Rheumatoid arthritis (RA) is an autoimmune disease that is mainly characterized by joint deterioration and decreased bone mineral density. The Dickkopf 1 protein (DKK1) exerts a negative regulatory function of the Wnt pathway involved in the differentiation of osteoblasts, and has been observed to be overexpressed in patients with RA.
ObjectiveTo provide updated information on current knowledge about the relationship between DKK1 serum levels and the presence of bone and joint damage in RA patients.
MethodA qualitative systematic review was carried out in the PubMed, Embase, Cochrane and Scielo databases using the terms Dickkopf 1, DKK1, Dickkopf related protein 1, Rheumatoid Arthritis, and Bone biomarker.
ResultsA total of 12 studies were chosen that met the requirements of the search. These included 7 prospective cohorts, 4 cross-sectional studies, and 1 clinical trial. Of the 12 studies reviewed, 10 analyzed the relationship between serum DKK1 levels and the presence of bone damage as the primary outcome. One of them analyzed this relationship as a secondary outcome and another one the RSP01/DKK1 ratio. The results to date seem to indicate that DKK1 could have an active role in advanced stages of RA, but not in the initial phase.
ConclusionsThe DKK1 protein plays an essential pathophysiological role in the decrease of bone mass and joint remodelling, depending on the stage of the disease in patients with RA. Its role as a biomarker or therapeutic strategy would be an interesting alternative still under study.
La artritis reumatoide (AR) es una enfermedad autoinmune caracterizada principalmente por deterioro articular y disminución de la densidad mineral ósea. La proteína Dickkopf 1 (DKK1) ejerce una función reguladora negativa de la vía Wnt comprometida con la diferenciación de osteoblastos y se ha observado que puede estar sobreexpresada en pacientes con AR.
ObjetivoProveer información actualizada sobre el conocimiento de la asociación entre los niveles séricos de DKK1 y la presencia de daño óseo y articular en pacientes con AR.
MétodoSe realizó una revisión sistemática cualitativa en las bases de datos Pubmed, Embase, Cochrane y Scielo utilizando los términos Dickkopf 1, DKK1, Dickkopf related protein 1, rheumatoid artrhitis, biomarcador, resorción ósea.
ResultadosSe escogieron 12 estudios que llenaban los requisitos de la búsqueda; 7 fueron cohortes prospectivas, 4 estudios de corte transversal y uno ensayo clínico. De los 12 estudios revisados, 10 analizaron la asociación entre niveles séricos de DKK1 y presencia de daño óseo como desenlace primario. Uno de ellos analizó esta asociación como desenlace secundario y otro la relación RSP01/DKK1. Los resultados hasta la fecha parecen indicar que la DKK1 tendría un papel activo en estadios avanzados de AR y no en la fase inicial.
ConclusionesLa proteína DKK1 desempeña un papel fisiopatológico esencial en la disminución de la masa ósea y la remodelación articular, dependiendo de la fase de la enfermedad, en pacientes con AR. Su papel como biomarcador o estrategia terapéutica sería una interesante alternativa aún en estudio.
Rheumatoid arthritis (RA) is a chronic, inflammatory disease, characterized by chronic synovitis, joint deterioration, and excessive bone loss.1 It affects between 0.5 and 2% of the world population, and is more common in women, with a peak of presentation between 50 and 60 years old.2,3 The aetiology of the disease is unknown; however, it is considered to be the result of the exposure to various environmental factors in genetically predisposed individuals.4 These genetic and environmental factors interact and trigger immune system alterations that lead to the production of