Patients with end stage liver disease may become critically ill prior to LT requiring admission to the intensive care unit (ICU). The high acuity patients may be thought too ill to transplant; however, often LT is the only therapeutic option. Choosing the correct liver allograft for these patients is often difficult and it is imperative that the allograft work immediately. Donation after cardiac death (DCD) donors provide an important source of livers, however, DCD graft allocation remains a controversial topic, in critically ill patients. Between January 2003-December 2008, 1215 LTs were performed: 85 patients at the time of LT were in the ICU. Twelve patients received DCD grafts and 73 received donation after brain dead (DBD) grafts. After retransplant cases and multiorgan transplants were excluded, 8 recipients of DCD grafts and 42 recipients of DBD grafts were included in this study. Post-transplant outcomes of DCD and DBD liver grafts were compared. While there were differences in graft and survival between DCD and DBD groups at 4 month and 1 year time points, the differences did not reach statistical significance. The graft and patient survival rates were similar among the groups at 3-year time point. There is need for other large liver transplant programs to report their outcomes using liver grafts from DCD and DBD donors. We believe that the experience of the surgical, medical and critical care team is important for successfully using DCD grafts for critically ill patients.
Liver transplantation (LT) is a life saving procedure for patients with end stage liver disease. Patients with end stage liver disease may become critically ill prior to LT requiring admission to the intensive care unit (ICU) and support including vasopressors, mechanical ventilation, and dialysis.1 There are often major concerns regarding patient survival and subsequent graft loss after LT. The high acuity patients may be thought too ill to transplant, however, often LT is the only therapeutic option. Choosing the correct liver allograft for these patients is often difficult and it is imperative that the allograft work immediately. In addition, there is a significant disparity between organ availability and the number of patients waiting for LT. Donation after cardiac death (DCD) donors provide an important source of livers that has been used to expand the donor pool.2 However, DCD livers are considered inferior grafts because of higher risk for primary non-function (PNF), hepatic artery thrombosis (HAT), and ischemic cholangiopathy (IC).3,4 DCD liver allograft allocation remains a controversial topic, especially in this high risk population of critically ill patients.5,6
We previously reported our experience in DCD liver recipients.7,8 Our liver transplant program has performed over 200 LT using liver grafts from DCD donors. To date there has not been any specific report in the literature regarding the short-and long-term graft and patient outcomes when DCD grafts were used for critically ill patients in the ICU. We reviewed our program's experience, specifically addressing peri-operative complications and short-and long-term outcomes of critically ill patients who received DCD liver grafts.
Material and MethodsMayo Clinic Florida database was searched for all patients between January 2003-December 2008 for patients who underwent LT while in the ICU. Of 1,215 LTs, 85 patients at the time of LT were in the ICU. Twelve patients received DCD grafts and 73 received donation after brain dead (DBD) grafts. Re-transplants and recipients receiving combined organ transplants were excluded, yielding 8 recipients of DCD grafts and 42 recipients of DBD grafts. A 2:1 match between recipients of DBD and DCD liver grafts using recipient age (± 10 years) and MELD score (± 5 points or both > 30 points) was attempted, however this proved difficult. For a more detailed analysis we were only able to match 8 DCD recipients to 13 DBD recipients using the aforementioned matching criteria. For a larger picture, the DBD recipients as a cohort were also reviewed and their variables were compared to those of DCD recipients. Group 1, includes the 8 recipients of DCD grafts. Group 2, includes 13 recipients in matched DBD control group. Group 3, includes 42 DBD recipients transplanted out of the ICU. Patient demographics including age, sex, primary liver disease, calculated Model for End-Stage Liver Disease (MELD) score on the day of LT, hospital or ICU stay care after LT, and complications. Operative data included recipient warm ischemia time (WIT) defined as time of removal of liver allograft from cold solution until reperfusion, operative time, and number of units packed red blood cells (PRBC) transfused. Donor characteristics recorded included donor age, race, sex, DCD warm ischemia time (DWIT) defined as withdrawal of support to initial flush of preservation solution, and cold ischemia time (CIT) defined as initial flush of preservation solution until the removal from cold in the recipient operation. Donor Risk Index (DRI) was calculated retrospectively for each case. Acute Physiologic and Chronic Health Evaluation II score (APACHE II) was also calculated for each patient based on data during the first 24 h of ICU admission and the 24 h prior to LT.
