Cirrhosis has gradually become a serious public health issue, especially the national prevalence of cirrhosis was 29.2% in northwest China. Recent evidence has revealed that intestinal barrier (IB) dysfunction results from and contributes to cirrhosis. Our previous results have indicated that insulin-like growth factors (IGF-1) improved the impaired IB function and downregulated high mobility group protein box-1 (HMGB-1). Nevertheless, the role of the IGF-1/HMGB1 axis in cirrhosis remains largely unknown.
Materials and methodsWestern blotting and qRT-PCR were used to detect protein and mRNA levels of related genes. The levels of AST, ALT, IL-1β, and TNF-α were examined using commercial kits. Immunofluorescence was used to evaluate the expression of HMGB1 in tissues.
ResultsIn carbon tetrachloride (CCl4)-treated rat, the levels of AST (380.12 vs. 183.97), ALT (148.12 vs. 53.56), IL-1β (155.94 vs. 55.60), and TNF-α (155.00 vs. 48.90) were significantly increased compared with the control group, while IGF-1 treatment significantly alleviated CCL4-induced inflammatory response and IB dysfunction by downregulating HMGB1-mediated the TLR4/MyD88/NF-κB signaling pathway. In vitro experiments, HMGB1 treatment promoted inflammatory cytokines secretion and reduced cell viability and tight junctions by activating the TLR4/MyD88/NF-κB signaling pathway in Caco-2 cells, but IGF-1 alleviated these effects.
ConclusionOur findings suggest that IGF-1 might serve as a potential therapeutic target for cirrhosis and IB dysfunction via inactivation of the TLR4/MyD88/NF-κB pathway through down-regulation HMGB1.
Cirrhosis has gradually become a leading cause of morbidity and mortality which is the 14th most common cause of death all over the world and the 4th in central Europe [1]. It leads to 1.03 million deaths per year in the world and 170,000 deaths per year in Europe [2]. Clinical practice indicates that cirrhosis is considered the end stage of various chronic liver diseases, often leading to death without liver transplantation and that the only preventive strategies are screening for esophageal varices and hepatocellular carcinoma. cirrhosis is characterized by portal hypertension and hepatic synthetic dysfunction [3]. Accumulating evidence has revealed IB dysfunction is strongly associated with the development of cirrhosis [4,5]. The intestinal barrier (IB) is necessary to maintain the integrity of the intestinal tract and the stability of the internal environment, and it is also responsible for the surveillance and elimination of bacteria, viruses, and antigen molecules [6]. IB dysfunction predisposes to portal hypertension and is a risk factor for complications of spontaneous bacterial peritonitis (SBP), which leads to decompensated cirrhosis [7]. Moreover, IB dysfunction can result in the formation of polymorphic nuclei and M cells, and induce the release of pro-inflammatory cytokines [8,9]. Meanwhile, the high levels of pro-inflammatory cytokines could further aggravate IB injury and liver dysfunction in patients with cirrhosis [10]. However, there is a lack of effective therapeutic drugs and novel targets for cirrhosis and IB dysfunction in clinical.
High-mobility-group box 1 (HMGB1) is normally located in the nucleus, and it acts as a DNA chaperon regulating transcription, replication, recombination, repair, and genome stability [3]. In response to certain stimuli (e.g., hypoxia), HMGB is released into the extracellular environment and is involved in the regulation of local infection, inflammation, and injury. Moreover, HMGB1 is a therapeutic target for inflammation-related disease via regulating the toll-like receptor 4 (TLR4)/NF-kappaB (NF-κB) signaling pathway [11,12]. Meanwhile, previous studies have shown that HMGB1 is not only an important cytokine that induces IB dysfunction in acute pancreatitis, but can also function as an induction factor for inflammatory bowel disease. Importantly, HMGB1 levels are significantly elevated in the serum of patients with chronic hepatitis and cirrhosis [13,14]. However, it is unclear whether HMGB1 is involved in the damage of the IB in cirrhosis.
Insulin-like growth factor 1 (IGF-1) has been reported to be found in muscle, bone, intestine, and liver [15,16]. IGF-1 can not only promote the growth and nutrition of various tissues, but also regulates IB function [17]. Similarly, our previous studies have demonstrated that IGF-1 treatment reduced HMGB1 levels and exhibited a protective effect on IB by increasing intestinal tight junctions in the cirrhosis model [18]. Moreover, IGF-1 exerts an anti-inflammatory effect on skeletal muscle cells by blocking the TLR4 pathway [19]. Nevertheless, the protective effect of the IGF-1/HMGB1/TLR4 signaling pathway in cirrhosis and IB dysfunction remains unclear.
The research aimed to investigate the regulatory role of the IGF-1/HMGB1/TLR4/NF-κB pathway in cirrhosis and IB dysfunction. In this study, we uncovered that IGF-1 administration significantly ameliorated carbon tetrachloride (CCL4)-induced cirrhosis and IB damage. Mechanistically, we verified whether IGF-1 exhibited a protective effect on cirrhosis induced by CCL4 via regulating the HMGB1/TLR4/NF-κB pathway. Furthermore, we established the HMGB1-treated Caco-2 cell model to examine the functional role of the IGF-1/HMGB1/TLR4/NF-κB pathway in regulating inflammation and tight junctions in vitro.
