Clinical guidelines recommend specific drugs for type 2 diabetes (T2D), hypertension, and dyslipidemia in patients with non-alcoholic fatty liver disease (NAFLD) and/or non-alcoholic steatohepatitis (NASH). We aimed to investigate the differences in prescription trends of antidiabetic, antihypertensive, and lipid-lowering drugs among adult patients according to the presence of comorbid NAFLD and/or NASH.
MethodsWe conducted a cross-sectional analysis using a large claims database from January 2013 to December 2020.
ResultsAmong 7,716,908 people, 47,157 patients with T2D, 180,050 with hypertension, and 191,348 with dyslipidemia were identified. A total of 8,897, 16,451, and 24,762 patients with NAFLD, as well as 435, 523, and 1033 patients with NASH, had T2D, hypertension, and dyslipidemia, respectively. Among antidiabetic drugs, sodium–glucose cotransporter-2 inhibitors (SGLT2is) and thiazolidine were more frequently prescribed to patients with NAFLD than to those without NAFLD (non-NAFLD) (thiazolidine: 1.4% and 2.8% and SGLT2is: 17.8% and 25.9% for non-NAFLD and NAFLD, respectively [2019–2020]). Among antihypertensive drugs, angiotensin II receptor antagonists exhibited a slightly higher prescription ratio in patients with NAFLD (33.6% vs. 39.0%). Regarding lipid-lowering drugs, fibrates were more frequently prescribed to patients with NAFLD (10.3% vs. 18.4%).
ConclusionsSpecific drugs tended to be prescribed to patients with NAFLD. However, the differences in prescription ratios were not considerable. Further investigation is required to confirm the effects of drugs on the prognosis of patients with NAFLD or NASH.
Non-alcoholic fatty liver disease (NAFLD) is the leading cause of liver disease worldwide [1,2]; however, no drug has been approved for the treatment of NAFLD or non-alcoholic steatohepatitis (NASH), a progressive type of NAFLD. Based on evidence from clinical studies, clinical guidelines from medical societies recommend specific classes of drugs for comorbid lifestyle-related diseases. In Japan, clinical guidelines from the Japanese Society of Gastroenterology (JSGE) recommend thiazolidine for patients with NASH and comorbid type 2 diabetes (T2D) due to its beneficial effect on liver histology improvement, such as steatosis, ballooning, and lobular inflammation [3]. Vitamin E (tocopherol) is also recommended for patients with NASH, although it is not approved for the treatment of NAFLD or NASH based on its beneficial effect on biochemical parameters (alanine aminotransferase and aspartate aminotransferase) and liver histology [3]. Clinical guidelines also mention statins for patients with dyslipidemia [3–5]. JSGE clinical guidelines recommend statins due to their beneficial effect on the liver histology of patients with NASH, although corroborating evidence is limited due to the small number of patients included [3]. Clinical guidelines from the American Association for the Study of Liver Disease (AASLD) recommend statins, considering that the NAFLD population is at risk of cardiovascular diseases [5]. Among antihypertensive drugs, angiotensin II receptor antagonists are recommended for NAFLD with comorbid hypertension based on limited evidence regarding their potentially beneficial effect on serum aminotransferase levels and liver histology, including hepatic necroinflammation and fibrosis [3]. Guidelines recently published by the JSGE and Japan Society of Hepatology recommend sodium–glucose cotransporter-2 inhibitors (SGLT2is) and glucagon-like peptide-1 (GLP-1) receptor agonists due to their beneficial effect on serum aminotransferase levels and liver histology [6]. In the same guidelines, angiotensin-converting enzyme inhibitors are also recommended for hypertension, in addition to angiotensin receptor blockers (ARBs) [6]. In the clinical guidelines for T2D, hypertension, and dyslipidemia, only renin–angiotensin system inhibitors have been recommended for the reduction of NASH-induced liver fibrosis, and no specific drug treatment has been recommended for patients with NAFLD or NASH in the guidelines for T2D and dyslipidemia [7–9]. In this clinical situation where recommendations on drug use are based on clinical evidence, differences in drug prescriptions between patients with lifestyle-related diseases and comorbid NAFLD or NASH and those without these liver complications remain unclear.
