Methylmalonic acidemia is an autosomal recessive disease, caused in 60% of cases by mutations in the MUT gene. The lack of l-methylmalonyl-CoA mutase leads to the accumulation of toxic compounds due to a metabolic block, leading to progressive encephalopathy and hyperammonemia that can end in death. Carrier couples are asymptomatic, and this disorder is not always included in the neonatal screening panel in Spain. Different reproductive options were explained to a couple with a previous affected son, and preimplantation genetic diagnosis was chosen. Both of them were carriers in heterozygosis of the same mutation c.671_678dup (p.V227fs) in exon 3 of the MUT gene.
Material and methodsFifteen short tandem repeat markers closely linked to the MUT gene were tested, and the mutation c.671_678dup was detected by performing multiplex fluorescent heminested polymerase chain reaction.
ResultsEight short tandem repeat markers were informative or partially informative, and the closest downstream and upstream to the MUT gene were selected in order to establish the disease-bearing haplotype. Nine blastomeres from nine embryos were analyzed, and one single embryo was transferred, resulting in a healthy birth.
DiscussionCustomized direct preimplantation genetic diagnosis is a valid reproductive alternative to avoid transmission of methylmalonic acidemia to offspring. The ad hoc protocol designed for this couple allowed the birth of a healthy boy.
La acidemia metilmalónica es una enfermedad autosómica recesiva causada por mutaciones en el gen MUT en el 60% de los casos. La deficiencia en la enzima L-metilmalonil-CoA mutasa provoca la acumulación de compuestos tóxicos debido a un bloque del metabolismo, produciendo una encefalopatía progresiva e hiperamonemia, que puede provocar la muerte. Las parejas portadoras son asintomáticas y esta enfermedad no está incluida en el cribado neonatal ampliado en España. Diferentes opciones reproductivas fueron explicadas a una pareja con un hijo previo afectado de acidemia metilmalónica, decidiendo realizar diagnóstico genético preimplantación. Ambos miembros de la pareja eran portadores en heterocigosis de la misma mutación, c.671_678dup (p.V227fs) en el exón 3 del gen MUT.
Material y métodosQuince marcadores microsatélite ligados al gen MUT y la mutación c.671_678dup fueron amplificados mediante reacción en cadena de la polimerasa multiplex semi anidada y fluorescente.
ResultadosOcho marcadores microsatélite resultaron informativos o parcialmente informativos y los más cercanos al gen fueron seleccionados para establecer el haplotipo portador de la mutación. Nueve blastómeros de nueve embriones fueron analizados y un solo embrión fue transferido, resultando en un nacimiento libre de la enfermedad.
DiscusiónEl diagnóstico genético preimplantación directo y personalizado es una alternativa reproductiva válida para evitar la transmisión de la acidemia metilmalónica a la descendencia. El protocolo ad hoc diseñado para esta pareja permitió el nacimiento de un niño sano.
It is usual that couples at risk of transmission of inherited metabolic diseases request reproductive options in order to have healthy offspring. Prenatal diagnosis (PND) based on amniocentesis or chorionic villus sampling analysis has traditionally been chosen. The development of assisted reproduction technologies offered new strategies as gamete donation from a studied donor. Nowadays, in vitro fertilization (IVF) combined with preimplantation genetic diagnosis (PGD) of embryos is well established, in order to select healthy embryos to transfer to the maternal uterus. It was first applied in 1990 (Handyside et al., 1990) as an alternative to PND and termination of pregnancy. The number of cases and diseases tested increases year by year (Moutou et al., 2014) and several strategies have been deeply reviewed (Van der Aa et al., 2013; Harper and SenGupta, 2012; Vendrell and Bautista-Llácer, 2012).