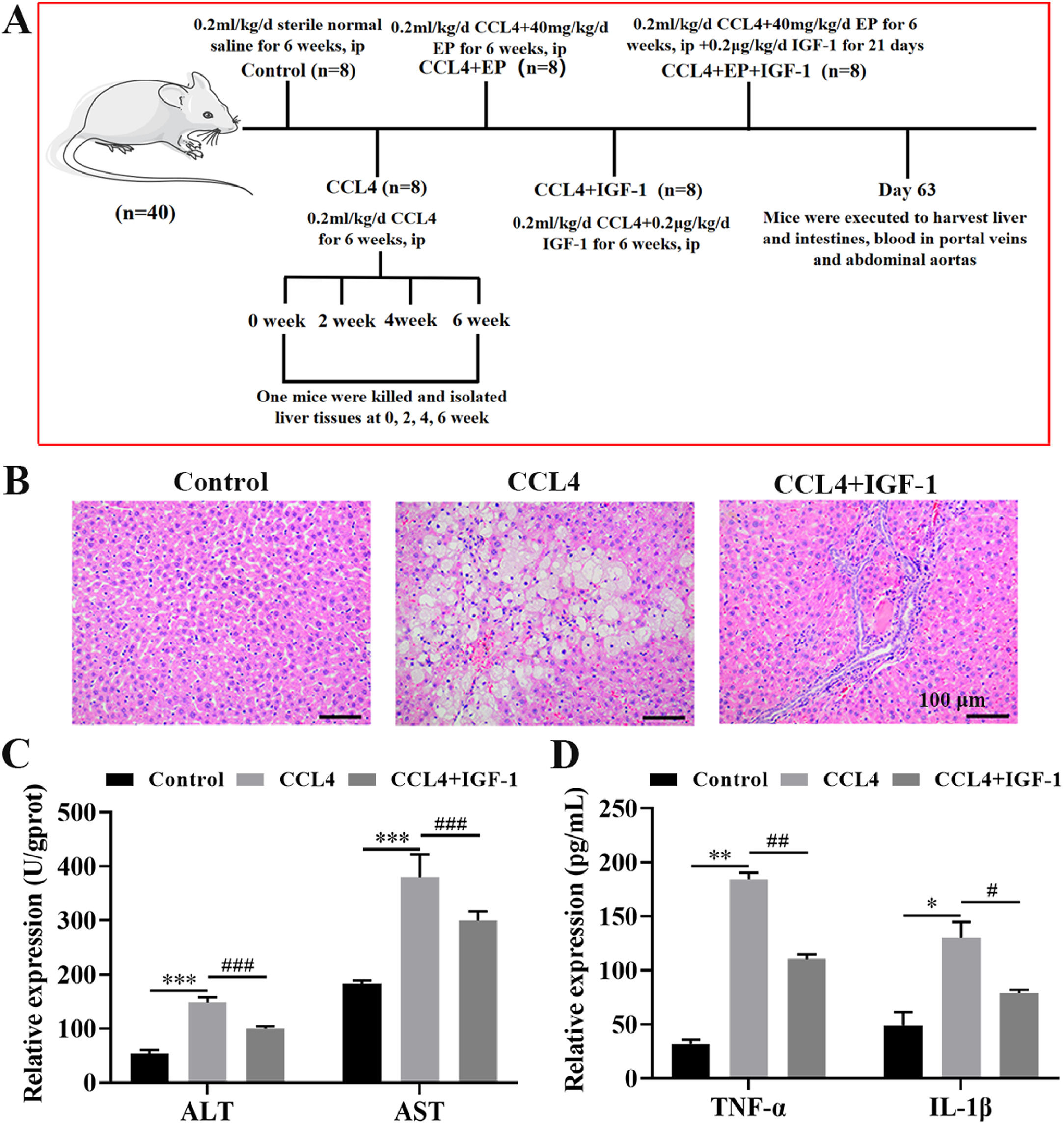
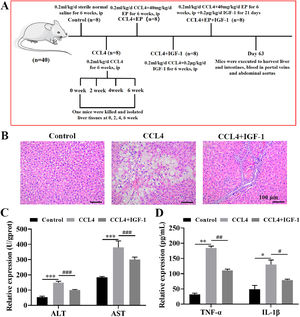
2Materials and methods2.1Animal experimentsThe protocols of all animal experiments were approved by the Experimental Animal Ethical Committee of Dalian Medical University. The animal model of CCL4-induced cirrhosis was established as previously described [18,20,21]. In brief, 40 male rats (6-week-old) were randomly divided into five groups (8 rats in each group) as follows: control group (intraperitoneal injection (i.p) of 0.2 mL/kg/d sterile normal saline for 6 weeks), cirrhosis model group (i.p of 0.2 mL/kg/d CCL4 for 6 weeks), cirrhosis model+ethyl pyruvate (EP) treatment group (i.p 0.2 mL/kg/d CCL4 and 40 mg/kg/d EP for 6 weeks), cirrhosis model+IGF-1 treatment group (i.p of 0.2 mL/kg/d CCL4 for 6 weeks followed by subcutaneously injection of 0.2 μg/kg/d recombinant human IGF-1 for 21 days), and cirrhosis model+EP+IGF-1 treatment group (i.p of 0.2 mL/kg/d CCL4 and 40 mg/kg/d EP for 6 weeks, followed by subcutaneously injection of 0.2 μg/kg/d recombinant human IGF-1 for 21 days). A detailed description of the experimental protocol is presented in Fig. 1A.
IGF-1 attenuates CCL4-induced cirrhosis in the rat. (A) The schematic presents the animal experiment schedules in this study. The rat was executed after receiving the desired treatment, and then liver, cecum tissues, and portal venous blood were collected. Histomorphology changes of the liver (B) were evaluated by H&E staining and photographed under a light microscope. Magnification: 200 ×, scale bars = 100 μm. (C) The levels of AST and ALT portal venous blood were examined using AST activity assay kit and ALT activity assay kit, respectively. (D) Serum levels of TNF-α and IL-1β were examined using commercial ELISA kits. *P<0.05, ⁎⁎P<0.01, ⁎⁎⁎P<0.001, vs. control group, #P<0.05, ##P<0.01, ###P<0.001, vs. CCL4 group.
Total RNA in tissues and Caco-2 cells was isolated and quantified, as previously described [18]. The relative expression levels of genes were normalized to β-actin levels and were calculated using the comparative CT (2–ΔΔCT) method. The primer sequences used are listed in Table 1.
The primer sequences for qRT-PCR
The frozen liver sections were incubated with HMGB1 antibody (1:100, A2553, ABclonal, China) at 4 °C overnight. Then, the slides were washed with PBS (5 min) three times and incubated with Cy3-labeled goat anti-rabbit IgG (1:200, A0516, Beyotime, China) for 60 min in a dark room. Then, the sections were washed using PBS three times and counterstained with DAPI (C1002, Beyotime, China). After washing with PBS, images of the stained specimens were captured under a microscope (Olympus BX53, Tokyo, Japan) using an image analysis system (Olympus DP73, Tokyo, Japan) after adding fluorescent mounting media (S2100, Solarbio, China).
2.4Measurements of alanine transaminase (ALT) and aspartate aminotransferase (AST) contentAll rats were anesthetized at day 63 using isoflurane and portal venous blood was collected. Serum contents of AST and ALT were assessed using corresponding AST and ALT activity kits (Wanleibio, China), following the manufacturer's instructions. The OD values were determined at 562 nm using a microplate reader (ELX-800, BIOTEK, USA) and relative activity was calculated using a standard curve.