antibodies such as the rheumatoid factor (RF) and anti-cyclic citrullinated peptides (anti-CCP), that finally result in the production of pro-inflammatory cytokines and the development of inflammatory arthritis.5 The loss of bone mass is a common characteristic in RA and it has been shown to be present since the early stages of the disease. In these patients, periarticular osteopenia is a significant characteristic. Furthermore, erosions at the pannus and adjacent bone tissue interphase may be identified, and these erosions progress rapidly and contribute to the deformities and morbidity that are typical of the disease. Since the severity and bone and joint involvement directly impact the short and long term prognosis in these patients, a large number of studies have tried to establish which factors identified early may predict negative outcomes. The major predictor of bone and joint deterioration in RA is the presence of erosions at the onset of the disease.6 However, our current knowledge, and the heterogeneity of the outcomes in patients with AR, make it mandatory to study new biomarkers to enable a more individualized treatment, addressed to therapeutic goals, which is currently referred to as T2T (treat to target) therapy. The recent evidence shows that DKK1 protein may play an active role in regulating bone biology.7,8
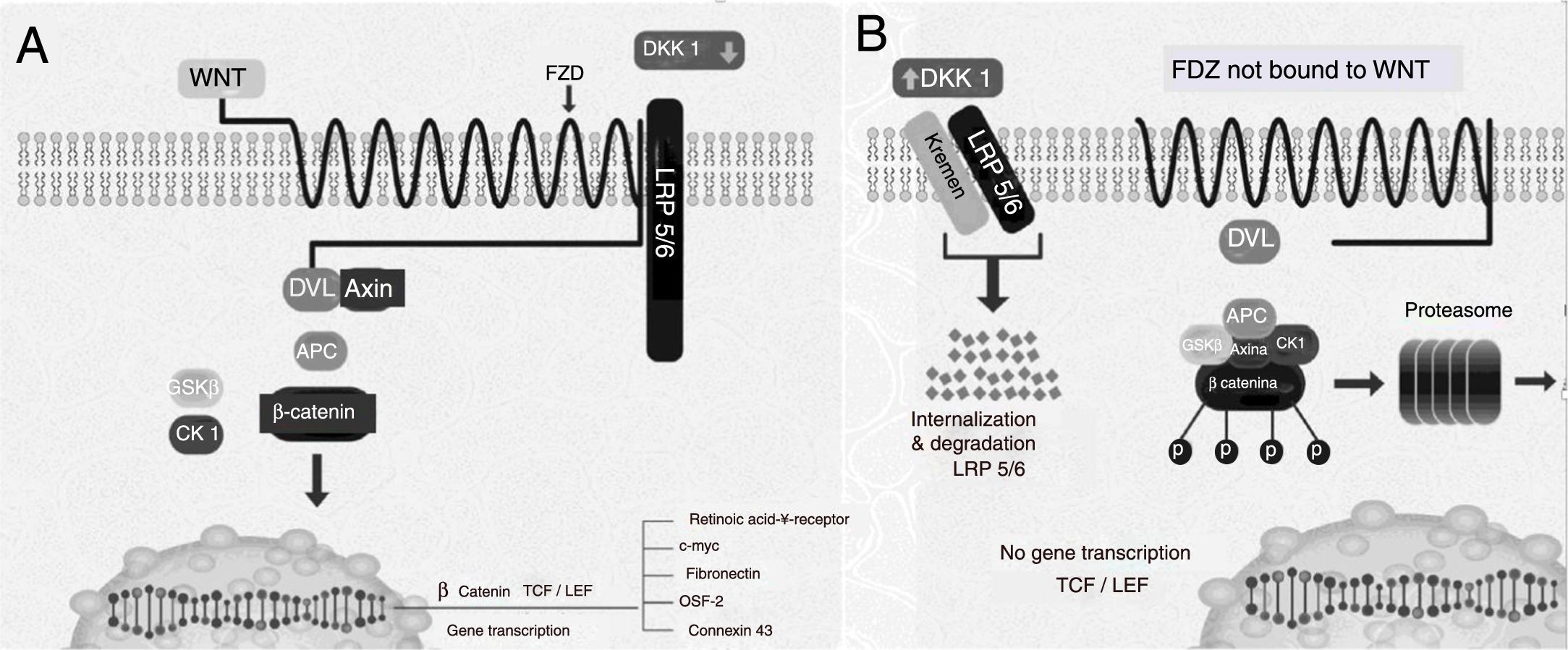
Dickkopf 1 negative regulation of the Wnt pathwayThe Wnt pathway activation induces different intracellular signals that are divided into two groups: the canonic or classical pathway and the non-canonic. The former is better known and presents an accumulation of β-catenin in the cytoplasm and subsequent translocation to the nucleus, where it modulates the transcription of various genes (Fig. 1A). Under baseline conditions, when it is not stimulated, a complex formed by several proteins such as axin, adenomatous polyposis coli (APC) protein, and glycogen synthase kinase-3β (GSK3β)<may be found.7 GSK3β phosphorylates β-catenin making to susceptible to binding to ubiquitin and to be subsequently degraded in the proteasome (Fig. 1B) In this manner, the intracellular levels of β-catenin are kept low. However, when the Wnt pathway is activated through the binding of the Wnt ligands to its receptor, the axin APC-GSK3β complex breaks down, reducing the phosphorylation activity of the GSK3β, and simultaneously reducing the phosphorylation of β-catenin and, consequently, its degradation in the proteasomes.9 Upon hypophosphorylation, it accumulates in the cytoplasm and translocates to the nucleus, where it regulates the gene expression through the activation of transcription factors such as TCF/LEF110 of molecules involved with the differentiation, proliferation, and maturation of osteoblasts, such as fibronectin, connexin 43, periostin, and the retinoic acid receptor γ.11 Wnt ligands act through the binding of receptors located in the cellular membrane (Fig. 1B). Several molecules with inhibiting action on the Wnt pathway have been described. The DKK1 inhibition of this pathway prevents the activation of the dishevelled protein (a protein associated with the Fzd receptor acting downstream), GSK3β continues to be activated and phosphorylates β-catenin, which then undergoes proteasomal degradation.7,12–14.
Components of the canonic pathway Wnt. (A) Gene activation and induction status. (B) At rest. APC: adenomatous polyposis coli protein; CK1: casein kinase 1; DKK1: Dickkopf1; Dvl: dishevelled; FZ: Frizzled receptor; GSK3β: glycogen synthase kinase 3β; LEF/TCF: lymphoid enhancer-binding factor/T-cell factors; LRP: LDL receptor-related protein; OSF-2: osteoblasts specific factor 2; WNT: Wingless.