Therefore, this study aimed to evaluate the differences in trends in drug use among adult patients of T2D, hypertension, and dyslipidemia according to the presence of comorbid NAFLD and/or NASH using a large Japanese administrative database.
2Methods2.1Source of dataData were extracted from a large database of health insurance claims, Mediscope®, which is developed and maintained by the Japan Medical Information Research Institute, Inc. (Tokyo, Japan) [10]. This database consists of medical, dental, dispensing, and diagnosis-procedure-combination claims data collected from 70 health insurers nationwide. The database contains information on age, sex, and all inpatient, outpatient, and pharmacy claims received from insurers. Claims included diagnoses classified according to the International Classification of Diseases, 10th Revision (ICD‐10), and prescribed medications. Dates were available for diagnosis, prescription, and dispensation.
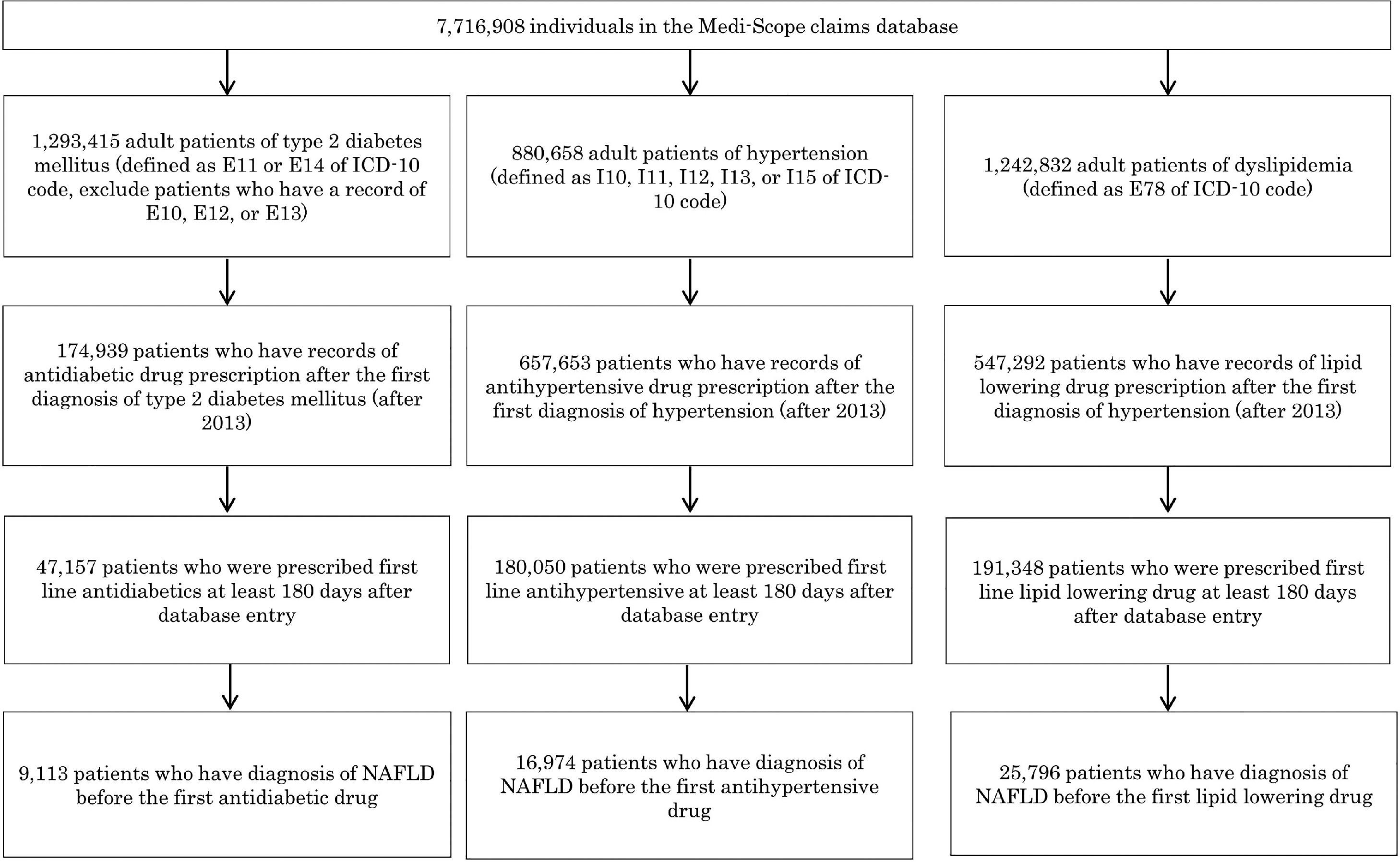
2.2PatientsThe dataset available on February 20, 2021, included 7,716,908 individuals covered by health insurers between January 2010 and December 2020. First, numbers of patients diagnosed with NAFLD (diagnosed according to K76.0 of the ICD-10 code) and/or NASH (diagnosis under other specified inflammatory liver disease; K75.8) were assessed. Prevalence of T2D (E11 and E14, excluding patients who had E10, E12, or E13 diagnoses), hypertension (I10, I11, I12, I13, and I15), and dyslipidemia (E78) in patients diagnosed with NAFLD and/or NASH was also assessed. Drug prescriptions for T2D, hypertension, and dyslipidemia were aggregated, as they were specifically mentioned in the clinical guidelines for NAFLD and NASH [3–5]. In the light of the study objective, patients over 20 years old were included. Patients had to be included in the database since equal or more than180 days prior to the initial prescription of drugs for T2D, hypertension, or dyslipidemia. In this study, we included prescription data after January 2013 because the number of individuals included in the dataset before 2012 was <25% of that after 2013 (Supplemental Fig. 1). For the analysis of drug