Branch-chained organic acidemias (OA) are paradigmatic examples for the personalized PGD. OA are life-threatening recessive disorders with a very low prevalence, and newborns may die before the condition has even been fully diagnosed. Furthermore, universal neonatal screening usually does not cover all the subtypes of OA. In this scenario, standardization of preventive genetic treatments is difficult. A customized reproductive protocol based on PGD is a rapid, direct, secure and cost-effective strategy. In this particular case, we present a family with an affected child of isolated methylmalonic acidemia due to l-methylmalonyl-CoA mutase deficiency (mut MMA; OMIM #251000). This is one of the most frequent forms of branch-chained organic acidemias, caused by mutations in the MUT gene (OMIM *609058) located in chromosome 6p12.3. The lack of the enzyme l-methylmalonyl-CoA mutase (MCM) provokes the accumulation of methylmalonic acid in blood and urine, resulting from the failure to convert methylmalonyl-CoA into succinyl-CoA during propionyl-CoA metabolism in the mitochondria. This metabolic blockage leads to progressive encephalopathy and hiperammonemia (Manoli and Venditti, 2005). Patients with mut MMA have been divided into two subgroups: mut° with no MCM activity and mut− with MCM residual activity in cells. Patients with the mut° type present the most acute phenotype, whereas mut− patient's symptoms are less severe and variable (Martinez et al., 2005). The real mut MMA prevalence remains unclear, as rates reported may include cases of other forms of methylmalonic acidemia (Manoli and Venditti, 2005). If not identified and treated on time, mut MMA could lead to coma and death. Carriers of mut MMA are aware of their condition after the birth of an affected son.
The aim of this case report is to show a personalized PGD protocol based on direct mutation detection on single cells biopsied from six-to-eight cells preimplantation embryos. To our knowledge, this is the first report of a successful application of PGD for mut MMA.
Materials and methodsFamily historyThe couple formed by a non-consanguineous 33 years old female and a 36 years old male attended to comprehensive preconception genetic counselling. They had an affected child of mut° MMA, carrying the mutation c.671_678dup (p.Val227fs) in homozygosis in exon 3 of the MUT gene. This frameshift mutation produces a change of the Valine residue in aminoacid 227, resulting in a premature stop codon at the protein level. The origin of this allelic variant has been previously reported in Spanish patients from the Basque country (Martinez et al., 2005), region where the couple comes from. The onset of the clinical phenotype in homozygous patients for this particular mutation is during the neonatal period, causing dead after a short period of time. The couple did not have any previous genetic study. However, as in an autosomal recessive disease, the parents are obligate asymptomatic carriers. After explaining the complete PGD process and answering their queries, the couple signed an informed consent form for PGD testing.
Preclinical informativity studyThe most important limitations in PGD for single gene disorders are the amount of DNA in a single cell, of about 6pg, and the impossibility of eventual re-analysis of the embryonic single cell. For these reasons, a preclinical design and validation on single cells (lymphocytes) is compulsory prior to performing the IVF cycle (Harton et al., 2011). The preclinical work-up has two main objectives: (i) to confirm the disease-causing mutation in the affected patient and both members of the couple and (ii) to find the informative polymorphic short tandem repeat (STR) markers linked to the gene, in order to establish de disease-bearing haplotypes and support the direct detection of the mutation. Moreover, polymerase chain reaction (PCR) amplification efficiency and allelic drop-out (ADO: the random non-amplification of one of the alleles in a heterozygous sample) rates on single cells are evaluated as the same optimized conditions are expected on blastomeres (Vendrell and Bautista-Llácer, 2012).
Preclinical studies and lymphocyte validation were performed prior to the IVF cycle, following recommended international guidelines on PGD (Harton et al., 2011) and the norm UNE-EN ISO 9001, 2000 (Vendrell et al., 2009).
Blood samples were requested from both members of the couple and buccal swabs from the affected son were provided. DNA extraction and PCR protocols were performed as previously published (Alberola et al., 2009). PCR was first performed on genomic DNA to confirm the mutation and to determine the informative STR markers, then on 1ng of diluted genomic DNA in a heminested multiplex PCR with the informative STR markers and finally on single lymphocytes collected from both parents with the same PCR conditions that worked for 1ng of genomic DNA. Oligonucleotides were tested in order to discard possible human DNA contamination.