Modified with authorization by Krishnan et al.13
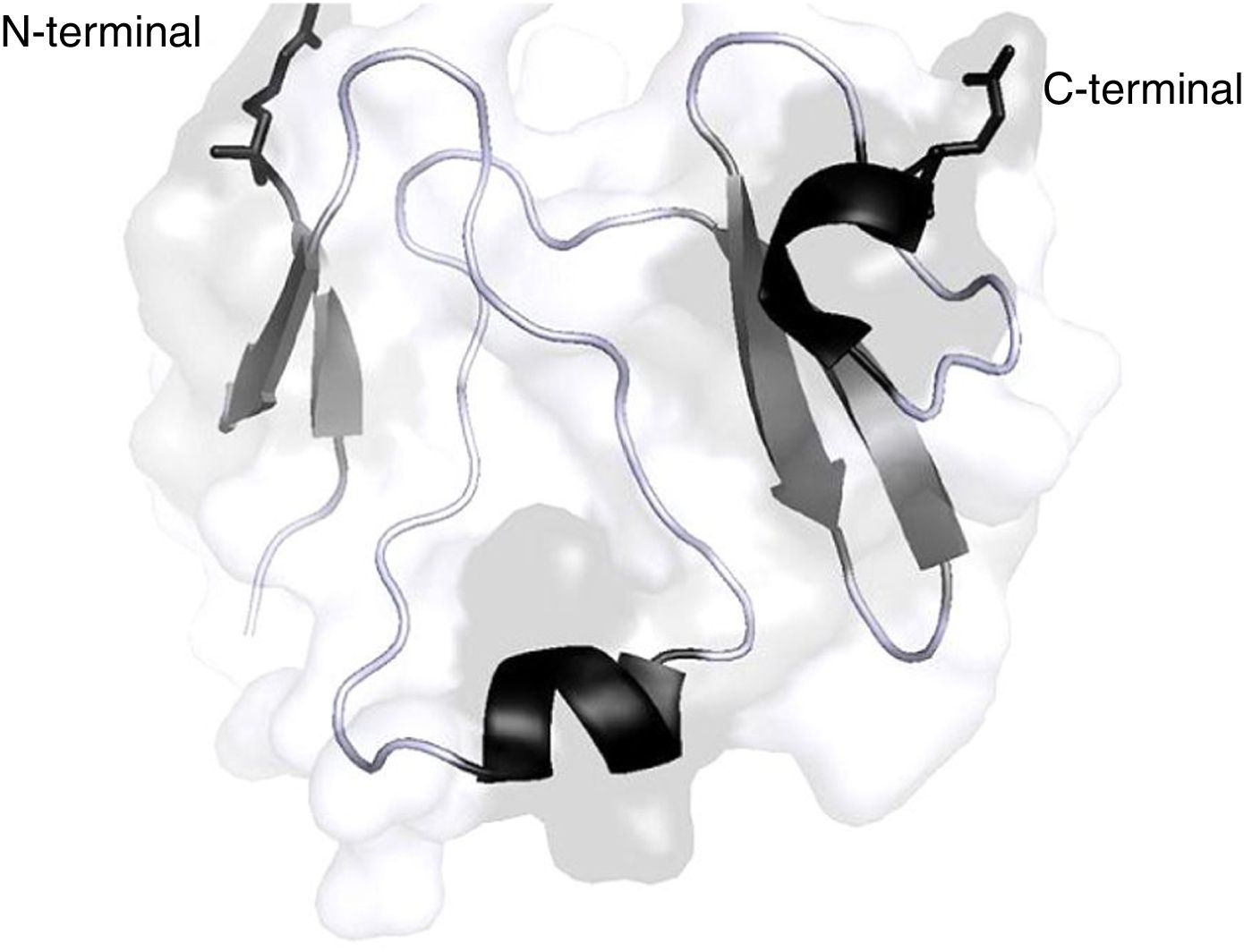
The 3 members of the DKK family (DKK1, DKK2, DKK4) are effective antagonists of the canonic Wnt-β catenin signalling pathway by binding directly and with significant affinity to LRP 5/6.15,16 The DKK molecule contains 2 conserved cysteine-rich domains (N or C, in accordance with their localization in the protein); these are connected through a binding domain17 (Fig. 2); the C domains of DKK1 and DKK2 may on their own inhibit the Wnt pathway.18,19 Studies on mutagenesis have revealed that one side of the C domain of DKK2 binds to LRP 5/6, while the other binds to Kremen,20 a molecule that modulates the antagonistic effect of DKK on the Wnt pathway. Moreover, the LRP 5/6 molecule may be divided into 3 regions: the ectodomain (ECD), the transmembrane domain, and the cytoplasmic domain.21 The ECD of LRP 5/6 also contains 4 β helical (P) domains comprising 4 amino acids (Tyr-Trp-Thr-Asp); each of the helical components couples to a domain similar to the epidermal growth factor (EGF) and then the ECD is made up by 3 type A receptor molecules of low density lipoprotein (LA).21 Several studies on mutagenesis indicate that the different Wnt molecules bind to different regions of the ECD of LRP 5/6.22,23 According to these studies, it can also be inferred that the P-EGF 3 and 4 domains that make up the ECD of LRP 5/6 are not needed for the Wnt1 pathway signalling, but are essential for DKK1 inhibition.24 The reason is that in the case of Wnt3a the binding surface to LRP 5/6 partially overlaps with the DKK1 binding site, so probably the antagonistic effect of DKK1 may be due to a mechanism of direct competition with the Wnt3a molecule for its binding site in LRP 5/6.25
The objective of this systematic review was to contribute with updated information on DKK1 protein and its association with osteoarticular deterioration in patients with RA.
Materials and methodsA systematic literature review of experimental studies, analytical observational studies (cohorts), and cross-sectional studies was conducted in order to assess the role of DKK1 protein and its association with osteoarticular outcomes (for example, bone erosions, joint damage, reduced bone mineral density [BMD]) in patients with early (≤2-years duration) or established RA. A search of primary studies, systematic reviews, and meta-analyses was conducted in the most important scientific databases, using the MeSH terms for each of the components of the PICO question (Problem, Intervention, Comparator, Outcomes).
Types of studiesControlled clinical trials, published and un-published cohort and cross-sectional studies written in English or Spanish, evaluating the association of the DKK1 levels with osteoarticular deterioration outcomes in patients with early or established RA were included. In all cases, the number of patients studied was more than 50. The analytical studies included all of the trials that considered exposure, target population and bias control strategies.