use, patients were categorized into two groups: (1) patients without NAFLD and (2) patients with NAFLD (NAFLD and/or NASH). Patients with NAFLD were defined as those having their initial diagnosis of NAFLD or NASH before the first prescription of antidiabetic, antihypertensive, or lipid-lowering drugs. In supplemental tabulations, patients with NAFLD were further divided into two subgroups: (1) patients without a NASH diagnosis and (2) patients diagnosed with NASH. Patients diagnosed with NASH were defined as those having their initial NASH diagnosis before the first prescription of the drugs. The prevalence of diseases that potentially affect drug selection (ischemic heart disease, I20–I25; heart failure, I50; stroke, I60–I64; and chronic kidney disease, N18 and N19) was assessed in each group.
2.3Prescription of drugsDrug prescriptions after disease diagnosis (e.g., an antidiabetic drug after T2D diagnosis) were included. Patients who had records of drug prescriptions before disease diagnosis (e.g., an antidiabetic drug before T2D diagnosis) were excluded from this survey. Drug categories were identified using Anatomical Therapeutic Classification (ATC) codes. Antidiabetic drugs (ATC code: A10) included thiazolidine, SGLT2i, GLP-1 receptor agonist, dipeptidyl peptidase-4 inhibitor (DPP-4i), biguanide, α-glucosidase inhibitor (α-GI), sulfonylurea (SU), glinide, long-acting insulin, and other insulins. Antihypertensive drugs (ATC codes: C2, C3, C7, C8, and C9) included ARBs, angiotensin-converting enzyme inhibitors (ACEi), calcium-channel blockers (CCBs), beta blockers, diuretics, and other antihypertensive drugs. Lipid-lowering drugs (ATC codes: C10 and C11) included statins, fibrates, ezetimibe, tocopherol (vitamin E), and other lipid-lowering drugs. The proportion of drug prescriptions was calculated for each 2-year cycle (2013–2014, 2015–2016, 2017–2018, and 2019–2020).
2.4Data analysesThis study was a cross-sectional survey that investigated trends in drug prescriptions, and the chi-squared test was used to summarize patient characteristics and proportions of prescription drugs. All data were analyzed using R with R studio software.