2.5ELISA analysis of interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α) levelsBlood was collected from the portal veins of the rat at the time points indicated. Then the content of IL-1β and TNF-α in the serum were determined using commercial ELISA kits (Wanleibio, China) by following the manufacturer's protocol.
2.6Cell treatmentTo screen for optimal concentration of HMGB1 on Caco-2 cells (the Chinese Academy of Medical Sciences, China), the Caco-2 cells were assigned into the following four groups: (1) control group: cells were routinely cultured for 24 h; (2) 0.1 μg/mL HMGB1 group: cells were cultured with Dulbecco's modified Eagle medium (DMEM) medium (Gibco, USA) containing 0.1 μg/mL HMGB1 for 24 h; (3) 1 μg/mL HMGB1 group: cells were cultured with DMEM medium containing 1 μg/mL HMGB1 for 24 h; (4) 10 μg/mL HMGB1 group: cells were cultured with DMEM medium containing 10 μg/mL HMGB1 for 24 h.
To investigate the effect of IGF-1 on the HMGB1-mediated protective effect on Caco-2 cells, cells were divided into the following four groups: (1) control group: cells were cultured routinely for 24 h; (2) 10 μg/mL HMGB1 group: cells were cultured with DMEM medium containing 10 μg/mL HMGB1 for 24 h; (3) IGF-1 group: cells were cultured with DMEM medium containing 100 ng/mL IGF-1 for 24 h; (4) HMGB1+IGF-1 group: cells were cultured with DMEM medium containing 10 μg/mL HMGB1and 100 ng/mL IGF-1 for 24 h.
2.7Cell counting kit-8 (CCK-8) assayCaco-2 cells in the logarithmic growth phase were plated in 96-well plates at a density of 5,000 cells per well. The cell viability in each group of the above-mentioned treatment groups was calculated using the CCK-8 kit (Solarbio, China). In brief, 10 μL of CCK-8 solution was added into each well and incubated at 37°C for 2 h following the manufacturer's instructions. Then, the plates were read using a microplate auto-reader (ELX-800, BIOTEK, USA) at a wavelength of 570 nm. Each experiment was performed in triplicate.
2.8Hematoxylin-eosin (H&E) stainingTo examine the histological changes in the liver and cecum tissues of the different groups, disassociated tissues were fixed using 4 % formaldehyde overnight. Then, the tissues were embedded in paraffin and sectioned at a thickness of 4 μm using rotary paraffin slicing microtome (RM2235, Leica, Germany). Changes in the structure were examined using standard protocols of H&E staining. After staining, the slides were observed under a microscope (Olympus BX53, Tokyo, Japan) and images were captured using an image analysis system (Olympus DP73, Tokyo, Japan).
2.9Western blotting analysisProtein expression levels in the tissues and cells were examined as previously described [18]. The primary antibodies for TLR4 (1:1,000), MyD88 (1:500), NF-κB p65 (1:500), Claudin-1 (1:500), and β-actin (1:2,000) were all purchased from the Cell Signaling Technology (USA). β-actin was chosen as the internal reference to be used to calculate the relative protein expression levels of the target genes.
2.10Statistical analysisAll data are presented as the mean ± standard deviation. Differences between the two groups were evaluated using Student's t-test (unpaired, two-tailed) or nonparametric tests. Differences between three or multiple groups were calculated by one-way ANOVA following the LSD-t test. Statistical analyses were performed using GraphPad Prism (version 6.01) software (La Jolla, USA) and SPSS 20.0 software (IBM, USA). A p-value of less than 0.05 was considered to be statistically significant.
3Results3.1IGF-1 treatment alleviated cirrhosis induced by CCL4 in vivoH&E staining results demonstrated that rats in the modal group developed cirrhosis, as evidenced by the disordered lobular structure, ballooning, inflammatory cell infiltration, fibrotic septa, and pseudo-lobule formation in the CCL4 group (Fig. 1B). Importantly, IGF-1 administration significantly improved liver injury induced by CCL4 (Fig. 1B). Moreover, we also examined changes in liver function by detecting the level of ALT and AST in portal venous blood. As expected, the levels of AST and ALT were enhanced (approximately 2.8 times and 2 times, respectively) in the CCL4 alone group compared with the control group (Fig. 1C), while IGF-1 administration effectively attenuated these effects. Given the important role that inflammation plays in the initiation and progression of cirrhosis, we detected the levels of two important pro-inflammatory cytokines, including TNF-α and IL-1β. As anticipated, the levels of TNF-α and IL-1β in the CCL4 alone group were elevated compared with the control group (Fig. 1D). Meanwhile, treatment with IGF-1 significantly rescued the expression of inflammatory cytokines induced by CCL4. Collectively, these results suggested that IGF-1 may exert a protective effect against CCL4-induced liver dysfunction in vivo.