Type of participantsPatients older than 18 years with a diagnosis of early or established RA.
Type of interventionDetermination of the serum DKK1 levels.
Type of outcomesAssociation of the serum DKK1 levels with bone erosions, decreased BMD or joint damage.
Electronic searchA database search was conducted using the above-mentioned PICO strategy. The Medline search was conducted with a combination of key words and filters recommended by Pubmed. The search was limited to articles published in English and Spanish.
Search termsA structured literature search was conducted using the electronic databases Pubmed, Embase, Cochrane and Scielo, with the key words ((«Arthritis, Rheumatoid»[Mesh]) AND «DKK1 protein, human» [Supplementary Concept]) AND «Bone Resorption»[Mesh] AND «Biomarker»).
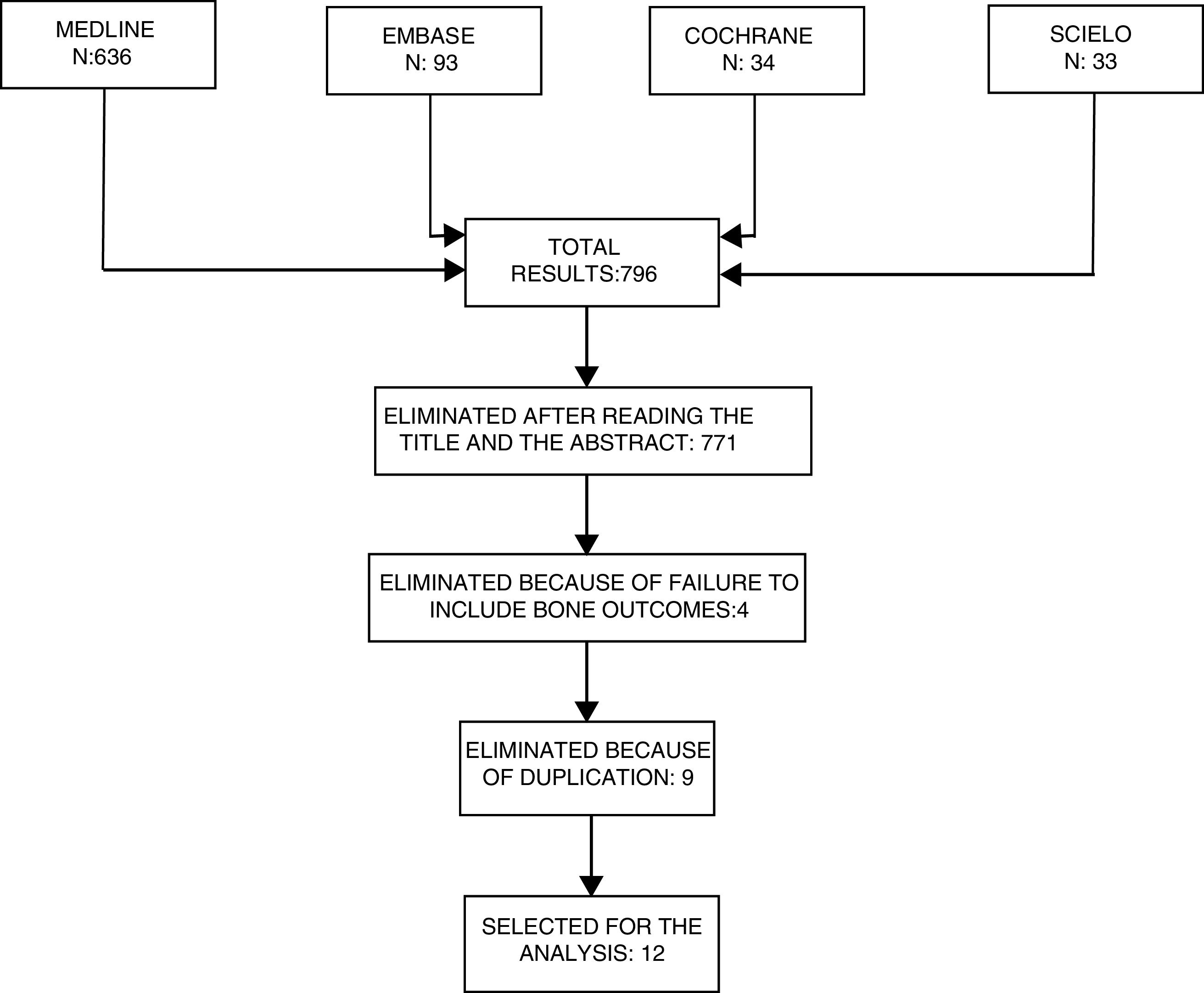
Selection of studiesThe abstracts of the articles identified were reviewed to eliminate any irrelevant articles. Subsequently, any articles chosen were independently reviewed by the authors ACR and JBG, to check for compliance with the inclusion criteria (Fig. 3).
Data miningThe studies that met the inclusion criteria were analyzed for data mining. The data were independently extracted by 2 of the authors (ACR and JBG) and the results were then re-assessed by the other authors (JCM, RVO and CRS) for consistency of the data. The data extracted from each study were summarized in 2 tables, for early and established RA, respectively, including the number of patients, age, sex, radiological score used, type of osteoarticular outcome, frequency measurements and association and statistical significance.
A meta-analysis was not performed because of the heterogeneity of the studies.
Missing dataSome studies failed to report the DKK1 levels and others failed to report the frequency and association. In the case of the latter, the statistical significance was used to compare against the direct results of the study.