2.5Ethical considerationsThis study was approved by the Institutional Review Board of Tohoku University School of Medicine on October 28, 2020 (registration number: 2020-1-667).
3Results3.1Characteristics of patients with NAFLD in the databaseThe database included 283,200 patients with NAFLD and 9,146 patients with NASH. Between 2013 and 2020, the number of patients with NAFLD increased (Table 1); however, the increase in the number of patients was in concordance with the total number of patients included in the database (Supplemental Fig. 1). The mean age of patients with NAFLD was consistent throughout the study period. The prevalence of T2D increased slightly during the study period among patients with NAFLD (Table 1).
Patient characteristics of NAFLD.
Abbreviations: NAFLD = nonalcoholic fatty liver disease, SD = standard deviation, T2D = type 2 diabetes mellitus.
A total of 47,157 eligible patients with T2D were divided into two groups: (1) 38,044 T2D patients without NAFLD (non-NAFLD group) and (2) 9,113 T2D patients with NAFLD (NAFLD group) (Fig. 1 and Table 2). The mean age and sex ratio were similar between the two groups (Fig. 1 and Table 2). The prevalence of the comorbidities was similar between the two groups, except for the prevalence of dyslipidemia, which was higher in the NAFLD group (79.2–83.9%) than in the non-NAFLD group (61.4–64.6%) (Table 2).
Characteristics of the T2D study population.
Abbreviations: NAFLD = nonalcoholic fatty liver disease, SD = standard deviation.
Among the patients with T2D, SGLT2i prescriptions increased from 2013 to 2020 (Table 3). Thiazolidine, DPP-4i, α-GI, SU, and glinide prescriptions decreased slightly. Compared to the non-NAFLD group, the NAFLD group was more likely to receive thiazolidine (4.1% and 1.4% for the non-NAFLD group and 7.3% and 2.8% for the NAFLD group in 2013–2014 and 2019–2020, respectively); nonetheless, thiazolidine prescriptions were not considerably frequent among patients with NAFLD. The NAFLD group was more likely to receive SGLT2i than the non-NAFLD group (2.5% and 17.8% for the non-NAFLD group and 3.5% and 25.3% for the NAFLD group in 2013–2014 and 2019–2020, respectively). In the NAFLD group, patients diagnosed with NASH were more likely to receive thiazolidine and SGLT2i than those without a NASH diagnosis (thiazolidine: 6.8% and 2.4% for patients without a NASH diagnosis and 20.0% and 10.6% for patients diagnosed with NASH, respectively; SGLT2i: 3.5% and 25.3% for patients without a NASH diagnosis and 7.5% and 37.1% for patients diagnosed with NASH, respectively) (Supplemental Table 3).
Prescription ratio for antidiabetic drugs in T2D study population.
| Non-NAFLD | NAFLD | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Year period | 2013-2014 | 2015-2016 | 2017-2018 | 2019-2020 | p-valuea | 2013-2014 | 2015-2016 | 2017-2018 | 2019-2020 | p-valuea |
| Drug class (%) | ||||||||||
| Thiazolidine | 4.1 | 2.4 | 1.7 | 1.4 | <0.001 | 7.3 | 3.5 | 3.0 | 2.8 | <0.001 |
| SGLT2is | 2.5 | 8.3 | 13.5 | 17.8 | <0.001 | 3.7 | 12.8 | 20.6 | 25.9 | <0.001 |
| GLP-1 receptor agonists | 0.2 | 0.1 | 0.3 | 0.4 | <0.001 | 0.3 | 0.1 | 0.4 | 0.9 | <0.001 |
| DPP-4is | 52.7 | 49.5 | 44.8 | 41.0 | <0.001 | 52.5 | 47.4 | 44.6 | 40.5 | <0.001 |
| Biguanide | 18.8 | 20.5 | 22.0 | 23.8 | <0.001 | 21.5 | 25.1 | 24.2 | 25.4 | 0.066 |
| α-GI | 9.2 | 6.3 | 4.4 | 3.9 | <0.001 | 8.7 | 5.5 | 4.8 | 3.9 | <0.001 |
| SU | 9.3 | 5.5 | 3.5 | 2.9 | <0.001 | 6.7 | 4.6 | 2.6 | 2.1 | <0.001 |
| Glinide | 1.5 | 1.1 | 0.9 | 0.8 | <0.001 | 1.5 | 0.7 | 0.6 | 0.4 | 0.001 |
| Long-acting insulin therapy | 1.8 | 2.0 | 2.4 | 2.5 | <0.001 | 1.3 | 0.9 | 1.4 | 1.2 | 0.711 |
| Other insulin therapy | 14.8 | 17.6 | 19.8 | 19.7 | <0.001 | 8.8 | 9.7 | 9.5 | 9.1 | 0.863 |
Abbreviations: α-GI = α-glucosidase inhibitors, DPP-4i = dipeptidyl peptidase-4 inhibitors, GLP-1 = glucagon-like peptide-1, NAFLD = nonalcoholic fatty liver disease, SGLT2i = sodium glucose cotransporter 2 inhibitor, SU = sulfonyl urea.