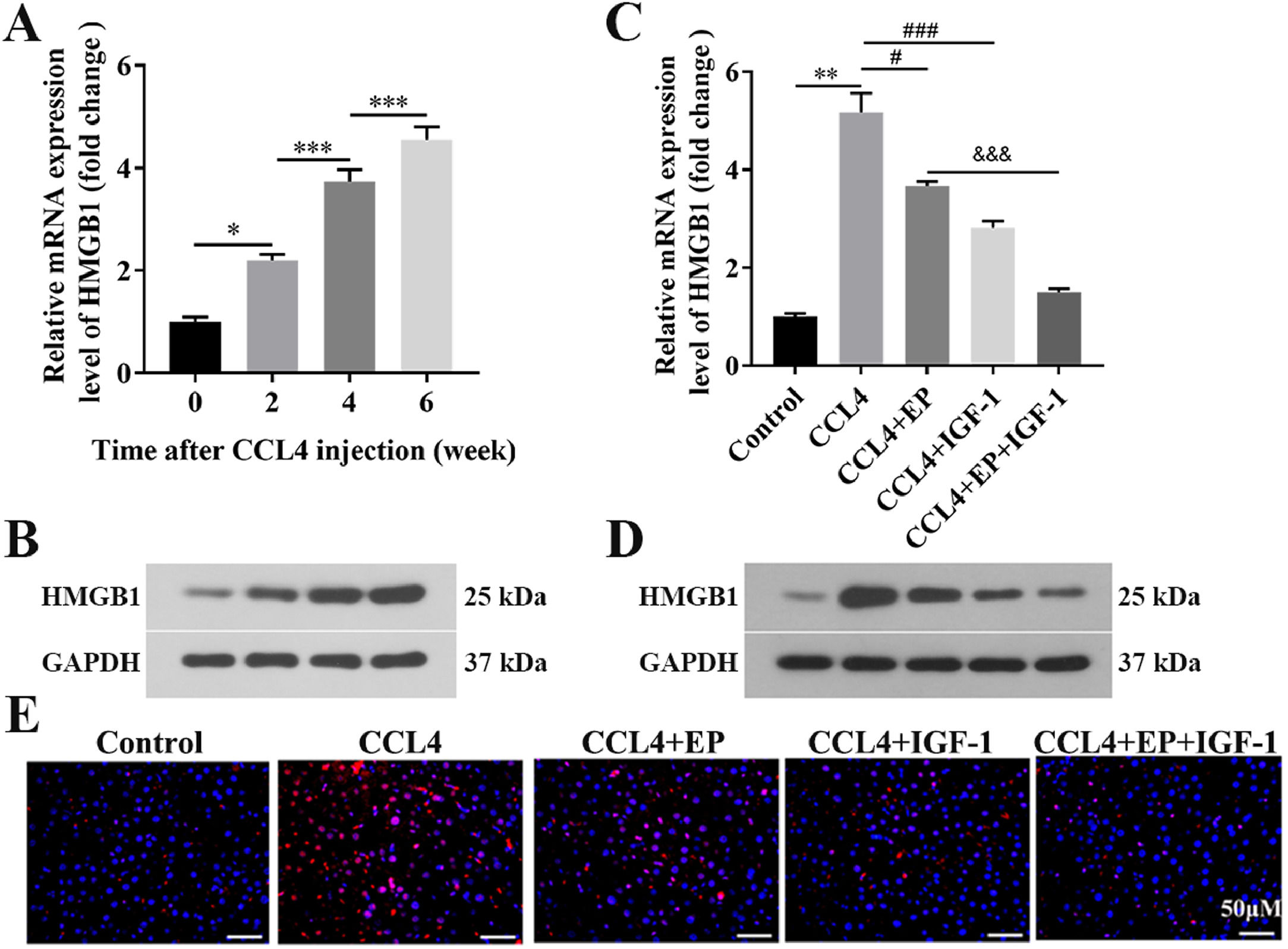
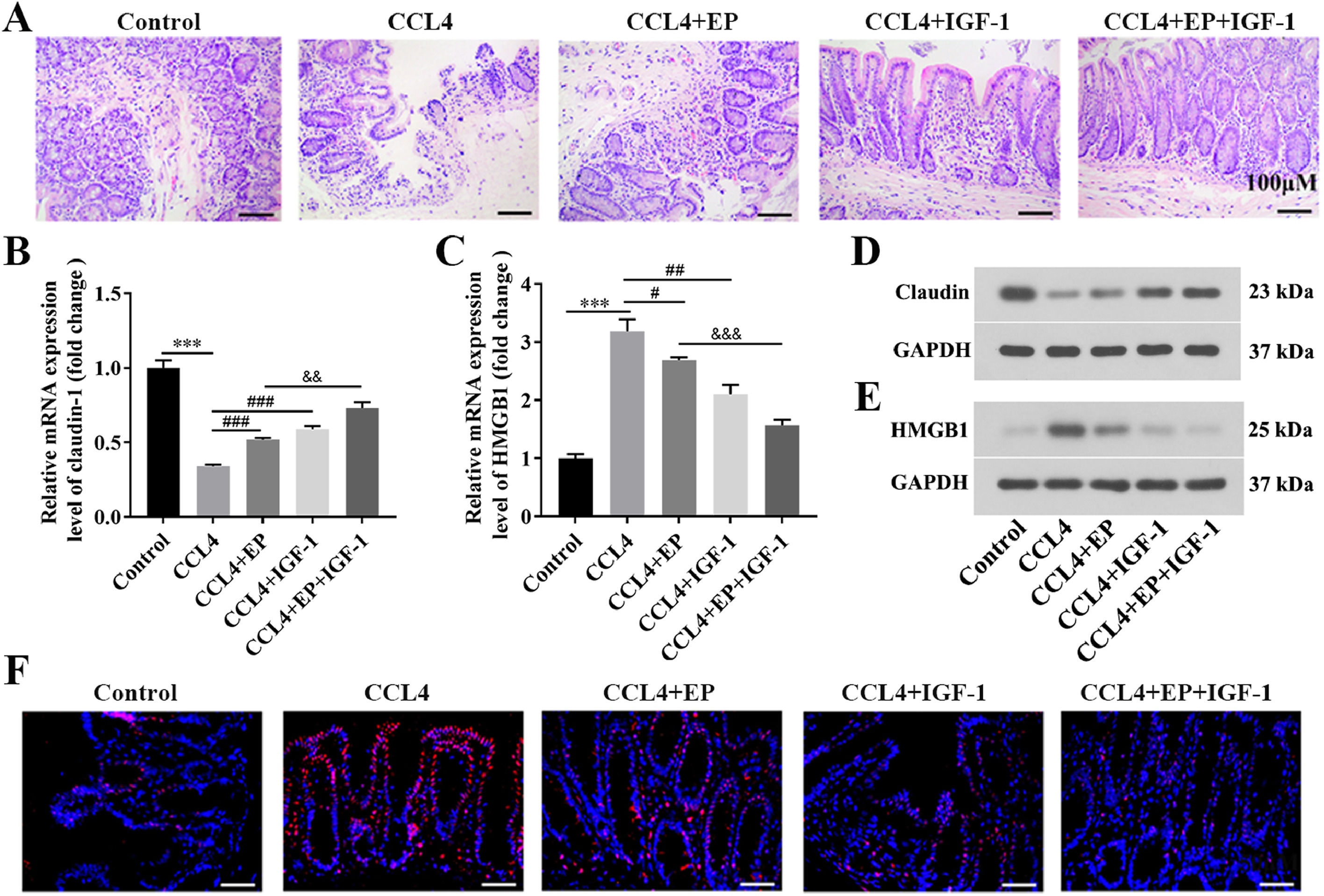
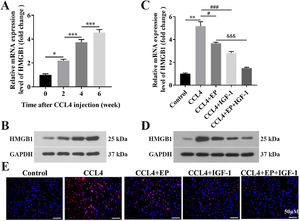
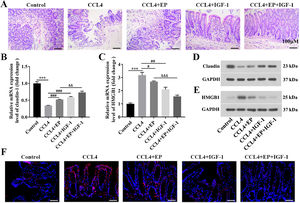
3.2HMGB1 was elevated in both liver and cecum tissues of rats in the cirrhosis modelWe further explored whether HMGB1 was involved in the protective role of IGF-1 in CCL4-induced cirrhosis. The results showed that CCL4 promoted both mRNA (Fig. 2A) and protein (Fig. 2B) levels of HMGB1 in liver tissues of rats in a time-dependent manner. However, IGF-1 administration or EP (a specific inhibitor of HMGB1) treatment reduced both mRNA (Fig. 2C) and protein (Fig. 2D) levels of HMGB1 in liver tissues of CCL4-treated rats, as well as IGF-1 plus EP treatment significantly decreased the expression of HMGB1 compared with the CCL4+IGF-1 group and CCL4+EP group. Moreover, IF analysis results showed that the immunopositively profiles of HMGB1 in the CCL4 alone group were increased compared with the control group (Fig. 2E), but both EP and IGF-1 treatment notably reduce HMGB1 abundance compared with the CCL4+IGF-1 group and CCL4+EP group. H&E staining showed that the infiltration of numerous inflammatory cells, edema, and desquamation of the villus, and the disappearance of goblet cells and crypt structures were observed in the CCL4 alone group, suggesting the IB structure and function were damaged (Fig. 3A). Of note, both IGF-1 and EP treatment significantly improved these pathological changes and restore IB function in CCL4-treated rat (Fig. 3A). Moreover, we assessed IB function by detecting the level of claudin-1, an important tight junction protein. Consistent with the H&E results, both mRNA (Fig. 3B) and protein (Fig. 3D) levels of claudin-1 were significantly downregulated in the CCL4 alone group compared with the control group, while IGF-1 and EP could reverse the inhibitory effect of CCL4 on the expression of claudin-1 in cecum tissues of rats. Meanwhile, we also examined both mRNA and protein levels of HMGB1 in cecum tissues of rats. The results showed that HMGB1 was significantly up-regulated in the CCL4 alone group compared with the control group (Fig. 3C, 3E), while both EP and IGF-1 administration notably reduced HMGB1 expression in cecum tissues of CCL4-treated rat. Furthermore, the IF analysis of HMGB1 expression in cecum tissues was similar to the above results (Fig. 3F). Collectively, these results indicated that HMGB1 was involved in CCL4-induced cirrhosis and impaired IB function.