In the same group, a prospective cohort study was performed in 136 patients with established RA, in whom the baseline HGF and DKK1 levels were measured, in addition to a baseline densitometry study, which was repeated then at 1, 2 and 5 years. In this study, the baseline DKK1 levels were correlated with increased periarticular bone loss at one year, and this result was maintained after doing a multivariate analysis.36
In the study by Choi et al. in 2014, they found that patients with RA and radiographic progression of the disease had a significant increase in the DKK1 serum levels and in the DKK1/R-Spondin (DKK1 antagonist). In the multivariate analysis, the DKK1 serum levels alone were not associated with radiological progression, but the DKK1/R-Spondin index was.37
Finally, in 2015 Rossini et al. conducted a cross-sectional trial in 154 postmenopausal women with established RA, matched by age, sex, body mass index (BMI), and vitamin D levels. The DKK1 levels were measured and the radiological evaluation was conducted, in addition to measuring the spinal and hip BMD. The results indicated that in the patients with RA, the age-adjusted DKK1 levels, the PTH levels and the duration of the disease were significantly higher in patients with bone erosions.38
ResultsWith the search terms used, 839 titles were identified in the various data bases, and 814 were excluded after applying the eligibility criteria to the respective titles and abstracts. Then any duplicate studies were excluded and additionally, 3 studies that failed to include osteoarticular outcomes were also eliminated. Out of the 12 studies selected, 11 were written in English and one in Spanish; 7 were prospective cohorts, 4 were cross-sectional studies, and one was a clinical trial. Of the studies reviewed, 10 analyzed the association between DKK1 and the presence of bone damage as the primary outcome. One of them analyzed this association as a secondary outcome and yet another one, the RSP01/DKK1 ratio.
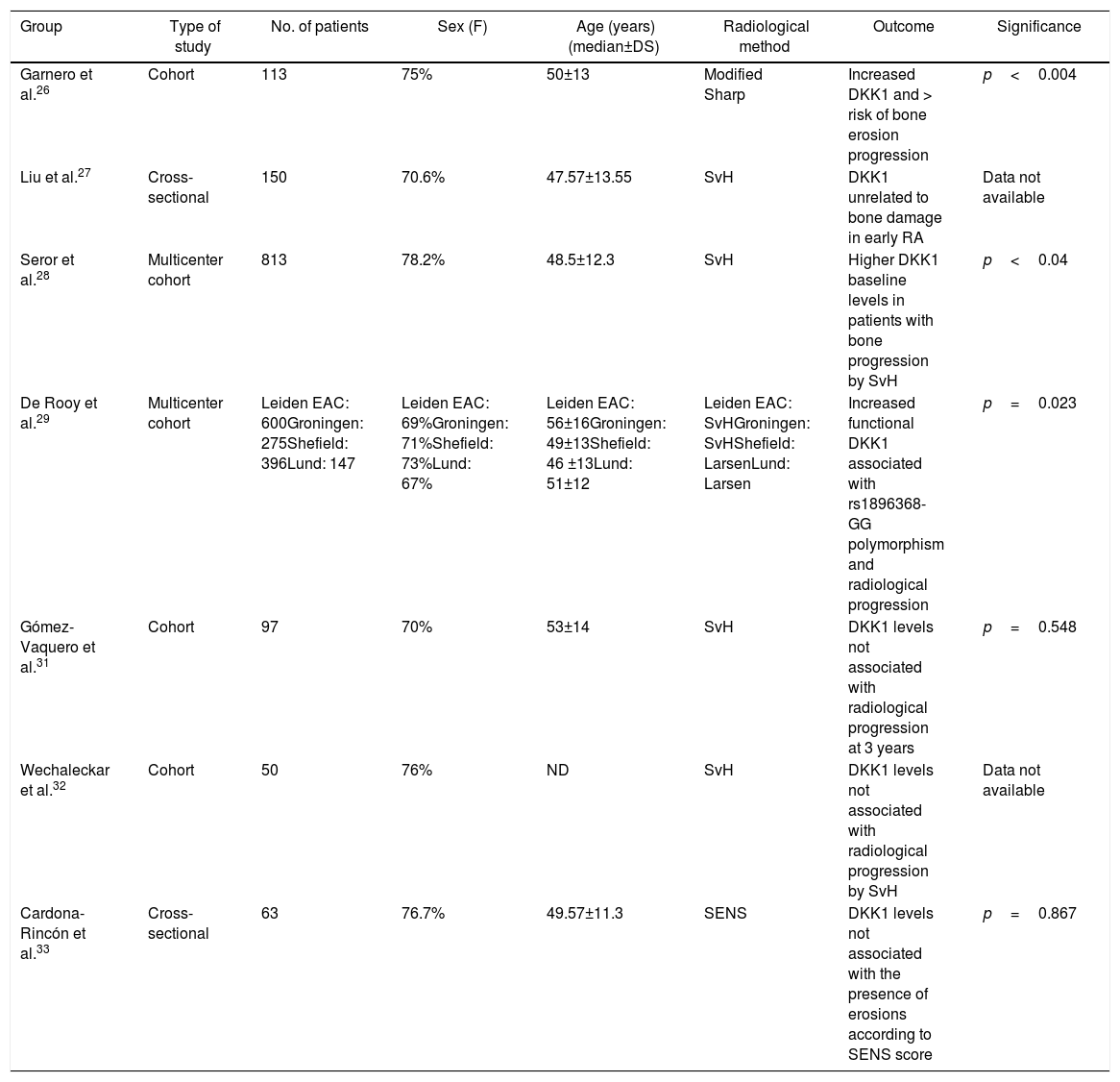
Relationship between the Dickkopf 1 levels and osteoarticular damage in early rheumatoid arthritisSeven studies analyzed the relationship between the DKK1 levels and the presence of bone or joint damage progression in early RA, 5 of which were cohorts and 2 were cross-sectional26–32 (Table 1).