The study protocol was reviewed and approved by Mayo Clinic Institutional Review Board.
In DCD donors, withdrawal of support and declaration of death were in strict compliance with donor hospital policies. The transplant team was not involved in the withdrawal process. An independent physician from the donor hospital, separate from the organ procurement organization and the transplant center, was assigned to withdraw artificial life support and provide end of life care to the patient. Following the declaration of death by the independent physician a further 2-5 min of mandatory observation was performed as described in the 1997 Institute of Medicine Guidelines.9 During the 2-5 minute waiting period, the patient was transported to the operating room (if not already there) and prepared for organ recovery. Heparin was administered to the patient according to the donor hospital policy. Following the 2-5-min wait period a rapid retrieval technique was performed as described previously.7,8 All liver grafts were procured by surgeons from our center and University of Wisconsin solution was used for flush and organ preservation for all DCD and DBD procurements. All LT were performed using the piggyback technique without a porto-caval shunt, venonenous bypass or caval clamping. Standard triple-drug immunosuppression with tacroli-mus, mycophenolate mofetil and corticosteroids was used in all cases.
Comparisons between DCD vs. matched DBD recipients, and DCD vs. overall DBD groups were assessed separately. Mann-Whitney U test and chi-square test were utilized to examine continuous and categorical variables, respectively. Kaplan-Meier method and log-rank test was used to estimate and compare graft and patient survival among the groups. Level of significance was set at a P value of < 0.05. All analyses were performed using SPSS version 17 (Chicago, IL, USA).
ResultsBetween January 2003 and December 2008, 50 critically ill patients in the ICU underwent primary LT. Forty-two patients received DBD liver grafts while 8 patients received DCD liver grafts.
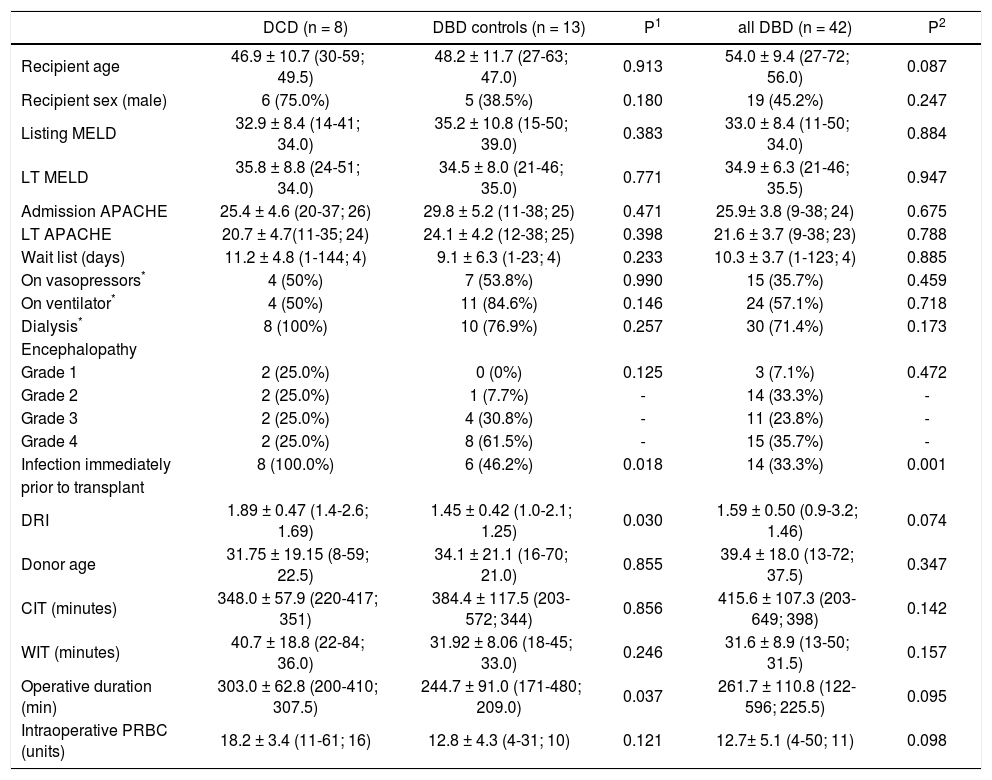
Statistically significant differences among the groups were not apparent with respect to recipient age, raw MELD scores at the time of listing and at LT, APACHE II scores at ICU admission and at LT, wait list days and pre-LT ICU length of stay (Tables 1 and 2). Metabolic encephalopathy, acute renal failure requiring renal replacement therapy, GI hemorrhage, respiratory failure, acute/acute on chronic liver failure were causes of ICU admission (Table 1). Percentages of patients on vasopressors immediately before LT in each group were similar. Likewise, percentages of patients on renal replacement therapy and on ventilator were similar in each study group (Table 1).