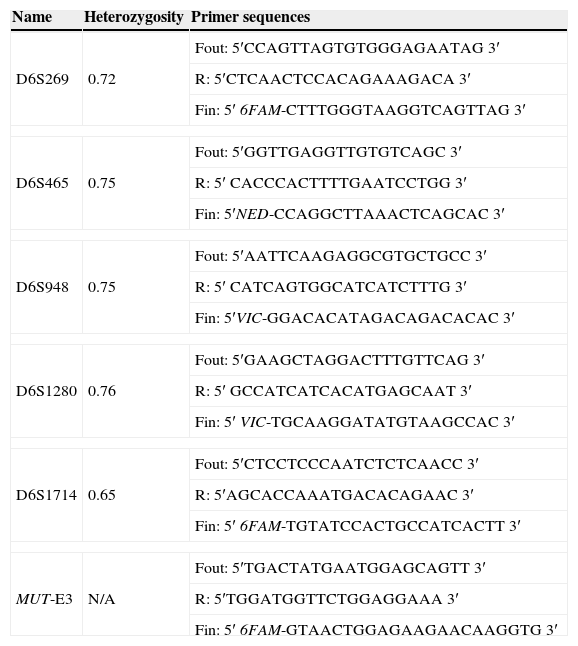
Fifteen STR markers closely linked to the MUT gene were tested. Single cells were isolated and lysed as previously described (Cui et al., 1989). The molecular analysis protocol was the same for lymphocytes (pre clinical set-up) and blastomeres (clinical PGD cycle). PCR amplification was performed using the outer and inner oligonucleotide primers listed in Table 1. First round multiplex PCR containing the external primers was carried out in a total volume of 25μL containing 200μM dNTP, 1.5mM MgCl2, 1× PCR buffer, 1 unit Go-Taq Hot Start polymerase (Promega, USA) and 0.4μM each of the outer forward and reverse primers. One microlitre from the first round PCR was used as template for separate second round PCR reactions with fluorescently labelled inner forward and reverse primers in a total volume of 10μL. PCR reactions were undertaken using a TProffesional standard Thermocycler (Biometra, Germany). Seventeen thermal cycles were performed for the outer primers and 30 for the inner primers at 95°C for 1min, 55°C for 45s, and 72°C for 1min. The cycling was preceded by a 5min initial denaturation step at 95°C and ended with an additional elongation step of 7min at 72°C. Amplified inner products were electrophoresed in an automated genetic analyser 3730xl (Applied Biosystems, USA). Results were analyzed with the Genemapper v3.7 software (Applied Biosystems, USA).
Primer sequences for the STR markers used in the preimplantation genetic diagnosis of methylmalonic acidemia.
| Name | Heterozygosity | Primer sequences |
|---|---|---|
| D6S269 | 0.72 | Fout: 5′CCAGTTAGTGTGGGAGAATAG 3′ |
| R: 5′CTCAACTCCACAGAAAGACA 3′ | ||
| Fin: 5′ 6FAM-CTTTGGGTAAGGTCAGTTAG 3′ | ||
| D6S465 | 0.75 | Fout: 5′GGTTGAGGTTGTGTCAGC 3′ |
| R: 5′ CACCCACTTTTGAATCCTGG 3′ | ||
| Fin: 5′NED-CCAGGCTTAAACTCAGCAC 3′ | ||
| D6S948 | 0.75 | Fout: 5′AATTCAAGAGGCGTGCTGCC 3′ |
| R: 5′ CATCAGTGGCATCATCTTTG 3′ | ||
| Fin: 5′VIC-GGACACATAGACAGACACAC 3′ | ||
| D6S1280 | 0.76 | Fout: 5′GAAGCTAGGACTTTGTTCAG 3′ |
| R: 5′ GCCATCATCACATGAGCAAT 3′ | ||
| Fin: 5′ VIC-TGCAAGGATATGTAAGCCAC 3′ | ||
| D6S1714 | 0.65 | Fout: 5′CTCCTCCCAATCTCTCAACC 3′ |
| R: 5′AGCACCAAATGACACAGAAC 3′ | ||
| Fin: 5′ 6FAM-TGTATCCACTGCCATCACTT 3′ | ||
| MUT-E3 | N/A | Fout: 5′TGACTATGAATGGAGCAGTT 3′ |
| R: 5′TGGATGGTTCTGGAGGAAA 3′ | ||
| Fin: 5′ 6FAM-GTAACTGGAGAAGAACAAGGTG 3′ | ||
Fout, forward outside primer; Fin, forward inside primer; R, reverse primer; N/A, not applicable.