Studies on the association between serum DKK1 levels and bone damage in early RA.
| Group | Type of study | No. of patients | Sex (F) | Age (years)(median±DS) | Radiological method | Outcome | Significance |
|---|---|---|---|---|---|---|---|
| Garnero et al.26 | Cohort | 113 | 75% | 50±13 | Modified Sharp | Increased DKK1 and > risk of bone erosion progression | p<0.004 |
| Liu et al.27 | Cross-sectional | 150 | 70.6% | 47.57±13.55 | SvH | DKK1 unrelated to bone damage in early RA | Data not available |
| Seror et al.28 | Multicenter cohort | 813 | 78.2% | 48.5±12.3 | SvH | Higher DKK1 baseline levels in patients with bone progression by SvH | p<0.04 |
| De Rooy et al.29 | Multicenter cohort | Leiden EAC: 600Groningen: 275Shefield: 396Lund: 147 | Leiden EAC: 69%Groningen: 71%Shefield: 73%Lund: 67% | Leiden EAC: 56±16Groningen: 49±13Shefield: 46 ±13Lund: 51±12 | Leiden EAC: SvHGroningen: SvHShefield: LarsenLund: Larsen | Increased functional DKK1 associated with rs1896368-GG polymorphism and radiological progression | p=0.023 |
| Gómez-Vaquero et al.31 | Cohort | 97 | 70% | 53±14 | SvH | DKK1 levels not associated with radiological progression at 3 years | p=0.548 |
| Wechaleckar et al.32 | Cohort | 50 | 76% | ND | SvH | DKK1 levels not associated with radiological progression by SvH | Data not available |
| Cardona-Rincón et al.33 | Cross-sectional | 63 | 76.7% | 49.57±11.3 | SENS | DKK1 levels not associated with the presence of erosions according to SENS score | p=0.867 |
RA: rheumatoid arthritis; DKK1: Dickkopf 1; F: sex female; NA: not available; SENS: Simple Erosion Narrowing Score; SvH: Sharp-van der Heijde score.
In 2008, Garnero et al. did a prospective study with 113 patients with early RA, who were undergoing treatment with etanercept or methotrexate as monotherapy. The baseline DKK1 levels were measured and serological and radiological follow-up was done with the Sharp-van der Heijde (SvH) score at 1 year. They found that the elevated initial levels of DKK1 were associated with a higher risk of progression of bone erosion and these changes were unrelated to age, sex, baseline radiological damage, CRP levels or the level of disease activity.26
Subsequently, Liu et al. analyzed the DKK1 levels and osteoprotegerin in 150 patients with early RA and in 150 patients with established RA; they found that the elevated levels of DKK1 were associated with increasing bone damage based on the SvH score and clinical activity based on the DAS28 index in the group of patients with established RA.27
More recently, Seror et al. analyzed the cohort of French patients with early RA known as ESPOIR. In this prospective cohort, the serum DKK1 baseline levels, the acute phase reactants, a clinimetric measurement of the disease activity and a baseline and after 2-years evaluation of the bone damage based on SvH levels were conducted. Of the total number of patients with RA in this cohort, 110 had bone erosions at the start of follow-up, and this finding was significantly correlated with several factors such as anti-CCP, age, female gender, elevated CRP, ESR and DKK1 levels. In the multivariate analysis, the DKK1 levels were independently associated with signs of radiological progression of damage by SvH.28