Recipient and donor demographics and intra-operative data.
| DCD (n = 8) | DBD controls (n = 13) | P1 | all DBD (n = 42) | P2 | |
|---|---|---|---|---|---|
| Recipient age | 46.9 ± 10.7 (30-59; 49.5) | 48.2 ± 11.7 (27-63; 47.0) | 0.913 | 54.0 ± 9.4 (27-72; 56.0) | 0.087 |
| Recipient sex (male) | 6 (75.0%) | 5 (38.5%) | 0.180 | 19 (45.2%) | 0.247 |
| Listing MELD | 32.9 ± 8.4 (14-41; 34.0) | 35.2 ± 10.8 (15-50; 39.0) | 0.383 | 33.0 ± 8.4 (11-50; 34.0) | 0.884 |
| LT MELD | 35.8 ± 8.8 (24-51; 34.0) | 34.5 ± 8.0 (21-46; 35.0) | 0.771 | 34.9 ± 6.3 (21-46; 35.5) | 0.947 |
| Admission APACHE | 25.4 ± 4.6 (20-37; 26) | 29.8 ± 5.2 (11-38; 25) | 0.471 | 25.9± 3.8 (9-38; 24) | 0.675 |
| LT APACHE | 20.7 ± 4.7(11-35; 24) | 24.1 ± 4.2 (12-38; 25) | 0.398 | 21.6 ± 3.7 (9-38; 23) | 0.788 |
| Wait list (days) | 11.2 ± 4.8 (1-144; 4) | 9.1 ± 6.3 (1-23; 4) | 0.233 | 10.3 ± 3.7 (1-123; 4) | 0.885 |
| On vasopressors* | 4 (50%) | 7 (53.8%) | 0.990 | 15 (35.7%) | 0.459 |
| On ventilator* | 4 (50%) | 11 (84.6%) | 0.146 | 24 (57.1%) | 0.718 |
| Dialysis* | 8 (100%) | 10 (76.9%) | 0.257 | 30 (71.4%) | 0.173 |
| Encephalopathy | |||||
| Grade 1 | 2 (25.0%) | 0 (0%) | 0.125 | 3 (7.1%) | 0.472 |
| Grade 2 | 2 (25.0%) | 1 (7.7%) | - | 14 (33.3%) | - |
| Grade 3 | 2 (25.0%) | 4 (30.8%) | - | 11 (23.8%) | - |
| Grade 4 | 2 (25.0%) | 8 (61.5%) | - | 15 (35.7%) | - |
| Infection immediately | 8 (100.0%) | 6 (46.2%) | 0.018 | 14 (33.3%) | 0.001 |
| prior to transplant | |||||
| DRI | 1.89 ± 0.47 (1.4-2.6; 1.69) | 1.45 ± 0.42 (1.0-2.1; 1.25) | 0.030 | 1.59 ± 0.50 (0.9-3.2; 1.46) | 0.074 |
| Donor age | 31.75 ± 19.15 (8-59; 22.5) | 34.1 ± 21.1 (16-70; 21.0) | 0.855 | 39.4 ± 18.0 (13-72; 37.5) | 0.347 |