IGF-1 significantly reduced the level of HMGB1 in liver tissues. Live tissues were collected from the rat in the model group (CCL4 alone) at four-time points: before CCL4 injection, 0, 2, 4, and 6 weeks. Then qRT-PCR (A) and western blotting (B) were employed to detect the mRNA and protein levels of HMGB1 at different time points. The qRT-PCR (C) and western blotting (D) were used to detect the mRNA and protein level of HMGB1 in the liver tissues. (E) Immunofluorescence was applied to examine the levels of HMGB1 in liver tissues.
IGF-1 significantly reduced the level of HMGB1 in cecum tissues. All rats were executed on day 63 following IGF-1 injection and cecum tissues were collected. (A) H&E staining was used to observe the histological change of cecum tissues. Scale bars = 100 μm. The mRNA and protein levels of claudin-1 (B, D) and HMGB1 (C, E) in cecum tissues were examined using qRT-PCR and western blotting, respectively. (F) Immunofluorescence analysis of HMGB1 distribution in cecum tissues. Magnification: 400 ×, scale bars = 50 μm. ⁎⁎⁎P<0.001, vs. control group, #P<0.05, ###P<0.001, vs. CCL4 group, &&P<0.01, &&&P<0.001, vs. CCL4+EP group.
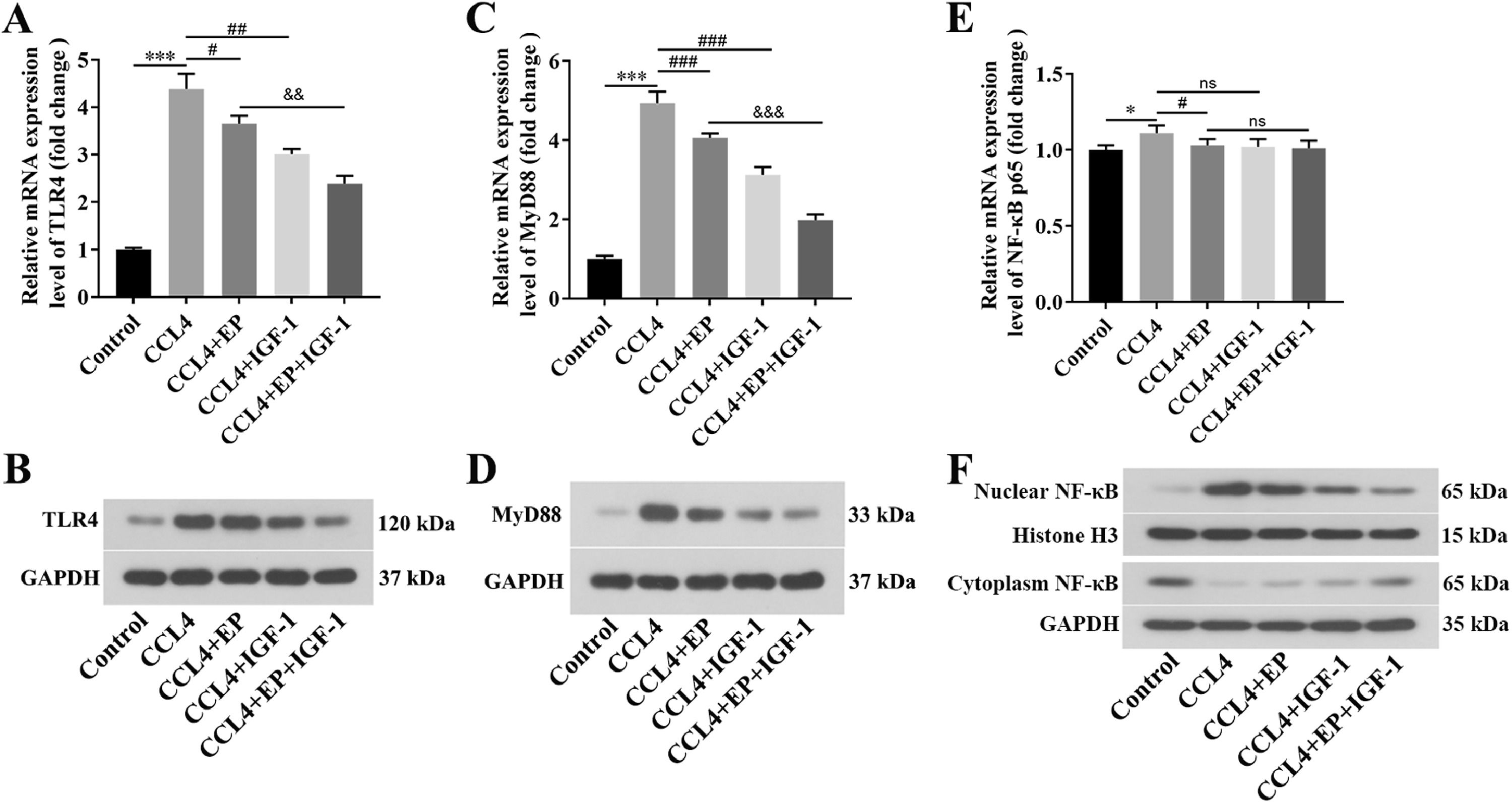
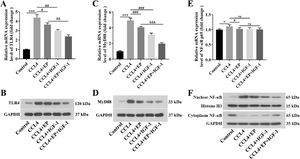
We further explored whether HMGB1 contributed to cirrhosis and impaired IB function by activating the TLR4/MyD88/NF-κB signaling pathway. As expected, both mRNA and protein levels of TLR4 (Fig. 4A, 4B) and MyD88 (Fig. 4C, 4D) in the CCL4 alone group were significantly elevated compared with the control group. In contrast, treatment with EP and IGF-1 alone or in combination could effectively downregulate the promotion effect of CCL4 on the TLR4/MyD88 signaling pathway (Fig. 4A-4D). Moreover, the NF-κB signaling pathway was notably activated in the CCL4 alone group (Fig. 4E, 4F), as evidenced by the increased nuclear translocation of NF-κB expression. However, treatment with EP and IGF-1 alone or in combination could notably reverse the activation of the NF-κB pathway induced by CCL4 (Fig. 4E, 4F). Collectively, these results suggest that IGF-1 exerted a protective role in CCL4-induced cirrhosis by downregulation of HMGB-1 and blocking the TLR4/MyD88/NF-κB signaling pathway.