GeneLoc: Exon-based integration of human genome maps. Bioinformatics 19(S1). URL: http://genecards.weizmann.ac.il/geneloc.
A PGD working scheme includes, in all cases, an IVF cycle in order to generate embryos in the laboratory. The woman received a routine superovulation procedure based on the antagonist protocol with Puregon (Merck Sharp & Dohme de España S.A, Spain) for 10 days, Decapeptyl (IPSEN PHARMA S.A, USA) two days before oocyte retrieval, and Orgalutran (Merck Sharp & Dohme de España S.A, Spain) for 5 days after 5 days of Puregon treatment. Twenty mature oocytes were retrieved, and thirteen two-pronucleus zygotes were produced after intracytoplasmic sperm injection (ICSI). Nine evolutionary embryos were cryopreserved at four-cell stage, 48h after ICSI (day +2), in order to transfer in a non-stimulated endometrium. In the following menstrual cycle, endometrial preparation was done with oestrogen and progesterone administered in a sequential regime. After thawing, nine embryos survived and nine cleaving embryos were available for biopsy on day +3. Only one blastomere was obtained per embryo. Biopsy procedures were performed according to previously described protocols (Alberola et al., 2009).
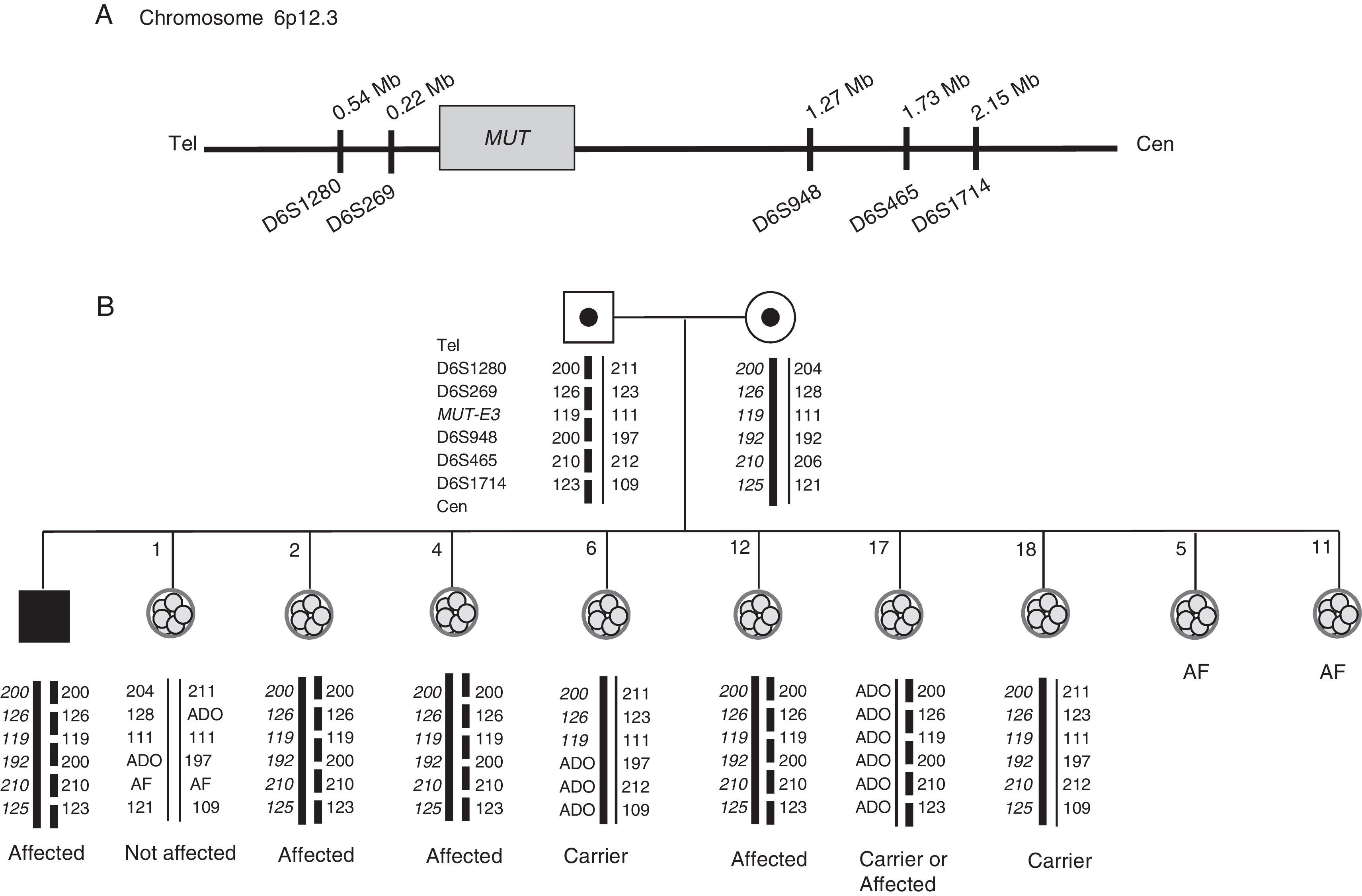
ResultsOne STR marker resulted informative, and six partially informative. Consequently, the closest markers upstream and downstream of the gene were chosen: D6S1280, D6S269, D6S948, D6S465 and D6S1714 (Fig. 1A). Once the diagnosis strategy to detect the c.671_678dup (p.V227fs) mutation in the MUT gene was established, validation of the technique was carried out as described elsewhere (Alberola et al., 2011; Vendrell et al., 2011). Amplification failure and ADO rates for each PCR amplicon were estimated. ADO rates were 0% (0/25) for c.671_678dup mutation and each STR marker (0/50). PCR efficiency for each amplicon was 100% (50/50).
Preimplantation genetic diagnosis (PGD) of methylmalonic acidemia. (A) Position of the short tandem repeat markers with respect to the MUT gene on chromosome 6. (B) Pedigree showing haplotypes of the family members involved in the preclinical work-up and embryos generated in the PGD cycle. The thick black line represents the maternal disease-bearing haplotype. The discontinuous line represents the paternal disease-bearing haplotype. The maternal disease-bearing alleles are shown in bold and italics; the paternal disease-bearing alleles are displayed in bold. AF: amplification failure; ADO: allele drop-out; cen: centromere; tel: telomere; Mb: megabases.