A total of 180,050 eligible patients with hypertension were divided into the following groups: (1) 163,076 non-NAFLD patients and (2) 16,974 patients with NAFLD (Fig. 1 and Table 4). The mean age was similar between the two groups (Table 4). The proportion of male patients was slightly higher in the NAFLD group than in the control group. The prevalence of T2D and dyslipidemia was higher in the NAFLD group (51.8–65.5% and 73.5–74.2%, respectively) than in the non-NAFLD group (33.9–45.2% and 39.2–43.7%, respectively) (Table 4).
Characteristics of the hypertension study population.
Abbreviations: NAFLD = nonalcoholic fatty liver disease, SD = standard deviation, T2D = type 2 diabetes.
In general, the proportion of prescriptions for each category of antihypertensive drugs was consistent throughout the study period (Table 5). The proportion of ARB prescriptions was slightly higher in the NAFLD group than in the non-NAFLD group (33.6% of the non-NAFLD group and 39.0% of the NAFLD group in 2019–2020). In the NAFLD group, the proportion of ARB prescriptions was also slightly higher among patients diagnosed with NASH than in those without a NASH diagnosis (38.8% of patients without a NASH diagnosis and 43.2% of patients diagnosed with NASH in 2019–2020) (Table 5).
Prescription ratio for antihypertensive drugs in hypertension study population.
| Non-NAFLD | NAFLD | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Year period | 2013-2014 | 2015-2016 | 2017-2018 | 2019-2020 | p-valuea | 2013-2014 | 2015-2016 | 2017-2018 | 2019-2020 | p-valuea |
| Drug class (%) | ||||||||||
| ARBs | 41.6 | 37.5 | 35.6 | 33.6 | <0.001 | 45.9 | 43.1 | 39.5 | 39.0 | <0.001 |
| ACE inhibitors | 3.2 | 3.1 | 2.7 | 2.4 | <0.001 | 4.0 | 3.5 | 3.1 | 2.5 | <0.001 |
| CCBs | 50.4 | 52.6 | 55.6 | 58.0 | <0.001 | 46.0 | 46.7 | 51.1 | 52.6 | <0.001 |
| Beta blockers | 8.1 | 8.9 | 8.4 | 8.6 | 0.623 | 7.2 | 8.0 | 8.0 | 7.7 | 0.25 |
| Diuretics | 5.2 | 5.1 | 4.8 | 5.1 | 0.236 | 5.7 | 5.1 | 4.6 | 4.3 | 0.005 |
| Others | 3.2 | 2.7 | 2.3 | 2.4 | <0.001 | 2.5 | 2.1 | 1.6 | 1.9 | 0.087 |
Abbreviations: ARB = angiotensin receptor blocker, ACE = angiotensin converting enzyme, CCB = calcium channel blocker, NAFLD = nonalcoholic fatty liver disease.