IGF-1 inactivation of the TLR4/MyD88/NF-κB signaling pathway mediated by HMGB1 in cirrhosis rats. qRT-PCR was employed to examine the mRNA levels of TLR4 (A), MyD88 (C), and NF-κB (E) in each group. The protein levels of TLR4 (B), MyD88 (D), and NF-κB (F) were detected using western blotting. *P<0.05, ⁎⁎⁎P<0.001, vs. control group, #P<0.05, ##P<0.01, ###P<0.001, vs. CCL4 group, &&P<0.01, &&&P<0.001, vs. CCL4+EP group.
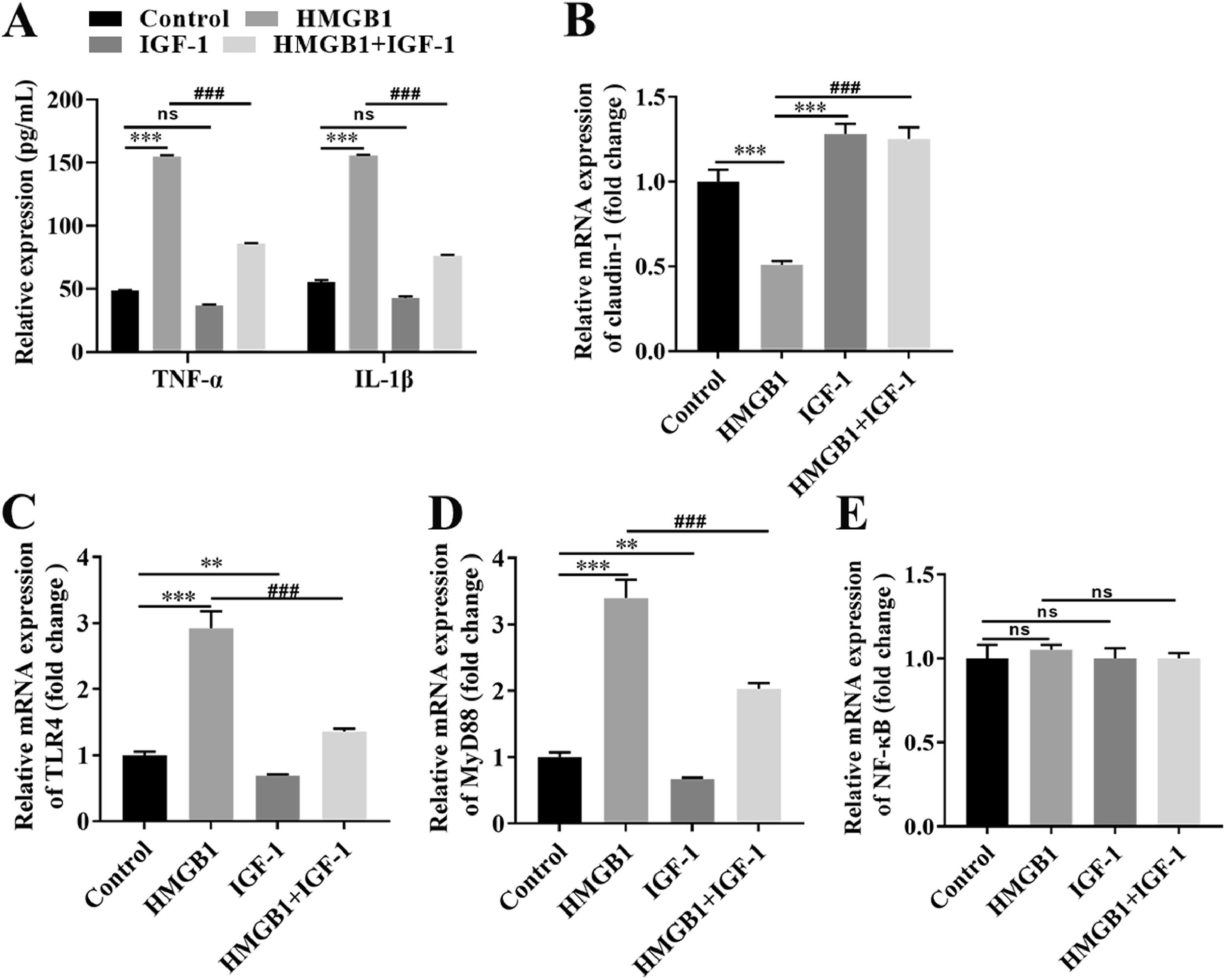
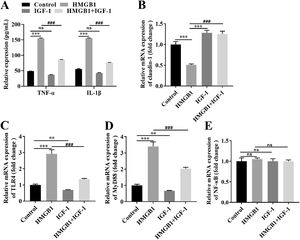
Based on our in vivo findings, we further investigated the functional role of the IGF-1/HMGB1 axis in Caco-2 cells. First, we conducted broader-ranged dose-response studies to screen the toxicity of HMGB-1 in Caco-2 cells. As shown in Fig. S1A, treatment with HMGB1 at the concentration of 0.1 μg/mL, 1 μg/mL, and 10 μg/mL significantly reduced cell viability in a dose-dependent manner. Besides, HMGB1 alone treatment could activate the TLR4/MyD88/NF-κB signaling pathway in Caco-2 cells (Fig. S1B-1G). Meanwhile, our results also showed that 1 μg/mL HMGB1 treatment significantly increased the expression levels of IL-1β and TNF-α compared with the control group (Fig. 5A), while combined with HMGB1 and 100 ng/mL IGF-1 treatment partially decreased the levels of IL-1β and TNF-α compared with the HMGB1 alone group (Fig. 5A). Next, we also found that HMGB1 treatment alone dramatically reduced the expression of claudin-1 at both mRNA (Fig. 5B) and protein (Fig. S1A) levels compared with the control group, but HMGB1 plus IGF-1 treatment notably enhanced claudin-1 expression. Furthermore, HMGB1 alone treatment resulted in activation of the TLR4/MyD88/NF-κB signaling pathway, as evidence by the increased levels of TLR4 (Fig. 5C; Fig. S1B), MyD88 (Fig. 5D; Fig. S1C), and the nuclear translocation of NF-κB (Fig. 5E; Fig. S1D). However, IGF-1 plus HMGB1 treatment partially decreased the expression of TLR4, MyD88, and NF-κB. Taken together, the aforementioned results suggest IGF-1 inhibited IB damages induced by HMGB1 by blocking the TLR4/MyD88/NF-κB signaling pathway in vitro.
IGF-1 blocked the TLR4/MyD88/NF-κB signaling pathway in Caco-2 cells via reducing HMGB-1. Caco-2 cells were treated with HMGB-1, IGF-1, or HMGB-1 combination with IGF-1 for the indicated time. (A) The levels of IL-1β and TNF-α were examined using commercial ELISA kits. The mRNA expression of claudin-1 (B), TLR4 (C), MyD88 (D), and NF-κB (E) in Caco-2 cells were examined using qRT-PCR. ⁎⁎P<0.01, ⁎⁎⁎P<0.001, vs. control group, ##P<0.01, ###P<0.001, vs. HMGB1 group.