Nine embryos were biopsied obtaining nine single blastomeres (Fig. 1B). Amplification failed in two blastomeres. Total ADO and amplification efficiency rates were 26.8% (11/41) and 97.6% (41/42) respectively. All embryos were successfully diagnosed except embryo number 17: as there was ADO of the maternal haplotype and the disease-bearing paternal haplotype was present, transfer was not recommended.
Six embryos reached the blastocyst stage and embryo number 1 was transferred. A single pregnancy was achieved, resulting in the birth of a healthy boy.
DiscussionIn this case-report we show, for the first time, the birth of a healthy boy after the application of an ad-hoc protocol for mut MMA on single cells from preimplantation embryos. Nowadays, PGD is a well-established reproductive option for couples genetically at-risk and it has extended worldwide, depending on the countries’ regulations. Serious recessive disorders are specially indicated, where couples are only aware of their carrier status when children show symptoms and are diagnosed, as they are asymptomatic. Concerning mut MMA, and particularly the mutation the couple carry, newborns die shortly after birth (Martinez et al., 2005). Furthermore, mut MMA and other OA are not included in the reduced neonatal screening panels. In Spain, neonatal screening schemes cover a different number of genetic disorders, varying from 2–3 to 20 diseases depending on the region. The Interregional Counsel of the National Health System approved in July of 2013 the creation of a unique newborn screening panel, including seven diseases: congenital hypothyroidism, phenylketonuria, cystic fibrosis, medium-chain acyl-CoA dehydrogenase deficiency, long-chain 3-OH acyl-CoA dehydrogenase deficiency, glutaric acidemia and sickle cell anaemia (https://www.msssi.gob.es/profesionales/saludPublica/prevPromocion/cribadoNeonatal.htm). However, mut MMA is not included in this panel. In this case, the mut° phenotype of MMA shows the most severe form of the disease, with poor prognosis. Besides, patients with this phenotype are nonresponsive to B12 treatment. For these reasons, PGD is an effective reproductive option for couples wishing to have healthy offspring.