A total of 191,348 eligible patients with dyslipidemia were divided into 165,553 non-NAFLD patients and 25,796 patients with NAFLD (Fig. 1 and Table 6). The mean age was similar between the two groups (Table 6). The ratio of male patients was slightly higher in the NAFLD group. The prevalence of T2D was higher in the NAFLD group (52.3–66.0%) than in the non-NAFLD group (39.0–52.0%) (Table 6).
Characteristics of the dyslipidemia study population.
Abbreviations: NAFLD = nonalcoholic fatty liver disease, SD = standard deviation, T2D = type 2 diabetes.
In general, the proportion of prescriptions for each category of lipid-lowering drugs was consistent throughout the study period (Table 7). The NAFLD group was more likely to receive fibrates than the non-NAFLD group (9.4% and 10.3% for the non-NAFLD group and 15.0% and 18.4% for the NAFLD group in 2013–2014 and 2019–2020, respectively). Conversely, the NAFLD group was less likely to receive statins than the non-NAFLD group (77.3% and 78.9% for the non-NAFLD group and 70.0% and 70.0% for the NAFLD group in 2013–2014 and 2019–2020, respectively), although the difference in proportions of prescriptions was not considerably vast. In the NAFLD group, patients diagnosed with NASH were more likely to receive tocopherol (vitamin E) than those without a NASH diagnosis (5.4% and 5.8% for patients without a NASH diagnosis and 13.0% and 18.0% for patients diagnosed with NASH in 2013–2014 and 2019–2020, respectively) (Supplemental Table 7). Patients diagnosed with NASH were less likely to receive statins than those without a NASH diagnosis (70.6% and 70.8% for patients without a NASH diagnosis and 47.8% and 53.0% for patients diagnosed with NASH in 2013–2014 and 2019–2020, respectively) (Supplemental Table 7).
Prescription ratio for lipid lowering drugs in dyslipidemia study population.
| Non-NAFLD | NAFLD | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Year period | 2013-2014 | 2015-2016 | 2017-2018 | 2019-2020 | p-valuea | 2013-2014 | 2015-2016 | 2017-2018 | 2019-2020 | p-valuea |
| Drug class (%) | ||||||||||
| HMG-CoA reductase inhibitors | 77.3 | 77.9 | 77.6 | 79.1 | <0.001 | 70.0 | 69.5 | 69.8 | 70.0 | 0.687 |
| Fibrates | 9.6 | 9.1 | 9.3 | 10.3 | <0.001 | 15.0 | 15.6 | 15.2 | 18.4 | <0.001 |
| Ezetimibe | 3.7 | 3.2 | 3.2 | 3.1 | <0.001 | 6.8 | 5.2 | 4.8 | 4.3 | <0.001 |
| Tocopherol | 6.5 | 6.3 | 6.6 | 5.3 | <0.001 | 5.6 | 5.9 | 6.9 | 6.4 | 0.192 |
| Others | 4.1 | 4.8 | 4.6 | 3.8 | <0.001 | 4.3 | 5.3 | 5.6 | 3.6 | 0.002 |
Abbreviations: HMG-CoA = hydroxymethylglutaryl-CoA, NAFLD = nonalcoholic fatty liver disease.
To the best of the authors’ knowledge, this is the first study to evaluate the differences in prescription trends of drugs for adult patients of T2D, hypertension, and dyslipidemia according to the presence of comorbid NAFLD using a large Japanese administrative database. Prescription patterns of specific drugs among non-NAFLD patients were consistent with those among patients with NAFLD, with modest differences. Among patients with NAFLD, differences in prescription patterns were observed between patients with and those without a NASH diagnosis. These differences appeared to be influenced by the recommendations in the clinical guidelines for NAFLD and NASH. Notably, the number of patients with NASH was relatively modest.