Despite a better understanding of cirrhosis and IB dysfunction, there is still a lack of effective clinical treatments. In this study, we found that IGF-1 could attenuate CCL4-induced cirrhosis through the down-regulation of HMGB-1 in vivo. Moreover, downregulation of HMGB1 by EP or IGF-1 treatment significantly improved IB dysfunction. In vitro experiments, our data showed that HMGB1 treatment contributed to inflammation and reduced cell viability and tight junctions via activating the TLR4/MyD88/NF-κB pathway in Caco-2 cells, but addition IGF-1 alleviated these effects. Functionally, IGF-1 suppressed CCL4-induced cirrhosis and improved IB function by reducing HMGB-1 levels and inactivation the TLR4/MyD88/NF-κB pathway.
Recently, increasing evidence has demonstrated that IGF-1 is involved in cirrhosis, for example, Miyauchi and colleagues found that there is a negative correlation between serum IGF-1 levels and liver fibrosis markers expression in type 2 diabetes mellitus patients [22]. Another study found that upregulation of IGF-1 could improve cell longevity and alleviate liver fibrosis by blocking hepatocyte premature senescence induced by oxidative stress [23]. Of note, the reduction of IGF-1 was associated with the serological parameters of liver damage, and IGF-1 overexpression was reduced with liver fibrosis progression [24]. In the present study, IGF-1 treatment effectively improved inflammation and IB dysfunction induced by CCL4 in the cirrhosis rat model. Similarly, Aksu et al. found that the serum and liver levels of IGF-1 were decreased in diabetic rats, and the reduction of blood IGF-1 levels correlates with hepatic injury [25], suggesting circulating IGF-1 may have predictive value for determining hepatic injury and acts as an effective therapeutic target for liver damage related diseases. Moreover, a study revealed that IGF-1 treatment alleviates oxidatively modified low-density lipoprotein induced inflammation by reducing HMGB1 [26]. Furthermore, treatment with IGF-1 notably improved the intestinal mucosal barrier of rats with necrotizing enterocolitis by reducing inflammatory response [27].