Specifically, isolated MMA is a very rare recessive condition with a variable prevalence across countries, being 1/48,000–1/61,000 in North America, 1/26,000 in China, 1/115,000 in Italy or 1/169,000 in Germany (reviewed by Manoli and Venditti, 2005). Genetically, it is a polygenic disorder with five candidate genes described to date and different phenotypes. Mutations in the MUT gene cause complete (mut0 enzymatic subtype) or partial (mut− enzymatic subtype) deficiency of the enzyme methylmalonyl-CoA mutase. Reduced synthesis of its cofactor 5′-deoxyadenosylcobalamin, associated with cblA, cblB, or cblD-variant 2 complementation groups are caused by mutations in the MMAA, MMAB, and MMADHC genes, respectively. Also, deficient activity of methylmalonyl-CoA epimerase is encoded by the MCEE gene. This variable phenotypic spectrum is the reason why clinical neonatal management is crucial in order to orientate the molecular genetic diagnosis.
In the context of low prevalent disorders as mut MMA, our first-choice method is always a personalized PGD protocol. Ad hoc PGD protocols allow a rapid, robust and cost-effective detection of mutations directly on single blastomeres, genotyping the embryos and detecting eventual recombination, ADO and contamination by simultaneous STR-based haplotyping by fluorescent multiplex PCR. Furthermore, this approach has been recently evaluated by the ESHRE PGD Consortium as a valid, robust and of a high diagnostic value (Dreesen et al., 2014).
Blastomere ADO rates in this cycle (26.8%) were not in line with ADO rates obtained in the preclinical study with lymphocytes (0%). Amplification efficiency in blastomeres and lymphocytes was not as different (97.6% and 100% respectively). However, it has been previously published that ADO rates and amplification efficiency may vary depending on the specimen tested, and what causes ADO remains unclear (Thornhill et al., 2005; Wilton et al., 2009; Bautista-Llácer et al., 2010). Moreover, two embryos failed to amplify, probably due to DNA degradation or absence of nucleus. Embryo number 17 could not be diagnosed due to absence of the maternal haplotype. This could be due to a monosomy in chromosome 6, although further studies would be necessary to prove it. In this sense, combined studies of monogenic disease plus aneuploidy would help to elucidate what is really ADO and what is a true chromosome loss (unpublished observations).
This strategy, ad hoc protocols plus direct mutation detection, represents clear benefits versus haplotyping: (i) it decreases the possibility of adverse misdiagnosis in cases where the disease-bearing haplotypes are wrongly assigned (Altarescu et al., 2008), where a few STR markers are available, or when they are poorly distributed along the region of study (Wilton et al., 2009) and (ii) it allows to perform PGD for non-informative couples, when family members are not available in the informativity study or carriers of de novo mutations. Several strategies have been described based on direct genotyping by studying single spermatozoa in the carrier male or polar body in the female, in order to establish the disease-bearing haplotype (Rechitsky et al., 2011).
In summary, customized direct PGD is a valid reproductive alternative to avoid transmission of mut MMA to offspring. The ad hoc protocol designed for this couple allowed the birth of a healthy boy. The application of different mutation-based direct methodologies allows increasing the number of diseases for which PGD is possible. However, the important challenge of direct analysis is to know the mutation responsible for the disorder. This is complicated in disorders of very low prevalence or in polygenic diseases. In this sense, accurate preconception genetic counselling is highly recommended in couples at risk of transmitting genetic diseases to offspring.
Conflicts of interestNone declared.