| CIT (minutes) | 348.0 ± 57.9 (220-417; 351) | 384.4 ± 117.5 (203-572; 344) | 0.856 | 415.6 ± 107.3 (203-649; 398) | 0.142 |
| WIT (minutes) | 40.7 ± 18.8 (22-84; 36.0) | 31.92 ± 8.06 (18-45; 33.0) | 0.246 | 31.6 ± 8.9 (13-50; 31.5) | 0.157 |
| Operative duration (min) | 303.0 ± 62.8 (200-410; 307.5) | 244.7 ± 91.0 (171-480; 209.0) | 0.037 | 261.7 ± 110.8 (122-596; 225.5) | 0.095 |
| Intraoperative PRBC (units) | 18.2 ± 3.4 (11-61; 16) | 12.8 ± 4.3 (4-31; 10) | 0.121 | 12.7± 5.1 (4-50; 11) | 0.098 |
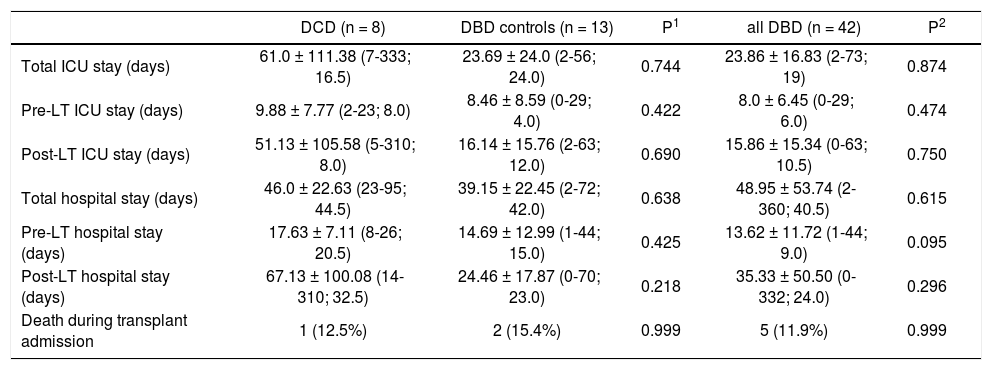
Peri-transplant information.
| DCD (n = 8) | DBD controls (n = 13) | P1 | all DBD (n = 42) | P2 | |
|---|---|---|---|---|---|
| Total ICU stay (days) | 61.0 ± 111.38 (7-333; 16.5) | 23.69 ± 24.0 (2-56; 24.0) | 0.744 | 23.86 ± 16.83 (2-73; 19) | 0.874 |
| Pre-LT ICU stay (days) | 9.88 ± 7.77 (2-23; 8.0) | 8.46 ± 8.59 (0-29; 4.0) | 0.422 | 8.0 ± 6.45 (0-29; 6.0) | 0.474 |
| Post-LT ICU stay (days) | 51.13 ± 105.58 (5-310; 8.0) | 16.14 ± 15.76 (2-63; 12.0) | 0.690 | 15.86 ± 15.34 (0-63; 10.5) | 0.750 |
| Total hospital stay (days) | 46.0 ± 22.63 (23-95; 44.5) | 39.15 ± 22.45 (2-72; 42.0) | 0.638 | 48.95 ± 53.74 (2-360; 40.5) | 0.615 |
| Pre-LT hospital stay (days) | 17.63 ± 7.11 (8-26; 20.5) | 14.69 ± 12.99 (1-44; 15.0) | 0.425 | 13.62 ± 11.72 (1-44; 9.0) | 0.095 |
| Post-LT hospital stay (days) | 67.13 ± 100.08 (14-310; 32.5) | 24.46 ± 17.87 (0-70; 23.0) | 0.218 | 35.33 ± 50.50 (0-332; 24.0) | 0.296 |
| Death during transplant admission | 1 (12.5%) | 2 (15.4%) | 0.999 | 5 (11.9%) | 0.999 |
Data is presented as mean ± SD (range; median). P1: DCD vs. DBD controls. P2: DCD vs. all DBD.
Nine out of the 42 DBD recipients presented with acute liver failure (ALF); four patients were in the matched DBD group; none of the DCD recipients had ALF.
