appropriate intestinal preparation, mucous removal and the use of indigo carmine are of extreme importance (31). The use of gastroscopes is preferred for colonoscopies due to the flexibility of gastroscopes and because most resection frequently occurs in retroversion. Because the colon wall is very thin, elevation of the lesion with larger volumes of saline or a similar solution decreases the risk of perforation. Several solutions have been used for submucosal injection (15,36), and in the present study, Mannitol and hyaluronic acid with indigo carmine were used, facilitating the visualization of the best resection plane. Ideally, submucosal elevation should be maintained throughout the procedure, but many injections are necessary to achieve this goal. The use of hyaluronic acid makes the procedure more expensive but also facilitates dissection because it maintains submucosal elevation for a longer duration.
The duration of ESD and the incidence of complications are directly related to the training and knowledge of the procedure. The average gastric resection time was 119 min (60-240 min), and the most difficult resections were performed in the posterior wall of the pre-pyloric region, cardia and incisura angularis. The average time for colon and rectal resection was 163 min. The rectal lesion that was 55 mm in size was the most difficult and required the longest time (420 min) due to bleeding that occurred during the procedure. As endoscopic bleeding is difficult to control due to the small visual field, the endoscopist is required to control the bleeding at all times while maintaining a clean resection area.
In addition, the identification of vessels is very important, and they must be coagulated before continuing the dissection. Therefore, it is vital that the endoscopist has a good view and uses a “cap” at the tip of the endoscope.
In gastric resections, bleeding is the most frequent complication. Oka et al. (11) reported bleeding incidences during and after EMR of 7.6% and 3.9% as compared to 22.6% and 6.2% after ESD. However, the necessity of a blood transfusion is rare.
The incidence of gastric perforation post-EMR and ESD can vary from 0% to 4% and 0% to 5%, respectively. Watanabe et al. (14) did not observe any differences related to the occurrence of perforation when comparing EMR with ESD (3.2% versus 4.2%, p>0.05). However, Oda et al. (19) reported that the incidence of perforation for EMR was 0.5% as compared to 9.7% for ESD. The most common complication in colorectal procedures is perforation, which demonstrates rates between 2.3% and 10.4%, followed by bleeding (19,28,38,39).
In this casuistic, gastric resections did not present early complications, in contrast with colorectal procedures. One case of bleeding in rectal resection was controlled with endoscopic clips and electrical cauterization. No hemodynamic instability was observed, and a transfusion was not necessary. However, the bleeding made the procedure last much longer. The authors also note that the bleeding was more frequent in rectal resections than colon resections. In another case, rectal perforation was easily diagnosed during the procedure and successfully treated with endoscopic clips and antibiotics. The patient remained in the hospital for five days without any other complications. As the ESD procedure is long, it is frequently accompanied by abdominal pain secondary to colon distension with air, which can be minimized with the use of carbonic gas (40).
Despite initial difficulties, en bloc resection was possible in all cases and may be an impetus for training our team in this technique.
The initiation of endoscopic submucosal resection in the Gastrocentro Unit was accompanied with difficulties at the onset related to the acquisition of materials and procedure cost. However, this new technique also brought a series of advantages, such the training of the nursing team in complex procedures and training of endoscopists in the morphological evaluation of lesions and more precise handling of the endoscope. Technological incorporation and histopathological study of specimens by pathologists were additional benefits. The Gastrocentro Unit is not a pioneer for ESD in Brazil, and there are a few centers that perform this procedure routinely. Thus, we hope that the use of ESD will become more prevalent among these others centers.
Although it is more difficult and has a higher rate of complications in colorectal procedures, our results demonstrate that ESD remains a feasible technique.
No potential conflict of interest was reported.
Santos JO contributed to the design, data collection and analysis, text elaboration and performed the endoscopy surgery. Miyajima N performed the endoscopy surgery and contributed to the discussion. Carvalho R contributed to the pathology analysis and contributed to the discussion. Leal RF contributed to the discussion. Ayrizo ML contributed to the data collection and discussion. Coy CS contributed to the design, data analysis, text elaboration and performed the endoscopy surgery.