Thiazolidine presents certain concerns regarding its safety, such as weight gain, fracture, heart failure, and bladder cancer [11–14]. In the non-NAFLD and NAFLD groups, decreasing trends in thiazolidine prescriptions were observed. In the NAFLD group, a higher proportion of thiazolidine prescriptions was observed in patients diagnosed with NASH than in those without a NASH diagnosis, although this drug did not have the highest proportion of prescriptions among patients with T2D and comorbid NASH. The proportion of thiazolidine prescriptions in patients diagnosed with NASH was maintained throughout the study period. It was suggested that thiazolidine maintained its proportion of prescriptions among patients diagnosed with NASH due to evidence regarding its beneficial effect on NAFLD and NASH [3,15–17]. The proportion of SGLT2i prescriptions increased throughout the study period in all groups, which is consistent with a previous study reporting the trends of antidiabetic drugs in Japan [18]. A higher proportion of prescriptions was observed in the NAFLD group than in the non-NAFLD group, especially after 2015. In the NAFLD group, the proportion of SGLT2i prescriptions was even higher in patients diagnosed with NASH than in those without a NASH diagnosis, though a definitive assessment of the 2013–2014 period was difficult due to the small number of patients diagnosed with NASH. SGLT2is were recently mentioned in the clinical guidelines issued in 2020 [6]; however, a higher proportion of prescriptions was observed before the issuance of these guidelines. Several studies have reported the efficacy of SGLT2i in NAFLD or NASH after 2016 [19–22]. However, these studies provided limited evidence to support the efficacy of SGLT2is due to their study designs (open-label, single-arm, and limited-sample-sized studies or those with endpoints such as ALT or AST), and valid evidence regarding the beneficial effect of SGLT2is on the prognosis of patients with NAFLD or NASH has not been demonstrated. A recent study in Japan reported that SGLT2is were more frequently prescribed to patients with T2D with higher body mass indexes (BMIs), and hemoglobin A1c and BMI were independently associated with SGLT2i prescriptions [23]. A relatively high BMI has been reported as a risk factor for fatty liver. In addition, among patients with diabetes, those with relatively high liver fat content have reportedly been associated with a higher BMI [24]. A relatively high BMI and NAFLD are known risk factors for cardiovascular diseases, and SGLT2is reduce the risk of cardiovascular diseases [25–27]. It is possible that SGLT2is were selected for patients with NAFLD who had higher BMIs.
Among antihypertensive drugs, ARBs were the only drugs recommended for patients with NAFLD/NASH and comorbid hypertension. Some studies have demonstrated the efficacy of ARBs on biochemical parameters and liver histology [28–31]. Both clinical evidence and clinical-guideline recommendations have been suggested to influence a higher proportion of drug prescriptions in the NAFLD group. However, the difference in the proportion of ARB prescriptions between the non-NAFLD and NAFLD groups was not vast. Among antihypertensive drugs, CCBs have predominantly been used; however, the popularity of ARB use is approaching that of CCBs [32]. It has been suggested that ARBs are used regardless of the presence of NAFLD. In addition, clinical evidence appears to suggest that the beneficial effect of selecting ARBs is less robust than that of CCBs.
In the non-NAFLD and NAFLD groups, statins were the most frequently prescribed drugs, with an enormous difference in prescription proportion compared to other drug classes. Accumulating clinical evidence reveals that the lowering of low-density lipoprotein cholesterol by statins potentially mitigates the risk of atherosclerotic cardiovascular diseases [7]. Although statins are recommended for patients with NAFLD and NASH based on their efficacy in both liver function improvement and prevention of cardiovascular disease, which is reportedly a high risk in patients with NAFLD, [3–5] a lower proportion of statin prescriptions was observed in the NAFLD group than in the non-NAFLD group, especially among patients with NASH. Statins are considered to be associated with the risk of liver injury [5]. In package inserts of statins, it is noted that statins should be avoided in patients with a history of liver injury, and they pose a risk of worsening liver injury in patients with cirrhosis. The prevalence of cirrhosis was notably higher in the NAFLD group, especially in patients diagnosed with NASH than in the non-NAFLD group (Supplemental Table 8); thus, this was suggested to be the cause of the relatively lower proportion of prescriptions in the NAFLD group. Another hypothesis is that statins were avoided in patients with NAFLD due to their risk of liver injury, although in the AASLD NAFLD/NASH clinical guidelines, it is noted that the risk of severe liver injury is rare among patients with NASH [5]. Remarkably, fibrates, which are not mentioned in the guidelines, exhibited a higher proportion of prescriptions in the NAFLD group than in the non-NAFLD group. Fibrates are recommended for hypertriglyceridemia. In some reports, patients with NAFLD (or fatty liver) have been reported to exhibit higher serum triglyceride levels than those without NAFLD [33,34]. Relatively high serum triglyceride levels and hypertriglyceridemia prevalence might have been the causes of the higher proportion of fibrate prescriptions in the NAFLD group. In patients diagnosed with NASH, the proportion of vitamin E prescriptions was notably higher than that in patients without a NASH diagnosis. In Japan, vitamin E has not been approved for the treatment of NASH, though it is recommended by clinical guidelines. Apparently, in this clinical setting, vitamin E was likely to be selected as the treatment option in patients with dyslipidemia and comorbid NASH compared to those without a NASH diagnosis. However, the proportion of vitamin E prescriptions was not predominant in patients diagnosed with NASH and comorbid dyslipidemia.