Cold ischemia and warm ischemia times were similar among the groups. Total operative time was higher in the DCD group in comparison to the matched DBD group (p = 0.037). Similarly, intraoperative PRBC transfusion requirement was higher in DCD group; however, this difference did not reach statistical significance. DCD grafts had statistically higher DRI scores when compared to those in the matched DBD group (p = 0.030).
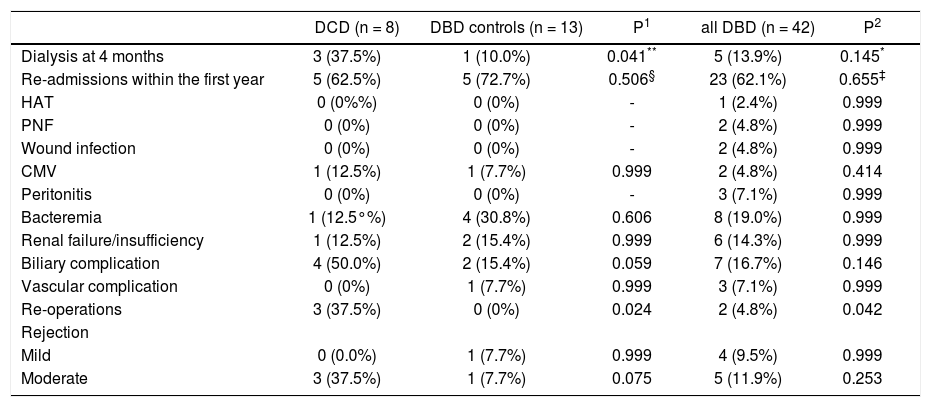
Total ICU and hospital stay were similar among the groups (Table 2). There were no intra-operative deaths. One patient (12.5%) in the DCD group, 2 patients (15.4%) in matched DBD group and 5 patients (11.9%) in DBD cohort died during transplant hospitalization. In the DCD group, 4 patients (50%) had biliary complications while 2 patients (15.4%) in matched DBD group and 7 patients (16.7%) in overall DBD group had biliary complications (Table 3). Five patients (62.5%) in the DCD group, 5 patients (45.5%) in matched DBD group and 23 patients (62.1%) in DBD cohort required readmission within the first year following LT (p = NS). These patients had complicated post-transplant course: excluding 1 patient who died in the perioperative period, 4 patients (57.1%) in the DCD group had biliary complications requiring reoperation and/or endoscopic procedures; 3 patients (42.8%) remained on renal replacement therapy at 4 month time point; 3 patients (42.8%) had moderate acute rejection requiring steroid bolus treatment. None of the patients in the DCD group had IC.
Complications after LT.
| DCD (n = 8) | DBD controls (n = 13) | P1 | all DBD (n = 42) | P2 | |
|---|---|---|---|---|---|
| Dialysis at 4 months | 3 (37.5%) | 1 (10.0%) | 0.041** | 5 (13.9%) | 0.145* |
| Re-admissions within the first year | 5 (62.5%) | 5 (72.7%) | 0.506§ | 23 (62.1%) | 0.655‡ |
| HAT | 0 (0%%) | 0 (0%) | - | 1 (2.4%) | 0.999 |
| PNF | 0 (0%) | 0 (0%) | - | 2 (4.8%) | 0.999 |
| Wound infection | 0 (0%) | 0 (0%) | - | 2 (4.8%) | 0.999 |
| CMV | 1 (12.5%) | 1 (7.7%) | 0.999 | 2 (4.8%) | 0.414 |
| Peritonitis | 0 (0%) | 0 (0%) | - | 3 (7.1%) | 0.999 |
| Bacteremia | 1 (12.5°%) | 4 (30.8%) | 0.606 | 8 (19.0%) | 0.999 |
| Renal failure/insufficiency | 1 (12.5%) | 2 (15.4%) | 0.999 | 6 (14.3%) | 0.999 |
| Biliary complication | 4 (50.0%) | 2 (15.4%) | 0.059 | 7 (16.7%) | 0.146 |
| Vascular complication | 0 (0%) | 1 (7.7%) | 0.999 | 3 (7.1%) | 0.999 |
| Re-operations | 3 (37.5%) | 0 (0%) | 0.024 | 2 (4.8%) | 0.042 |
| Rejection | |||||
| Mild | 0 (0.0%) | 1 (7.7%) | 0.999 | 4 (9.5%) | 0.999 |
| Moderate | 3 (37.5%) | 1 (7.7%) | 0.075 | 5 (11.9%) | 0.253 |
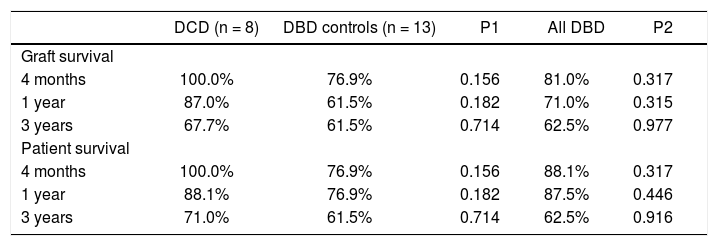
While there were differences in graft and survival among the groups at 4 month and 1 year time points, the differences did not reach statistical significance (Table 4). The graft and patient survival rates were similar among the groups at 3-year time point.