Previous studies have demonstrated that HMGB1 plays an important role in the pathogenesis of liver injury, fibrosis, and liver cancer [13]. For instance, a recent study revealed that HMGB1 could effectively accelerate the progression of chronic liver injury to hepatocarcinogenesis, while HMGB1 deficiency attenuated these changes [14]. Numerous pre-clinical and clinical studies have indicated that the concentrations of circulating HMGB1 in liver failure animals and patients are notably elevated and positively associated with gut bacterial translocation, as well as multiple organ failure [28]. Therefore, HMGB1 inhibition may represent a potential therapeutic strategy for the prevention and treatment of fibrosis. In the present study, our data showed that downregulation of HMGB1 attenuated CCL4-induced cirrhosis and IB dysfunction by inactivation of the TLR4/MyD88/NF-κB signaling pathway in vivo. Ge et al. showed that HMGB1 was essential for the pathogenesis of liver injury and liver fibrosis [29]. Meanwhile, Chen and colleagues demonstrated that HMGB1 downregulation attenuated intestinal mucosal barrier dysfunction in mice with acute pancreatitis [30]. Yang et al. showed that anti-HMGB1 neutralizing antibody ameliorated gut barrier dysfunction via reducing bacterial translocation to mesenteric lymph nodes [31]. Moreover, previous studies have found that HMGB1 promoted inflammation to accelerate disease progression by activation of the TLR4/NF-κB pathway, such as HMGB1 promoted lipopolysaccharide-induced acute lung injury via activating the TLR4/NF-κB and PI3K/Akt/mTOR pathways [32]. Shang et al. found that silencing of HMGB1 reduced the levels of inflammatory cytokines via blocking the TLR4/MyD88/NF-κB pathway in asthma [33]. In line with previous studies, our results showed that HMGB1 treatment significantly promoted inflammatory response and reduced cell viability and tight junctions by activating the TLR4/MyD88/NF-κB pathway in Caco-2 cells. Moreover, Sappington and colleagues found that HMGB1 overexpression impaired IB function in mice and increased the permeability of Caco-2 cell enterocytic monolayers [34]. Another study showed that inhibition of HMGB1 alleviated the progression of acute necrotizing pancreatitis via enhancing tight junction and stabilizing intestinal permeability [35].
However, there are some limitations in this study. For instance, this study did not validate the influence of HMGB-1 levels (knockdown or overexpression) on cirrhosis, IB function, or the activation of the downstream signaling pathway. Furthermore, activation of the TLR4/MyD88/NF-κB pathway should be inhibited using an inhibitor of the gene knockdown method to verify its mediation role.
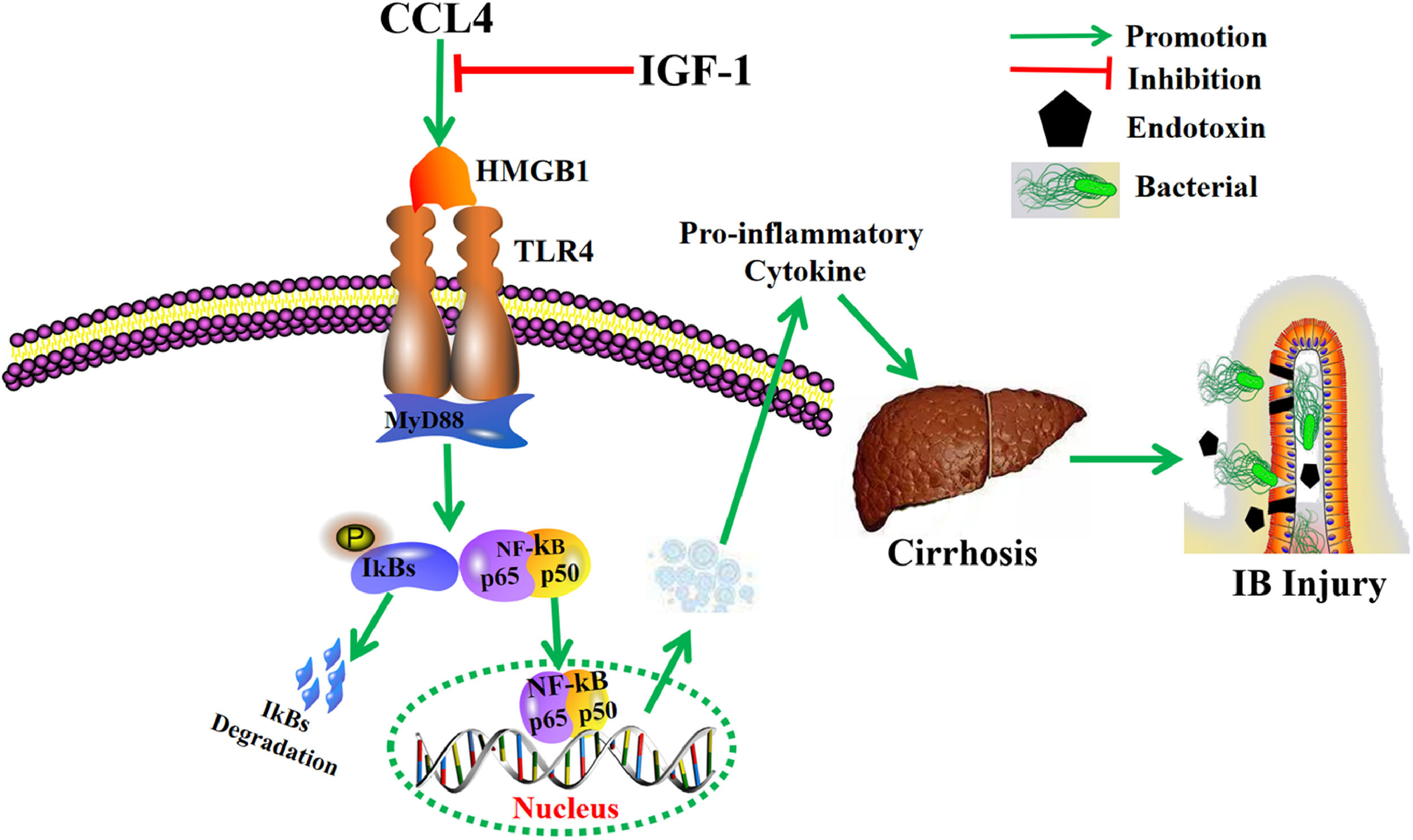
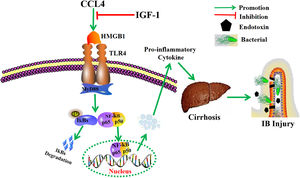
5ConclusionThis study further elucidates the potential role of IGF-1 for the prevention and treatment of cirrhosis and improvement of IB function (Fig. 6). These findings suggest that IGF-1 may be a potential target drug for the treatment of cirrhosis and the prevention of IB injury.
Authors’ contributionsTianyu Zhao and Ying Zhu made substantial contributions to conception and design. Liying Yao, Liu Liu and Na Li made acquisition and analysis of data. Tianyu Zhao and Ying Zhu have been involved in drafting and revising the manuscript. All authors have read and approved the final manuscript.
This work was supported by the Key Program of Liaoning Province National Natural Science Foundation (Grant Number: 20180530102).