In 2013, de Rooy et al. conducted a single nucleotide polymorphisms (SNP) study of different components of the Wnt pathway in 4 European cohorts (LeidenNED, Groningen-NED, Sheffield-UK and Lund-SWE), in which 8 SNP of DKK1, 44 of LRP-5, 16 of Kremen-1 and 9 of sclerostin (SOST) were analyzed, and regression models were performed to establish the association between the polymorphisms of these molecules and their serum levels, as well as the radiological progression using 2 different scores (SvH and Larsen). The results showed that in the Leiden cohort, 6 SNP of DKK1, 3 of sclerostin, one of Kremen and 10 of LRP-5 were significantly associated with joint damage progression. According to the meta-analysis conducted, 3 SNP of DKK1 were significantly associated with joint damage progression and 2 SNP of scletostin had a tendency towards statistical significance. One SNP of DKK1 was associated with higher blood DKK1 levels (rs1896368, p=0.02).29
A study by Miceli-Richard et al. tried to emulate the findings by de Rooy et al. in the French cohort ESPOIR; however, none of the 10 SNPs of DKK1 studied was associated with progression of structural damage according to the SvH score.30
Further studies have been unable to find an association between the levels of DKK1 and radiographic alterations in patients with early RA.31–33
Gómez-Vaquero et al. conducted a cohort study in 97 patients with early RA (median 1.6 years of evolution). Following an average follow-up of 3.3 years, the average SvH score progression was 0.88±2.2 units. The multivariate analysis showed that certain factors such as age (OR per year=1.10; p=0.003) an d elevated CRP (OR=1.29; p=0.005), but not the DKK1 levels, were associated with radiological progression according to SvH.31
Similarly, Wechaleckar et al., in 2016, conducted an initial cohort study in 50 patients with RA of <1 year of evolution, with double positivity for RF and anti-CCP. No association was found between the baseline levels of DKK1 and the radiological progression according to SvH, after one year of follow-up.32
Finally, in 2017 Cardona-Rincón et al. submitted the results of a cross-sectional trial in 63 patients with early RA. Once again, no significant statistical correlation was found between the levels of DKK1 and variables such as the disease activity according to DAS28 or radiological damage based on the SENS score.33
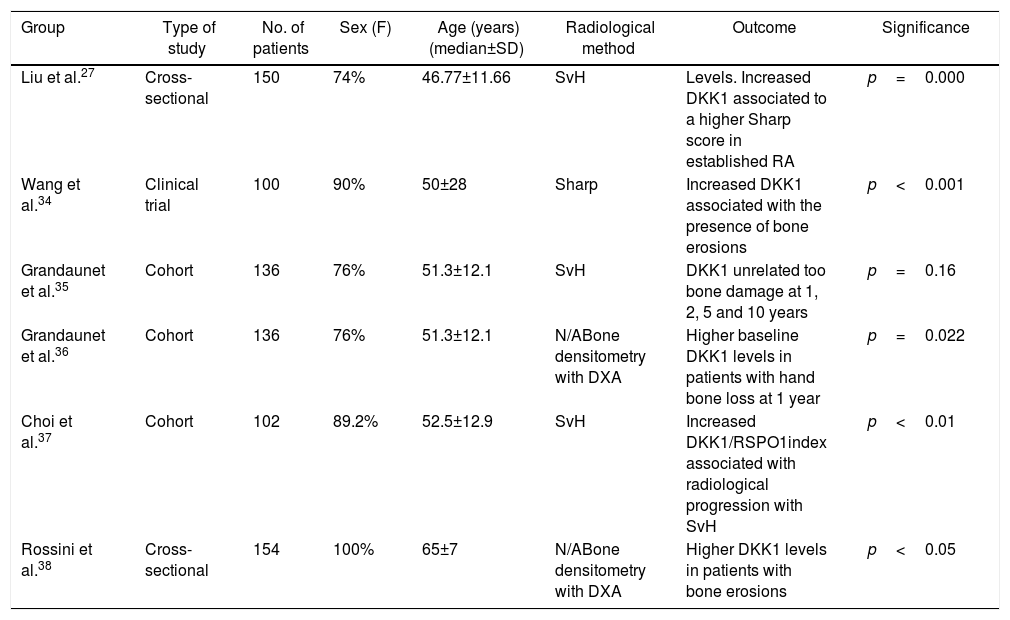
Relationship between the Dickkopf 1 levels and osteoarticular damage in established rheumatoid arthritisOther studies aimed at establishing a correlation between the elevated levels of DKK1 and osteoarticular deterioration have been conducted in patients with established RA (Table 2).
Studies on the association between serum DKK1 and bone damage in established RA.
| Group | Type of study | No. of patients | Sex (F) | Age (years)(median±SD) | Radiological method | Outcome | Significance |
|---|---|---|---|---|---|---|---|
| Liu et al.27 | Cross-sectional | 150 | 74% | 46.77±11.66 | SvH | Levels. Increased DKK1 associated to a higher Sharp score in established RA | p=0.000 |
| Wang et al.34 | Clinical trial | 100 | 90% | 50±28 | Sharp | Increased DKK1 associated with the presence of bone erosions | p<0.001 |
| Grandaunet et al.35 | Cohort | 136 | 76% | 51.3±12.1 | SvH | DKK1 unrelated too bone damage at 1, 2, 5 and 10 years | p=0.16 |
| Grandaunet et al.36 | Cohort | 136 | 76% | 51.3±12.1 | N/ABone densitometry with DXA | Higher baseline DKK1 levels in patients with hand bone loss at 1 year | p=0.022 |
| Choi et al.37 | Cohort | 102 | 89.2% | 52.5±12.9 | SvH | Increased DKK1/RSPO1index associated with radiological progression with SvH | p<0.01 |
| Rossini et al.38 | Cross-sectional | 154 | 100% | 65±7 | N/ABone densitometry with DXA | Higher DKK1 levels in patients with bone erosions | p<0.05 |
RA: rheumatoid arthritis; DKK1: Dickkopf 1; DXA: dual X-ray absorptiometry; F: female gender; N/A: not applicable; RSPO1: R-spondin 1; SvH: Sharp-van der Heijde score.
Three cohorts, 2 cross-sectional studies, and a clinical trial studied the association between DKK1 and bone outcomes in established RA.