Graft and patient survival.
| DCD (n = 8) | DBD controls (n = 13) | P1 | All DBD | P2 | |
|---|---|---|---|---|---|
| Graft survival | |||||
| 4 months | 100.0% | 76.9% | 0.156 | 81.0% | 0.317 |
| 1 year | 87.0% | 61.5% | 0.182 | 71.0% | 0.315 |
| 3 years | 67.7% | 61.5% | 0.714 | 62.5% | 0.977 |
| Patient survival | |||||
| 4 months | 100.0% | 76.9% | 0.156 | 88.1% | 0.317 |
| 1 year | 88.1% | 76.9% | 0.182 | 87.5% | 0.446 |
| 3 years | 71.0% | 61.5% | 0.714 | 62.5% | 0.916 |
P1: DCD vs. DBD controls. P2: DCD vs. all DBD.
LT is the only treatment modality for patients with end-stage liver disease. Success of LT depends on recipient's overall condition, the quality of the liver graft, as well as intra-operative factors. In patients who need urgent liver transplantation, selection of appropriate liver grafts affect short and long-term outcomes. The optimal distribution of available cadaveric liver grafts has been a crucial part of liver transplant practice.10 While in an ideal system every liver transplant candidate receives an appropriate liver graft, a dilemma emerges as a result of the significant discrepancy between the supply and demand of liver grafts: should any available liver graft be used for a patient in the ICU with liver failure or should we wait for a good quality liver graft with the inherent risk of patient death on the wait list or increased risk of disease progression to an unacceptable surgical risk for that particular patient.11 Also, from ethical standpoint a utility versus equality dilemma emerges: is it justified to use scarce organs for sick patients in the ICU-with the understanding of potentially poor prognosis-or should those organs be used for patients with relatively mild liver disease? DCD has emerged as a source for liver grafts that can alleviate the discrepancy between the supply and demand of livers. DCD numbers increased in the first part of the last decade, however, the enthusiasm of using these grafts declined after 2007 because of lower graft survival rates and significant biliary problems related to these grafts.8,12 Liver grafts from DCD donors are considered higher risk organs.13 Therefore, the decision to use a DCD organ for a critically ill recipient must balance higher rates of complications and graft failure against that recipient's imminent risk of death from liver failure.