This study has certain limitations. First, the study utilized data from administrative claims, which did not include the clinical status of patients, such as vitals and laboratory parameters. Therefore, we could not assess important confounding factors, including the severity of T2D, hypertension, and dyslipidemia. Differences in the severity of these diseases among the three tested groups are possible. In addition, the database was based on claims to employee health insurers; thus, it mainly comprised the working-age population and had limited data regarding the older population. It has been reported that there are many patients with NAFLD in Japan, especially women [35,36]. This population imbalance included in the database might have affected the generalizability of our findings. Finally, it is apparent that patients who were clinically diagnosed with NAFLD or NASH did not necessarily have a disease code for NAFLD or NASH, thus causing underdiagnosis within the database. Sixty-two percent of patients with newly diagnosed diabetes have reportedly been associated with NAFLD in a study with subjects who underwent health checkups [37]. In this study, patients with T2D and comorbid NAFLD/NASH merely constituted 24% of the study population (Fig. 1), which is smaller than that reported previously [37]. Furthermore, considering the estimated NAFLD prevalence of approximately 18% in Japan [38], the number of patients in the NAFLD and/or NASH groups also appeared smaller than that in the non-NAFLD/non-NASH groups among those with both hypertension and dyslipidemia (Fig. 1). In addition, based on estimates of NAFLD's natural history, in which approximately 25% of the NAFLD population progresses to NASH, the number of patients in the NASH group also appeared smaller. NAFLD and NASH are not adequately diagnosed in claims, possibly because there is no drug approved for the treatment of these liver diseases. It is further apparent that only a small population was diagnosed with NASH among patients with NAFLD because NASH diagnosis requires an invasive biopsy, and the treatment for patients diagnosed with NASH emphatically differs from that for patients without a NASH diagnosis. Therefore, the sensitivity of NASH diagnosis within the database was expectedly low. This potentially renders it difficult to accurately estimate the proportion of prescriptions for patients who are clinically diagnosed with NAFLD and NASH.
5ConclusionIn conclusion, this study provides information regarding the trends in drug prescriptions for T2D, hypertension, and dyslipidemia in patients with NAFLD. Further accumulation of clinical evidence regarding drugs used for T2D, hypertension, and dyslipidemia in terms of their beneficial effects on NAFLD and NASH is anticipated, especially their long-term benefits on the prognosis of patients. Such evidence potentially influences the appropriate management of NAFLD and NASH with comorbid lifestyle-related diseases.
FoundingThis research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of interestToshiya Machida is an employee of Pfizer R&D, Japan. Makoto Miyazaki is an employee of MSD K.K.. Toshiya Machida and Makoto Miyazaki are also research collaborators at Tohoku University, and they contributed to the present study independently of the companies they belong to. Nariyasu Mano received honoraria from Daiichi Sankyo Co., Ltd. The other authors declare no conflicts of interest.
The authors would like to thank JMIRI, Inc., who provided the claims database.