In 2011, a group of researchers led by Doctors Wang and Liu analyzed the levels of DKK1 in serum of 100 patients with established RA and 100 patients with other rheumatologic conditions such as osteoarthritis and anchylosing spondylitis, and found that the levels of this protein were significantly elevated in the RA group. Moreover, the levels of DKK1 correlated with the levels of CRP, ESR, and radiological changes according to the SvH score. This study also observed that patients with RA that were treated with anti TNF-α (infliximab) or anti-IL-1 (anakinra) presented decreased serum DKK1levels.34
Also in 2011, Grandaunet et al. conducted a prospective cohort study in 136 patients with established RA, with an average duration of the disease of 2.2 years. DKK1 serum levels and hepatocyte growth factor (HGF) were measured, and a radiological evaluation was conducted with the SvH score. This study found that elevated HGF levels but not the DKK1 levels were associated with osteoarticular damage.35
DiscussionRA is an autoimmune, inflammatory, chronic, and progressive disease, mainly characterized by damage to the small joints of the hands and feet and a generalized reduction in BMD. From the onset of the disease, patients may experience bone erosions; it is well known that the presence of bone erosions is the most important predictor for the occurrence of future erosions and bone deterioration,38–40 and is also associated with decreased BMD.41 Furthermore, the presence of certain factors such as elevated acute phase reactants, the number of swollen joints, the presence of RF or anti-CCP, female gender, the prevalence of upper rather than lower extremities involvement, age, and altered BMI, has shown to be associated with progression of radiological damage in patients with RA.42 Not all patients with RA have the same pattern of bone damage progression; hence the vital importance of being able to differentiate which patients will experience worse outcomes, in order to optimize resources and improve the clinical results.29
The knowledge of the components of the Wnt osteogenic (canonic) pathway and its negative regulators has improved over the past years. The role of DKK1 protein, as a molecule that plays different roles, has given rise to a lot of interest among the scientific community and has led to an increasing number of studies published about this molecule as a serological marker associated with different outcomes in different diseases, both rheumatologic such as ankylosing spondylitis,42 systemic lupus erythematous43,44 or osteoarthritis,45 and non-rheumatologic such as hepatocellular carcinoma,46 atherosclerotic disease and diabetes,47 inter alia. DKK1 studies have been made in RA, both in the synovial fluid48,49 and in the bone50; these studies have established the overexpression of this protein in the sites most affected by the pathology, which is irrefutable proof of the time and space relationship between DKK1 and the clinical or radiological findings in patients with RA. There is a very close relationship between elevated TNF-α and DKK1 levels in patients with RA, and it has been shown that cytokines such as TNF-α and IL-1β may indirectly stimulate the production of DKK1 through the fibroblast-like synoviocytes.7,48 Likewise, the treatment with anti-TNF-α antibodies in these patients has been associated with a decrease in the serum levels of DKK1,34,51,52 in the same way as with anti-IL-6 antibodies.53 This shows that the DKK1 levels may play an important role, not just in predicting bone outcomes, but also in the evaluation of the response to anti-TNF-α or anti-IL-6; however, proper studies are needed to be able to answer this question.
The results of the different studies analyzed in this systematic review show that DKK1 protein may be an additional tool for the clinician, in order to identify those patients that require more aggressive therapy. Nonetheless, there is little evidence in patients with early RA because of the short time of evolution (Table 1), keeping in mind the importance of forecasting outcomes from the onset of the disease. It must be said also that while the DKK1 levels are associated with bone erosions or mineral reduction, there was no significant association in these studies with the reduction in the joint space, which is consistent with the current knowledge about the involvement of the Wnt pathway rather than its negative regulators (DKK1, sclerostin) in joint remodelling of patients with RA.7
This review tried to divide the current RA DKK1 into two groups: patients with early RA and patients with established RA. However, since currently no consensus has been reached on the cut off point between the early and the late presentation of the disease, it was decided to classify the studies in accordance with the opinion of the authors thereof (for instance, in some studies, early RA was established as a disease duration of <2 years, while in others it was <1 year or <6 months). Another drawback of the serum DKK1 studies is that most of them measure the circulating DKK1 instead of the functional DKK1 (DKK1 bound to LRP 5/6). According to some authors, functional DKK1 correlates better with the bone resorption status in contrast to circulating DKK.54 Moreover, these studies have shown that most of the patients who overexpress DKK1 experience bone damage progression; however, it is yet unknown why some patients with normal or low DKK1 levels exhibit progression of radiological damage. It should also be mentioned that whilst some of the trials conducted so far have shown an association of DKK1 with progression of radiological damage, other studies such as those by Grandaunet et al.,35 Gómez-Vaquero et al.31 and Liu et al.,27 were not able to establish this association in the early RA subgroup. A likely explanation for this discrepancy could be the broad heterogeneity of the population studied, keeping in mind differences in the laboratory parameters of the DKK1levels, dissimilar populations studied, the duration of the disease, the concomitant biological or glucocorticoid therapy, inter alia. Additionally, the multiple and potential causes of pre-analysis alterations, such as diurnal variations, the comorbidities of the patient, and the technical conditions under which the measurements were taken, should be taken into consideration.
ConclusionsSeveral studies have been conducted, both in early and established RA patients, that have associated elevated serum DKK1 levels with adverse outcomes, including increased radiological progression of bone damage or decreased bone mass according to densitometry. Protein DKK1 plays a pathophysiological role in bone mass reduction and joint remodelling in patients with RA. Its role as a biomarker or therapeutic strategy in this population has not yet been established. The results to date seem to indicate that DKK1 could play an active role as a biomarker in advanced RA stages, but not in the initial phase.
FinancingThis review is supported by two ongoing projects: Hospital Militar Central (Code: 2015-047), Colombian Association of Rheumatology 2016 call for papers and Universidad El Bosque (Code: PCI 2016-8806).
Conflict of interestThe authors do not have any conflict of interests to disclose.
The authors acknowledge Dr. Krishnan Venkatesh for his kind contribution to the request for use authorization and amendments to Fig. 1.
Mrs. Brigitte Espinosa, librarian of the Medical Library, Deputy Directorate of Education and Scientific Research, Hospital Militar Central, for her advice.
Dr. Adriana Beltrán, Director of the Research Division, Deputy Directorate of Education and Scientific Research, Hospital Militar Central, for her advice in the design of the systematic review.
Please cite this article as: Cardona-Rincón AD, Bello-Gualtero JM, Munevar-Niño JC, Romero-Sánchez C, Valle-Oñate RR. Proteína Dickkopf 1 y deterioro osteoarticular en artritis reumatoide: revisión sistemática. Rev Colomb Reumatol. 2019;26:48–57.