Our program started using liver grafts from DCD donors in 1998. To date we have performed over 200 LT using DCD grafts representing the largest single institution experience in the US.8 While DCD procurement and use requires expertise, its use in critically ill patients should be balanced according to the program's previous DCD results versus availability of liver grafts in a particular region. Wait list time in our program has been one of the shortest in the US.14 Because of higher availability of liver grafts in our region, we have not needed to use DCD grafts very often for critically ill patients, as their MELD scores are often higher and patients are likely to receive a donor organ in a short time interval regardless of donor type. Ultimately the decision to accept a DCD liver graft should be based on close assessment of the LT candidate in the ICU and frequent communication between the accepting surgeon and the critical care team. A team decision regarding the risks of higher donor risk and subsequent morbidity and mortality vs. prolonged wait for transplant with also subsequent risk of morbidity and mortality should be considered. It is also now evident that transplant program's volume correlates with the results of using high risk organs: higher volume centers may have additional experience in using high-risk donors that could lead to greater allograft and recipient survival compared to low volume programs when grafts with higher risk profiles are used.15 Using high risk donors may result in reduced median transplant MELD scores and wait times which improve outcome and optimize organ utilization. This potential benefit must be weighed against the knowledge that high MELD and DRI are independent factors for graft failure.16 However, MELD and DRI as continuous variables are only 2 of the independent factors for graft failure, and other variables outside of MELD and DRI calculation such as patient age, pre-transplant functional status, and hemodynamic support requirements in the immediate pre-transplant period also play a significant role. When using high-risk donor grafts, experience and improved judgment comes from a combination of high procedure volume, adequate resource allocation and collaboration between the physicians taking care of these patients.
In our study, the recipients of DCD and DBD grafts had similar raw MELD and APACHE scores at the time of listing and transplantation. As a reflection of similar severity of illness, the percentages of patients on vasopressors, on the ventilator support and those requiring dialysis before LT were similar. There were no differences in ICU or hospital stay among the groups. DCD recipients required longer operating times, and received more intra-ope-rative PRBC transfusions, possibly as a result of early graft dysfunction that is more common with DCD liver grafts. Re-admission to the hospital during the first post-transplant year was common in all study groups; more than half of the patients were admitted to the hospital after initial transplant hos-pitalization and there were no differences among the study groups. In the recipients of DCD liver grafts, biliary complications were more frequent; however, this did not reach statistical significance, most likely due to small number of patients in the study groups. None of the recipients of DCD grafts experienced IC; all biliary complications were extra-hepatic bile duct complications such as bile leaks and strictures. This finding is consistent with our report on our DCD experience: the development of IC was not correlated with the degree of sickness of the recipient reflected by MELD score.8 Similar to our previous published experience with DCD grafts, the patient and graft survival at 4 months, 1 year and 3 year were similar for recipients of DCD compared to recipients of DBD grafts.8
The most appropriate use of scarce liver allografts continues to evolve. As the gap between supply and demand for liver grafts is widening world wide, livers from DCD donors should be considered. DCD grafts have been used more extensively since 1997 when Institute of Medicine determined that these organs are medically effective and ethically acceptable. DCDs are considered to be less than optimal for transplantation because of damaging effect of variable warm ischemia time before cold preservation. The combination of the limited donor organ pool, increased number of patients dying on the waiting list and financial constraints with a failing graft challenges the current organ allocation system to provide efficient distribution of a limited resource. The discrepancy between the number of available organs and increasing number of potential recipients will worsen until significant future advances are made in providing alternative management for chronic liver disease. Allocation of scarce liver grafts requires balancing of ethical principles: beneficence, fairness and utility.
This study summarizes our single center experience using DCD donors in critically ill recipients managed with uniform surgical technique and medical protocols throughout the study period with complete long-term follow-up. It also demonstrates the outcomes in these high-risk group of transplant candidates and the complications expected even with non-DCD donors. As such, it represents a unique contribution to the literature, but our study is limited by the relatively small sample size of critically ill patients who received DCD liver grafts, preventing multivariable analysis to determine overlapping factors.
The relative excess of ALF in the DBD group undoubtedly contributes to the higher acuity and delayed recovery of this group relative to other DBD recipients in the ICU. This aspect and the small numbers leave open the likelihood of a type 2 statistical error.
In conclusion, utilization of DCD liver grafts for critically ill patients resulted in similar graft and patient outcomes. There is need for other large liver transplant programs to report their outcomes using liver grafts from DCD and DBD donors. We believe that the experience of the surgical, medical and critical care team is important for successfully using DCD grafts for these critically ill patients. While utilization of DCD grafts is associated with higher rate of biliary problems, it can provide a chance for early LT and avoidance of a futile LT or death on the wait list. Each LT program should decide whether to use these grafts based on their overall experience with high-risk donors, the acuity of their critically ill patient and availability of liver grafts in their region.